AHC0881
antibody from Invitrogen Antibodies
Targeting: CXCL8
3-10C, AMCF-I, b-ENAP, GCP-1, GCP1, IL-8, IL8, K60, LECT, LUCT, LYNAP, MDNCF, MONAP, NAF, NAP-1, NAP1, SCYB8, TSG-1
Antibody data
- Antibody Data
- Antigen structure
- References [17]
- Comments [0]
- Validations
- Western blot [1]
- Other assay [8]
Submit
Validation data
Reference
Comment
Report error
- Product number
- AHC0881 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IL-8 (CXCL8) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Recombinant full-length protein
- Description
- Centrifuge vial prior to opening. Reconstitute in sterile water to 0.1-1.0 mg/mL.
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µg
- Storage
- -20°C
Submitted references Combined application of geranylgeranylacetone and amelogenin promotes angiogenesis and wound healing in human periodontal ligament cells.
Toll-Like Receptor 3 as a Recurrence Risk Factor and a Potential Molecular Therapeutic Target in Colorectal Cancer.
Prognostic Significance of Lymphocyte Infiltration and a Stromal Immunostaining of a Bladder Cancer Associated Diagnostic Panel in Urothelial Carcinoma.
Immunohistochemical analysis of IL-6, IL-8/CXCR2 axis, Tyr p-STAT-3, and SOCS-3 in lymph nodes from patients with chronic lymphocytic leukemia: correlation between microvascular characteristics and prognostic significance.
Validation and clinicopathologic associations of a urine-based bladder cancer biomarker signature.
The role of CXC-chemokine receptor CXCR2 and suppressor of cytokine signaling-3 (SOCS-3) in renal cell carcinoma.
Immunohistological pointers to a possible role for excessive cathelicidin (LL-37) expression by apocrine sweat glands in the pathogenesis of hidradenitis suppurativa/acne inversa.
Interleukin-8, a promising predictor for prognosis of pancreatic cancer.
Stress effects on FosB- and interleukin-8 (IL8)-driven ovarian cancer growth and metastasis.
Cytokines and growth factors expressed by human cutaneous melanoma.
Modification of the primary tumor microenvironment by transforming growth factor alpha-epidermal growth factor receptor signaling promotes metastasis in an orthotopic colon cancer model.
Clinical and biological significance of vascular endothelial growth factor in endometrial cancer.
Induction of CXC chemokines in A549 airway epithelial cells by trypsin and staphylococcal proteases - a possible route for neutrophilic inflammation in chronic rhinosinusitis.
Monocyte/macrophage and T-cell infiltrates in peritoneum of patients with ovarian cancer or benign pelvic disease.
Specific C-terminal cleavage and inactivation of interleukin-8 by invasive disease isolates of Streptococcus pyogenes.
Modulation of bone microenvironment with zoledronate enhances the therapeutic effects of STI571 and paclitaxel against experimental bone metastasis of human prostate cancer.
Differential expression of interleukin-8 and its receptors in the neuroendocrine and non-neuroendocrine compartments of prostate cancer.
Yamato H, Sanui T, Yotsumoto K, Nakao Y, Watanabe Y, Hayashi C, Aihara R, Iwashita M, Tanaka U, Taketomi T, Fukuda T, Nishimura F
Journal of cellular biochemistry 2021 Jul;122(7):716-730
Journal of cellular biochemistry 2021 Jul;122(7):716-730
Toll-Like Receptor 3 as a Recurrence Risk Factor and a Potential Molecular Therapeutic Target in Colorectal Cancer.
Yoshida T, Miura T, Matsumiya T, Yoshida H, Morohashi H, Sakamoto Y, Kurose A, Imaizumi T, Hakamada K
Clinical and experimental gastroenterology 2020;13:427-438
Clinical and experimental gastroenterology 2020;13:427-438
Prognostic Significance of Lymphocyte Infiltration and a Stromal Immunostaining of a Bladder Cancer Associated Diagnostic Panel in Urothelial Carcinoma.
Furuya H, Chan OTM, Hokutan K, Tsukikawa Y, Chee K, Kozai L, Chan KS, Dai Y, Wong RS, Rosser CJ
Diagnostics (Basel, Switzerland) 2019 Dec 28;10(1)
Diagnostics (Basel, Switzerland) 2019 Dec 28;10(1)
Immunohistochemical analysis of IL-6, IL-8/CXCR2 axis, Tyr p-STAT-3, and SOCS-3 in lymph nodes from patients with chronic lymphocytic leukemia: correlation between microvascular characteristics and prognostic significance.
Levidou G, Sachanas S, Pangalis GA, Kalpadakis C, Yiakoumis X, Moschogiannis M, Sepsa A, Lakiotaki E, Milionis V, Kyrtsonis MC, Vassilakopoulos TP, Tsirkinidis P, Kontopidou F, Kokoris S, Siakantaris M, Angelopoulou M, Papadaki H, Kavantzas N, Panayiotidis P, Patsouris E, Korkolopoulou P
BioMed research international 2014;2014:251479
BioMed research international 2014;2014:251479
Validation and clinicopathologic associations of a urine-based bladder cancer biomarker signature.
Zhang G, Gomes-Giacoia E, Dai Y, Lawton A, Miyake M, Furuya H, Goodison S, Rosser CJ
Diagnostic pathology 2014 Nov 12;9:200
Diagnostic pathology 2014 Nov 12;9:200
The role of CXC-chemokine receptor CXCR2 and suppressor of cytokine signaling-3 (SOCS-3) in renal cell carcinoma.
Stofas A, Levidou G, Piperi C, Adamopoulos C, Dalagiorgou G, Bamias A, Karadimou A, Lainakis GA, Papadoukakis S, Stravodimos K, Dimopoulos MA, Patsouris E, Gakiopoulou H, Korkolopoulou P
BMC cancer 2014 Mar 4;14:149
BMC cancer 2014 Mar 4;14:149
Immunohistological pointers to a possible role for excessive cathelicidin (LL-37) expression by apocrine sweat glands in the pathogenesis of hidradenitis suppurativa/acne inversa.
Emelianov VU, Bechara FG, Gläser R, Langan EA, Taungjaruwinai WM, Schröder JM, Meyer KC, Paus R
The British journal of dermatology 2012 May;166(5):1023-34
The British journal of dermatology 2012 May;166(5):1023-34
Interleukin-8, a promising predictor for prognosis of pancreatic cancer.
Chen Y, Shi M, Yu GZ, Qin XR, Jin G, Chen P, Zhu MH
World journal of gastroenterology 2012 Mar 14;18(10):1123-9
World journal of gastroenterology 2012 Mar 14;18(10):1123-9
Stress effects on FosB- and interleukin-8 (IL8)-driven ovarian cancer growth and metastasis.
Shahzad MM, Arevalo JM, Armaiz-Pena GN, Lu C, Stone RL, Moreno-Smith M, Nishimura M, Lee JW, Jennings NB, Bottsford-Miller J, Vivas-Mejia P, Lutgendorf SK, Lopez-Berestein G, Bar-Eli M, Cole SW, Sood AK
The Journal of biological chemistry 2010 Nov 12;285(46):35462-70
The Journal of biological chemistry 2010 Nov 12;285(46):35462-70
Cytokines and growth factors expressed by human cutaneous melanoma.
Elias EG, Hasskamp JH, Sharma BK
Cancers 2010 May 7;2(2):794-808
Cancers 2010 May 7;2(2):794-808
Modification of the primary tumor microenvironment by transforming growth factor alpha-epidermal growth factor receptor signaling promotes metastasis in an orthotopic colon cancer model.
Sasaki T, Nakamura T, Rebhun RB, Cheng H, Hale KS, Tsan RZ, Fidler IJ, Langley RR
The American journal of pathology 2008 Jul;173(1):205-16
The American journal of pathology 2008 Jul;173(1):205-16
Clinical and biological significance of vascular endothelial growth factor in endometrial cancer.
Kamat AA, Merritt WM, Coffey D, Lin YG, Patel PR, Broaddus R, Nugent E, Han LY, Landen CN Jr, Spannuth WA, Lu C, Coleman RL, Gershenson DM, Sood AK
Clinical cancer research : an official journal of the American Association for Cancer Research 2007 Dec 15;13(24):7487-95
Clinical cancer research : an official journal of the American Association for Cancer Research 2007 Dec 15;13(24):7487-95
Induction of CXC chemokines in A549 airway epithelial cells by trypsin and staphylococcal proteases - a possible route for neutrophilic inflammation in chronic rhinosinusitis.
Sachse F, von Eiff C, Stoll W, Becker K, Rudack C
Clinical and experimental immunology 2006 Jun;144(3):534-42
Clinical and experimental immunology 2006 Jun;144(3):534-42
Monocyte/macrophage and T-cell infiltrates in peritoneum of patients with ovarian cancer or benign pelvic disease.
Wang X, Deavers M, Patenia R, Bassett RL Jr, Mueller P, Ma Q, Wang E, Freedman RS
Journal of translational medicine 2006 Jul 6;4:30
Journal of translational medicine 2006 Jul 6;4:30
Specific C-terminal cleavage and inactivation of interleukin-8 by invasive disease isolates of Streptococcus pyogenes.
Edwards RJ, Taylor GW, Ferguson M, Murray S, Rendell N, Wrigley A, Bai Z, Boyle J, Finney SJ, Jones A, Russell HH, Turner C, Cohen J, Faulkner L, Sriskandan S
The Journal of infectious diseases 2005 Sep 1;192(5):783-90
The Journal of infectious diseases 2005 Sep 1;192(5):783-90
Modulation of bone microenvironment with zoledronate enhances the therapeutic effects of STI571 and paclitaxel against experimental bone metastasis of human prostate cancer.
Kim SJ, Uehara H, Yazici S, He J, Langley RR, Mathew P, Fan D, Fidler IJ
Cancer research 2005 May 1;65(9):3707-15
Cancer research 2005 May 1;65(9):3707-15
Differential expression of interleukin-8 and its receptors in the neuroendocrine and non-neuroendocrine compartments of prostate cancer.
Huang J, Yao JL, Zhang L, Bourne PA, Quinn AM, di Sant'Agnese PA, Reeder JE
The American journal of pathology 2005 Jun;166(6):1807-15
The American journal of pathology 2005 Jun;166(6):1807-15
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-IL-8 (CXCL8) Polyclonal Antibody(Product # AHC0881) and a 11kDa band corresponding to IL-8 (CXCL8) was observed in Human PBMC and U-87 MG with Thapsigargin and PTI treatment. Whole cell extracts (30 µg lysate) of U-87 MG (Lane 1), U-87 MG treated with PTI (1X for 4hr) (Lane 2), U-87 MG treated with Thapsigargin (1uM for 24hr) followed by PTI (1X for 4hr) (Lane 3) and Human PBMC (Lane 4) were electrophoresed using Novex™ 16% Tricine Protein Gel (Product # EC6695BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:1000 dilution) and detected by chemiluminescence with Goat anti-Rabbit IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A27036, 1:4000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
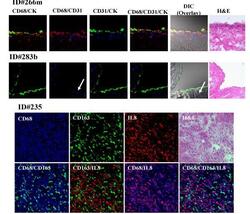
- Figure 3 Triple immunofluorescence costaining of frozen right peritoneal tissues were stained with CD68 (red), CD31 (blue), and keratin (CK) (green) antibodies (Rows 1 & 2) Row 1, peritoneal cells from a patient with EOC (ID 266 m) appear yellow from the colocalization of CD68 (red) and CK (green) on some surface mesothelial cells. CD31 staining (blue) indicates endothelial cells just under the mesothelium. Row 2, peritoneal cells from a patient with benign cystic teratoma of the ovary (ID 283b) show prominent staining for keratin in the single cell mesothelial layer but no staining for CD68 staining (red) and positive staining for endothelial cells (blue). Rows 3 and 4, peritoneal cells from patient ID#235 showed colocalization of CD68 (blue) and CD163 (green) appearing cyan color; CD68 (blue) and CXCL8 (red) costaining showed magenta effect and no color changed in CD163+ cells (green). Images were analyzed by confocal laser scanning microscopy (magnification 400x). H&E stained sections are shown for comparison.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Immunohistochemical expression of IL-8 (a), CXCR2 (b), IL-6 (c), tyrosine p-STAT-3 (d, e), SOCS-3 (f), and VEGF (g, h) in lymph nodes from CLL patients. (a) A CLL case with very few scattered IL-8 positive lymphoid cells. The inset shows an IL-8 positive cell in a higher magnification. Note the positive endothelial cells. (b) CXCR2 expression in a CLL lymph node. (c) IL-6 expression homogeneously distributed throughout the lymphoid tissue. (d) Tyrosine p-STAT-3 in a case showing a more pronounced immunoreactivity in the PCs. Note that endothelial cells are strongly positive. (e) Scattered tyrosine p-STAT-3 positive lymphoid cells along with positive endothelial cells in a CLL case. (f) Higher SOCS-3 expression in the PC in a CLL case. (g) (h) Pronounced VEGF immunoexpression in the PCs compared with the area outside the PCs. Higher magnification (h) showing the cytoplasmic immunoreactivity of VEGF. (PC: proliferation centers; E: endothelial cells).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Representative expression status for ANG, CA9, MMP9, MMP10, SERPINA1, APOE, SDC1, VEGFA, serpine1 and IL8 levels in tumor tissue. Insert Representative expression status for ANG, CA9, MMP9, MMP10, SERPINA1, APOE, SDC1, VEGFA, serpine1 and IL-8 levels in benign tissue. All images were captured at 400x magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 1 Representative expression status for ANG, CA9, MMP9, MMP10, A1AT, APOE, SDC1, VEGFA, PAI1 and IL8 in high-grade non-muscle invasive bladder tumor. Representative expression status for ANG, CA9, MMP9, MMP10, A1AT, APOE, SDC1, VEGFA, PAI1 and IL8 levels in benign tissue noted in square insert image. All images were captured at 400x magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 6 Figure GGA and rM180-treated hPDLCs promote angiogenesis in HUVECs. For the tube formation assay, HUVECs were cultured in matrigel-coated 96-well plates, and the supernatants from hPDLCs that were stimulated with or without GGA, rM180, or GGA + rM180 were added to the HUVECs (A) in the absence or (C) presence of neutralizing anti-Areg, anti-IL-8, anti-MCP-1, and anti-IL-6; (B) in the presence of the isotype control for 12 h at 37degC. (D) Tube formation was evaluated by counting the number of capillary tubes formed in at least 10 randomly selected fields per well. Scale bar = 200 mum. Data are represented as mean +- SD . n = 5. GGA, geranylgeranylacetone; hPDLC, human periodontal ligament cell; HUVEC, human umbilical vein endothelial cell. * p < .05; ** p < .01; *** p < .001
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
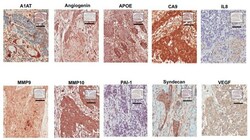
- Figure 1 Images of representative IHC staining for factors in primary melanoma (A) and metastatic melanoma (B).
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA