Antibody data
- Antibody Data
- Antigen structure
- References [8]
- Comments [0]
- Validations
- Flow cytometry [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- Q10480 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Anti-CD4 Monoclonal Antibody (S3.5), Qdot 605
- Antibody type
- Monoclonal
- Antigen
- Other
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- S3.5
- Vial size
- 25 µL
- Concentration
- Lot Dependent
- Storage
- 4° C, do not freeze
Submitted references Analysis of Intracellular Ca(2+) Mobilization in Human NK Cell Subsets by Flow Cytometry.
Qualification of a whole blood intracellular cytokine staining assay to measure mycobacteria-specific CD4 and CD8 T cell immunity by flow cytometry.
The novel tuberculosis vaccine, AERAS-402, is safe in healthy infants previously vaccinated with BCG, and induces dose-dependent CD4 and CD8T cell responses.
Distinct T-cell responses when BCG vaccination is delayed from birth to 6 weeks of age in Ugandan infants.
Heterologous vaccination against human tuberculosis modulates antigen-specific CD4+ T-cell function.
Differential regulatory T cell activity in HIV type 1-exposed seronegative individuals.
The Thai phase III trial (RV144) vaccine regimen induces T cell responses that preferentially target epitopes within the V2 region of HIV-1 envelope.
Functional capacity of Mycobacterium tuberculosis-specific T cell responses in humans is associated with mycobacterial load.
Theorell J, Bryceson YT
Methods in molecular biology (Clifton, N.J.) 2016;1441:117-30
Methods in molecular biology (Clifton, N.J.) 2016;1441:117-30
Qualification of a whole blood intracellular cytokine staining assay to measure mycobacteria-specific CD4 and CD8 T cell immunity by flow cytometry.
Kagina BM, Mansoor N, Kpamegan EP, Penn-Nicholson A, Nemes E, Smit E, Gelderbloem S, Soares AP, Abel B, Keyser A, Sidibana M, Hughes JE, Kaplan G, Hussey GD, Hanekom WA, Scriba TJ
Journal of immunological methods 2015 Feb;417:22-33
Journal of immunological methods 2015 Feb;417:22-33
The novel tuberculosis vaccine, AERAS-402, is safe in healthy infants previously vaccinated with BCG, and induces dose-dependent CD4 and CD8T cell responses.
Kagina BM, Tameris MD, Geldenhuys H, Hatherill M, Abel B, Hussey GD, Scriba TJ, Mahomed H, Sadoff JC, Hanekom WA, 018-402 Clinical Lab study team., Mansoor N, Hughes J, de Kock M, Whatney W, Africa H, Krohn C, Veldsman A, Kany AL, Douoguih M, Pau MG, Hendriks J, McClainc B, Benko J, Snowden MA, Hokey DA
Vaccine 2014 Oct 14;32(45):5908-17
Vaccine 2014 Oct 14;32(45):5908-17
Distinct T-cell responses when BCG vaccination is delayed from birth to 6 weeks of age in Ugandan infants.
Lutwama F, Kagina BM, Wajja A, Waiswa F, Mansoor N, Kirimunda S, Hughes EJ, Kiwanuka N, Joloba ML, Musoke P, Scriba TJ, Mayanja-Kizza H, Day CL, Hanekom WA
The Journal of infectious diseases 2014 Mar;209(6):887-97
The Journal of infectious diseases 2014 Mar;209(6):887-97
Heterologous vaccination against human tuberculosis modulates antigen-specific CD4+ T-cell function.
Dintwe OB, Day CL, Smit E, Nemes E, Gray C, Tameris M, McShane H, Mahomed H, Hanekom WA, Scriba TJ
European journal of immunology 2013 Sep;43(9):2409-20
European journal of immunology 2013 Sep;43(9):2409-20
Differential regulatory T cell activity in HIV type 1-exposed seronegative individuals.
Pattacini L, Murnane PM, Kahle EM, Bolton MJ, Delrow JJ, Lingappa JR, Katabira E, Donnell D, McElrath MJ, Baeten JM, Lund JM
AIDS research and human retroviruses 2013 Oct;29(10):1321-9
AIDS research and human retroviruses 2013 Oct;29(10):1321-9
The Thai phase III trial (RV144) vaccine regimen induces T cell responses that preferentially target epitopes within the V2 region of HIV-1 envelope.
de Souza MS, Ratto-Kim S, Chuenarom W, Schuetz A, Chantakulkij S, Nuntapinit B, Valencia-Micolta A, Thelian D, Nitayaphan S, Pitisuttithum P, Paris RM, Kaewkungwal J, Michael NL, Rerks-Ngarm S, Mathieson B, Marovich M, Currier JR, Kim JH, Ministry of Public Health–Thai AIDS Vaccine Evaluation Group Collaborators.
Journal of immunology (Baltimore, Md. : 1950) 2012 May 15;188(10):5166-76
Journal of immunology (Baltimore, Md. : 1950) 2012 May 15;188(10):5166-76
Functional capacity of Mycobacterium tuberculosis-specific T cell responses in humans is associated with mycobacterial load.
Day CL, Abrahams DA, Lerumo L, Janse van Rensburg E, Stone L, O'rie T, Pienaar B, de Kock M, Kaplan G, Mahomed H, Dheda K, Hanekom WA
Journal of immunology (Baltimore, Md. : 1950) 2011 Sep 1;187(5):2222-32
Journal of immunology (Baltimore, Md. : 1950) 2011 Sep 1;187(5):2222-32
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
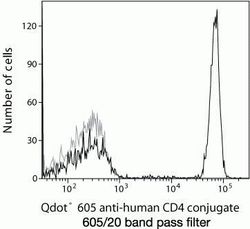
- Experimental details
- Qdot® 605 anti-human CD4 conjugate 605/20 band pass filter
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry