Antibody data
- Antibody Data
- Antigen structure
- References [48]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [24]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 17-0048-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD4 Monoclonal Antibody (OKT4 (OKT-4)), APC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The OKT4 monoclonal antibody reacts with human CD4, a 59 kDa cell surface glycoprotein expressed by the majority of thymocytes, a subpopulation of mature T cells (T-helper cells) and in low levels on monocytes. CD4 is a receptor for the human immunodeficiency virus (HIV). The OKT4 antibody recognizes a different epitope than the RPA-T4 monoclonal antibody, and these antibodies do not cross-block binding to each other's respective epitopes. Applications Reported: This OKT4 (OKT-4) antibody has been reported for use in flow cytometric analysis. Applications Tested: This OKT4 (OKT-4) antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.06 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 633-647 nm; Emission: 660 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- OKT4 (OKT-4)
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Plasma Extracellular Vesicle Subtypes May be Useful as Potential Biomarkers of Immune Activation in People With HIV.
In vitro and In vivo CD8(+) T Cell Suppression Assays.
Targeting Phosphatidylserine Enhances the Anti-tumor Response to Tumor-Directed Radiation Therapy in a Preclinical Model of Melanoma.
Diurnal Variation of Plasma Extracellular Vesicle Is Disrupted in People Living with HIV.
The TCR Repertoire Reconstitution in Multiple Sclerosis: Comparing One-Shot and Continuous Immunosuppressive Therapies.
Mesenchymal Stem and Stromal Cells Harness Macrophage-Derived Amphiregulin to Maintain Tissue Homeostasis.
A Cross-Reactive Small Protein Binding Domain Provides a Model to Study Off-Tumor CAR-T Cell Toxicity.
The Potential of Nanobody-Targeted Photodynamic Therapy to Trigger Immune Responses.
T-Lymphocyte Subset Distribution and Activity in Patients With Glaucoma.
Expression of CD20 after viral reactivation renders HIV-reservoir cells susceptible to Rituximab.
Fingolimod reduces circulating tight-junction protein levels and in vitro peripheral blood mononuclear cells migration in multiple sclerosis patients.
Complement receptor CD46 co-stimulates optimal human CD8(+) T cell effector function via fatty acid metabolism.
Clinically compliant spatial and temporal imaging of chimeric antigen receptor T-cells.
Accumulation of T-helper 22 cells, interleukin-22 and myeloid-derived suppressor cells promotes gastric cancer progression in elderly patients.
IL-6 receptor blockade corrects defects of XIAP-deficient regulatory T cells.
Targeted Disruption of TCF12 Reveals HEB as Essential in Human Mesodermal Specification and Hematopoiesis.
Ex vivo culture of human atherosclerotic plaques: A model to study immune cells in atherogenesis.
Hormonal vitamin D up-regulates tissue-specific PD-L1 and PD-L2 surface glycoprotein expression in humans but not mice.
Modulation of Endoplasmic Reticulum Stress Controls CD4(+) T-cell Activation and Antitumor Function.
Single-cell profiling reveals GPCR heterogeneity and functional patterning during neuroinflammation.
A new cell line for high throughput HIV-specific antibody-dependent cellular cytotoxicity (ADCC) and cell-to-cell virus transmission studies.
Tax and Semaphorin 4D Released from Lymphocytes Infected with Human Lymphotropic Virus Type 1 and Their Effect on Neurite Growth.
Function of Treg Cells Decreased in Patients With Systemic Lupus Erythematosus Due To the Effect of Prolactin.
PD-L1 expression and prognostic impact in glioblastoma.
Tumor-derived exosomes regulate expression of immune function-related genes in human T cell subsets.
Control of signaling-mediated clearance of apoptotic cells by the tumor suppressor p53.
Hyperreactive onchocerciasis is characterized by a combination of Th17-Th2 immune responses and reduced regulatory T cells.
The HIV-1 gp120 CD4-bound conformation is preferentially targeted by antibody-dependent cellular cytotoxicity-mediating antibodies in sera from HIV-1-infected individuals.
RNAi-Mediated CCR5 Knockdown Provides HIV-1 Resistance to Memory T Cells in Humanized BLT Mice.
Serum macrophage-derived chemokine/CCL22 levels are associated with glioma risk, CD4 T cell lymphopenia and survival time.
A comparison of DNA methylation specific droplet digital PCR (ddPCR) and real time qPCR with flow cytometry in characterizing human T cells in peripheral blood.
Aging and cytomegalovirus infection differentially and jointly affect distinct circulating T cell subsets in humans.
Immunologic human renal allograft injury associates with an altered IL-10/TNF-α expression ratio in regulatory B cells.
Reprogramming human endothelial cells to haematopoietic cells requires vascular induction.
Pathogenicity of pandemic H1N1 influenza A virus in immunocompromised cynomolgus macaques.
Experimental infection of macaques with a wild water bird-derived highly pathogenic avian influenza virus (H5N1).
Protection against H5N1 highly pathogenic avian and pandemic (H1N1) 2009 influenza virus infection in cynomolgus monkeys by an inactivated H5N1 whole particle vaccine.
Small molecule inhibitor of antigen binding and presentation by HLA-DR2b as a therapeutic strategy for the treatment of multiple sclerosis.
Association of a NOD2 gene polymorphism and T-helper 17 cells with presumed ocular toxoplasmosis.
A Francisella tularensis live vaccine strain that improves stimulation of antigen-presenting cells does not enhance vaccine efficacy.
Engineering HIV-1-resistant T-cells from short-hairpin RNA-expressing hematopoietic stem/progenitor cells in humanized BLT mice.
Prolactin down-regulates CD4+CD25hiCD127low/- regulatory T cell function in humans.
A mutation in the human Uncoordinated 119 gene impairs TCR signaling and is associated with CD4 lymphopenia.
Epigenetic biomarkers of T-cells in human glioma.
Epigenetic regulation of Foxp3 expression in regulatory T cells by DNA methylation.
Epigenetic regulation of Foxp3 expression in regulatory T cells by DNA methylation.
CD4-CCR5 interaction in intracellular compartments contributes to receptor expression at the cell surface.
Inhibition of gp160 and CD4 maturation in U937 cells after both defective and productive infections by human immunodeficiency virus type 1.
Bazié WW, Boucher J, Vitry J, Goyer B, Routy JP, Tremblay C, Trottier S, Jenabian MA, Provost P, Alary M, Gilbert C
Pathogens & immunity 2021;6(1):1-28
Pathogens & immunity 2021;6(1):1-28
In vitro and In vivo CD8(+) T Cell Suppression Assays.
Xie L, Liu G, Liu Y, Yu Y
Bio-protocol 2021 May 20;11(10):e4020
Bio-protocol 2021 May 20;11(10):e4020
Targeting Phosphatidylserine Enhances the Anti-tumor Response to Tumor-Directed Radiation Therapy in a Preclinical Model of Melanoma.
Budhu S, Giese R, Gupta A, Fitzgerald K, Zappasodi R, Schad S, Hirschhorn D, Campesato LF, De Henau O, Gigoux M, Liu C, Mazo G, Deng L, Barker CA, Wolchok JD, Merghoub T
Cell reports 2021 Jan 12;34(2):108620
Cell reports 2021 Jan 12;34(2):108620
Diurnal Variation of Plasma Extracellular Vesicle Is Disrupted in People Living with HIV.
Bazié WW, Goyer B, Boucher J, Zhang Y, Planas D, Chatterjee D, Routy JP, Alary M, Ancuta P, Gilbert C
Pathogens (Basel, Switzerland) 2021 Apr 24;10(5)
Pathogens (Basel, Switzerland) 2021 Apr 24;10(5)
The TCR Repertoire Reconstitution in Multiple Sclerosis: Comparing One-Shot and Continuous Immunosuppressive Therapies.
Amoriello R, Greiff V, Aldinucci A, Bonechi E, Carnasciali A, Peruzzi B, Repice AM, Mariottini A, Saccardi R, Mazzanti B, Massacesi L, Ballerini C
Frontiers in immunology 2020;11:559
Frontiers in immunology 2020;11:559
Mesenchymal Stem and Stromal Cells Harness Macrophage-Derived Amphiregulin to Maintain Tissue Homeostasis.
Ko JH, Kim HJ, Jeong HJ, Lee HJ, Oh JY
Cell reports 2020 Mar 17;30(11):3806-3820.e6
Cell reports 2020 Mar 17;30(11):3806-3820.e6
A Cross-Reactive Small Protein Binding Domain Provides a Model to Study Off-Tumor CAR-T Cell Toxicity.
Hammill JA, Kwiecien JM, Dvorkin-Gheva A, Lau VWC, Baker C, Wu Y, Bezverbnaya K, Aarts C, Heslen CW, Denisova GF, Derocher H, Milne K, Nelson BH, Bramson JL
Molecular therapy oncolytics 2020 Jun 26;17:278-292
Molecular therapy oncolytics 2020 Jun 26;17:278-292
The Potential of Nanobody-Targeted Photodynamic Therapy to Trigger Immune Responses.
Beltrán Hernández I, Angelier ML, Del Buono D'Ondes T, Di Maggio A, Yu Y, Oliveira S
Cancers 2020 Apr 15;12(4)
Cancers 2020 Apr 15;12(4)
T-Lymphocyte Subset Distribution and Activity in Patients With Glaucoma.
Yang X, Zeng Q, Göktas E, Gopal K, Al-Aswad L, Blumberg DM, Cioffi GA, Liebmann JM, Tezel G
Investigative ophthalmology & visual science 2019 Mar 1;60(4):877-888
Investigative ophthalmology & visual science 2019 Mar 1;60(4):877-888
Expression of CD20 after viral reactivation renders HIV-reservoir cells susceptible to Rituximab.
Serra-Peinado C, Grau-Expósito J, Luque-Ballesteros L, Astorga-Gamaza A, Navarro J, Gallego-Rodriguez J, Martin M, Curran A, Burgos J, Ribera E, Raventós B, Willekens R, Torrella A, Planas B, Badía R, Garcia F, Castellví J, Genescà M, Falcó V, Buzon MJ
Nature communications 2019 Aug 16;10(1):3705
Nature communications 2019 Aug 16;10(1):3705
Fingolimod reduces circulating tight-junction protein levels and in vitro peripheral blood mononuclear cells migration in multiple sclerosis patients.
Annunziata P, Cioni C, Masi G, Tassi M, Marotta G, Severi S
Scientific reports 2018 Oct 18;8(1):15371
Scientific reports 2018 Oct 18;8(1):15371
Complement receptor CD46 co-stimulates optimal human CD8(+) T cell effector function via fatty acid metabolism.
Arbore G, West EE, Rahman J, Le Friec G, Niyonzima N, Pirooznia M, Tunc I, Pavlidis P, Powell N, Li Y, Liu P, Servais A, Couzi L, Fremeaux-Bacchi V, Placais L, Ferraro A, Walsh PR, Kavanagh D, Afzali B, Lavender P, Lachmann HJ, Kemper C
Nature communications 2018 Oct 10;9(1):4186
Nature communications 2018 Oct 10;9(1):4186
Clinically compliant spatial and temporal imaging of chimeric antigen receptor T-cells.
Emami-Shahri N, Foster J, Kashani R, Gazinska P, Cook C, Sosabowski J, Maher J, Papa S
Nature communications 2018 Mar 14;9(1):1081
Nature communications 2018 Mar 14;9(1):1081
Accumulation of T-helper 22 cells, interleukin-22 and myeloid-derived suppressor cells promotes gastric cancer progression in elderly patients.
Chen X, Wang Y, Wang J, Wen J, Jia X, Wang X, Zhang H
Oncology letters 2018 Jul;16(1):253-261
Oncology letters 2018 Jul;16(1):253-261
IL-6 receptor blockade corrects defects of XIAP-deficient regulatory T cells.
Hsieh WC, Hsu TS, Chang YJ, Lai MZ
Nature communications 2018 Jan 31;9(1):463
Nature communications 2018 Jan 31;9(1):463
Targeted Disruption of TCF12 Reveals HEB as Essential in Human Mesodermal Specification and Hematopoiesis.
Li Y, Brauer PM, Singh J, Xhiku S, Yoganathan K, Zúñiga-Pflücker JC, Anderson MK
Stem cell reports 2017 Sep 12;9(3):779-795
Stem cell reports 2017 Sep 12;9(3):779-795
Ex vivo culture of human atherosclerotic plaques: A model to study immune cells in atherogenesis.
Lebedeva A, Vorobyeva D, Vagida M, Ivanova O, Felker E, Fitzgerald W, Danilova N, Gontarenko V, Shpektor A, Vasilieva E, Margolis L
Atherosclerosis 2017 Dec;267:90-98
Atherosclerosis 2017 Dec;267:90-98
Hormonal vitamin D up-regulates tissue-specific PD-L1 and PD-L2 surface glycoprotein expression in humans but not mice.
Dimitrov V, Bouttier M, Boukhaled G, Salehi-Tabar R, Avramescu RG, Memari B, Hasaj B, Lukacs GL, Krawczyk CM, White JH
The Journal of biological chemistry 2017 Dec 15;292(50):20657-20668
The Journal of biological chemistry 2017 Dec 15;292(50):20657-20668
Modulation of Endoplasmic Reticulum Stress Controls CD4(+) T-cell Activation and Antitumor Function.
Thaxton JE, Wallace C, Riesenberg B, Zhang Y, Paulos CM, Beeson CC, Liu B, Li Z
Cancer immunology research 2017 Aug;5(8):666-675
Cancer immunology research 2017 Aug;5(8):666-675
Single-cell profiling reveals GPCR heterogeneity and functional patterning during neuroinflammation.
Tischner D, Grimm M, Kaur H, Staudenraus D, Carvalho J, Looso M, Günther S, Wanke F, Moos S, Siller N, Breuer J, Schwab N, Zipp F, Waisman A, Kurschus FC, Offermanns S, Wettschureck N
JCI insight 2017 Aug 3;2(15)
JCI insight 2017 Aug 3;2(15)
A new cell line for high throughput HIV-specific antibody-dependent cellular cytotoxicity (ADCC) and cell-to-cell virus transmission studies.
Orlandi C, Flinko R, Lewis GK
Journal of immunological methods 2016 Jun;433:51-8
Journal of immunological methods 2016 Jun;433:51-8
Tax and Semaphorin 4D Released from Lymphocytes Infected with Human Lymphotropic Virus Type 1 and Their Effect on Neurite Growth.
Quintremil S, Alberti C, Rivera M, Medina F, Puente J, Cartier L, Ramírez E, Tanaka Y, Valenzuela MA
AIDS research and human retroviruses 2016 Jan;32(1):68-79
AIDS research and human retroviruses 2016 Jan;32(1):68-79
Function of Treg Cells Decreased in Patients With Systemic Lupus Erythematosus Due To the Effect of Prolactin.
Legorreta-Haquet MV, Chávez-Rueda K, Chávez-Sánchez L, Cervera-Castillo H, Zenteno-Galindo E, Barile-Fabris L, Burgos-Vargas R, Álvarez-Hernández E, Blanco-Favela F
Medicine 2016 Feb;95(5):e2384
Medicine 2016 Feb;95(5):e2384
PD-L1 expression and prognostic impact in glioblastoma.
Nduom EK, Wei J, Yaghi NK, Huang N, Kong LY, Gabrusiewicz K, Ling X, Zhou S, Ivan C, Chen JQ, Burks JK, Fuller GN, Calin GA, Conrad CA, Creasy C, Ritthipichai K, Radvanyi L, Heimberger AB
Neuro-oncology 2016 Feb;18(2):195-205
Neuro-oncology 2016 Feb;18(2):195-205
Tumor-derived exosomes regulate expression of immune function-related genes in human T cell subsets.
Muller L, Mitsuhashi M, Simms P, Gooding WE, Whiteside TL
Scientific reports 2016 Feb 4;6:20254
Scientific reports 2016 Feb 4;6:20254
Control of signaling-mediated clearance of apoptotic cells by the tumor suppressor p53.
Yoon KW, Byun S, Kwon E, Hwang SY, Chu K, Hiraki M, Jo SH, Weins A, Hakroush S, Cebulla A, Sykes DB, Greka A, Mundel P, Fisher DE, Mandinova A, Lee SW
Science (New York, N.Y.) 2015 Jul 31;349(6247):1261669
Science (New York, N.Y.) 2015 Jul 31;349(6247):1261669
Hyperreactive onchocerciasis is characterized by a combination of Th17-Th2 immune responses and reduced regulatory T cells.
Katawa G, Layland LE, Debrah AY, von Horn C, Batsa L, Kwarteng A, Arriens S, W Taylor D, Specht S, Hoerauf A, Adjobimey T
PLoS neglected tropical diseases 2015 Jan;9(1):e3414
PLoS neglected tropical diseases 2015 Jan;9(1):e3414
The HIV-1 gp120 CD4-bound conformation is preferentially targeted by antibody-dependent cellular cytotoxicity-mediating antibodies in sera from HIV-1-infected individuals.
Veillette M, Coutu M, Richard J, Batraville LA, Dagher O, Bernard N, Tremblay C, Kaufmann DE, Roger M, Finzi A
Journal of virology 2015 Jan;89(1):545-51
Journal of virology 2015 Jan;89(1):545-51
RNAi-Mediated CCR5 Knockdown Provides HIV-1 Resistance to Memory T Cells in Humanized BLT Mice.
Shimizu S, Ringpis GE, Marsden MD, Cortado RV, Wilhalme HM, Elashoff D, Zack JA, Chen IS, An DS
Molecular therapy. Nucleic acids 2015 Feb 17;4(2):e227
Molecular therapy. Nucleic acids 2015 Feb 17;4(2):e227
Serum macrophage-derived chemokine/CCL22 levels are associated with glioma risk, CD4 T cell lymphopenia and survival time.
Zhou M, Bracci PM, McCoy LS, Hsuang G, Wiemels JL, Rice T, Zheng S, Kelsey KT, Wrensch MR, Wiencke JK
International journal of cancer 2015 Aug 15;137(4):826-36
International journal of cancer 2015 Aug 15;137(4):826-36
A comparison of DNA methylation specific droplet digital PCR (ddPCR) and real time qPCR with flow cytometry in characterizing human T cells in peripheral blood.
Wiencke JK, Bracci PM, Hsuang G, Zheng S, Hansen H, Wrensch MR, Rice T, Eliot M, Kelsey KT
Epigenetics 2014 Oct;9(10):1360-5
Epigenetics 2014 Oct;9(10):1360-5
Aging and cytomegalovirus infection differentially and jointly affect distinct circulating T cell subsets in humans.
Wertheimer AM, Bennett MS, Park B, Uhrlaub JL, Martinez C, Pulko V, Currier NL, Nikolich-Žugich D, Kaye J, Nikolich-Žugich J
Journal of immunology (Baltimore, Md. : 1950) 2014 Mar 1;192(5):2143-55
Journal of immunology (Baltimore, Md. : 1950) 2014 Mar 1;192(5):2143-55
Immunologic human renal allograft injury associates with an altered IL-10/TNF-α expression ratio in regulatory B cells.
Cherukuri A, Rothstein DM, Clark B, Carter CR, Davison A, Hernandez-Fuentes M, Hewitt E, Salama AD, Baker RJ
Journal of the American Society of Nephrology : JASN 2014 Jul;25(7):1575-85
Journal of the American Society of Nephrology : JASN 2014 Jul;25(7):1575-85
Reprogramming human endothelial cells to haematopoietic cells requires vascular induction.
Sandler VM, Lis R, Liu Y, Kedem A, James D, Elemento O, Butler JM, Scandura JM, Rafii S
Nature 2014 Jul 17;511(7509):312-8
Nature 2014 Jul 17;511(7509):312-8
Pathogenicity of pandemic H1N1 influenza A virus in immunocompromised cynomolgus macaques.
Pham VL, Nakayama M, Itoh Y, Ishigaki H, Kitano M, Arikata M, Ishida H, Kitagawa N, Shichinohe S, Okamatsu M, Sakoda Y, Tsuchiya H, Nakamura S, Kida H, Ogasawara K
PloS one 2013;8(9):e75910
PloS one 2013;8(9):e75910
Experimental infection of macaques with a wild water bird-derived highly pathogenic avian influenza virus (H5N1).
Fujiyuki T, Yoneda M, Yasui F, Kuraishi T, Hattori S, Kwon HJ, Munekata K, Kiso Y, Kida H, Kohara M, Kai C
PloS one 2013;8(12):e83551
PloS one 2013;8(12):e83551
Protection against H5N1 highly pathogenic avian and pandemic (H1N1) 2009 influenza virus infection in cynomolgus monkeys by an inactivated H5N1 whole particle vaccine.
Nakayama M, Shichinohe S, Itoh Y, Ishigaki H, Kitano M, Arikata M, Pham VL, Ishida H, Kitagawa N, Okamatsu M, Sakoda Y, Ichikawa T, Tsuchiya H, Nakamura S, Le QM, Ito M, Kawaoka Y, Kida H, Ogasawara K
PloS one 2013;8(12):e82740
PloS one 2013;8(12):e82740
Small molecule inhibitor of antigen binding and presentation by HLA-DR2b as a therapeutic strategy for the treatment of multiple sclerosis.
Ji N, Somanaboeina A, Dixit A, Kawamura K, Hayward NJ, Self C, Olson GL, Forsthuber TG
Journal of immunology (Baltimore, Md. : 1950) 2013 Nov 15;191(10):5074-84
Journal of immunology (Baltimore, Md. : 1950) 2013 Nov 15;191(10):5074-84
Association of a NOD2 gene polymorphism and T-helper 17 cells with presumed ocular toxoplasmosis.
Dutra MS, Béla SR, Peixoto-Rangel AL, Fakiola M, Cruz AG, Gazzinelli A, Quites HF, Bahia-Oliveira LM, Peixe RG, Campos WR, Higino-Rocha AC, Miller NE, Blackwell JM, Antonelli LR, Gazzinelli RT
The Journal of infectious diseases 2013 Jan 1;207(1):152-63
The Journal of infectious diseases 2013 Jan 1;207(1):152-63
A Francisella tularensis live vaccine strain that improves stimulation of antigen-presenting cells does not enhance vaccine efficacy.
Schmitt DM, O'Dee DM, Horzempa J, Carlson PE Jr, Russo BC, Bales JM, Brown MJ, Nau GJ
PloS one 2012;7(2):e31172
PloS one 2012;7(2):e31172
Engineering HIV-1-resistant T-cells from short-hairpin RNA-expressing hematopoietic stem/progenitor cells in humanized BLT mice.
Ringpis GE, Shimizu S, Arokium H, Camba-Colón J, Carroll MV, Cortado R, Xie Y, Kim PY, Sahakyan A, Lowe EL, Narukawa M, Kandarian FN, Burke BP, Symonds GP, An DS, Chen IS, Kamata M
PloS one 2012;7(12):e53492
PloS one 2012;7(12):e53492
Prolactin down-regulates CD4+CD25hiCD127low/- regulatory T cell function in humans.
Legorreta-Haquet MV, Chávez-Rueda K, Montoya-Díaz E, Arriaga-Pizano L, Silva-García R, Chávez-Sánchez L, Moreno-Lafont M, Zenteno-Galindo E, Blanco-Favela F
Journal of molecular endocrinology 2012 Feb;48(1):77-85
Journal of molecular endocrinology 2012 Feb;48(1):77-85
A mutation in the human Uncoordinated 119 gene impairs TCR signaling and is associated with CD4 lymphopenia.
Gorska MM, Alam R
Blood 2012 Feb 9;119(6):1399-406
Blood 2012 Feb 9;119(6):1399-406
Epigenetic biomarkers of T-cells in human glioma.
Wiencke JK, Accomando WP, Zheng S, Patoka J, Dou X, Phillips JJ, Hsuang G, Christensen BC, Houseman EA, Koestler DC, Bracci P, Wiemels JL, Wrensch M, Nelson HH, Kelsey KT
Epigenetics 2012 Dec 1;7(12):1391-402
Epigenetics 2012 Dec 1;7(12):1391-402
Epigenetic regulation of Foxp3 expression in regulatory T cells by DNA methylation.
Lal G, Zhang N, van der Touw W, Ding Y, Ju W, Bottinger EP, Reid SP, Levy DE, Bromberg JS
Journal of immunology (Baltimore, Md. : 1950) 2009 Jan 1;182(1):259-73
Journal of immunology (Baltimore, Md. : 1950) 2009 Jan 1;182(1):259-73
Epigenetic regulation of Foxp3 expression in regulatory T cells by DNA methylation.
Lal G, Zhang N, van der Touw W, Ding Y, Ju W, Bottinger EP, Reid SP, Levy DE, Bromberg JS
Journal of immunology (Baltimore, Md. : 1950) 2009 Jan 1;182(1):259-73
Journal of immunology (Baltimore, Md. : 1950) 2009 Jan 1;182(1):259-73
CD4-CCR5 interaction in intracellular compartments contributes to receptor expression at the cell surface.
Achour L, Scott MG, Shirvani H, Thuret A, Bismuth G, Labbé-Jullié C, Marullo S
Blood 2009 Feb 26;113(9):1938-47
Blood 2009 Feb 26;113(9):1938-47
Inhibition of gp160 and CD4 maturation in U937 cells after both defective and productive infections by human immunodeficiency virus type 1.
Bour S, Boulerice F, Wainberg MA
Journal of virology 1991 Dec;65(12):6387-96
Journal of virology 1991 Dec;65(12):6387-96
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
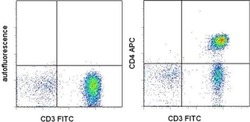
- Experimental details
- Staining of normal human peripheral blood cells with Anti-Human CD3 FITC (Product # 11-0037-42) and staining buffer (autofluorescence) (left) or Anti-Human CD4 APC (right). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 Effects of TEX on protein expression and functions of T cells. In ( a ) down-regulation of CD69 protein expression on the surface of responder CD4 + Tconv after co-incubation with TEX. Activated CD4 + Tconv were co-incubated with TEX (10 ug protein) produced by the PCI-13 cells or with PBS for 12 h. The CD69 expression levels on CD4 + Tconv were then determined by flow cytometry (MFI) and were converted into MESF units based on calibration curves established with fluorescent calibration beads. The bar graphs show data (mean values +- SD) from 3 independent experiments performed with CD4 + Tconv obtained from different normal donors. The asterisks indicate p values at p < 0.0005. In ( b ) changes in expression levels of CD39 protein on the surface of resting CD4 + CD39 + Treg co-incubated with TEX produced by the PCI-13 cell line or DEX. The exosomes were used at the concentration of 10 ng protein/ assay. Exogenous ATP was added as described in Methods. Flow cytometry ( right ) shows up-regulation of MFI for CD39 in a representative experiment, and the bar graph summarizes results of three experiments performed with Treg obtained from different donors. In ( c ), Production levels of 5' AMP, adenosine and inosine by resting CD4 + CD39 + Treg co-incubated with TEX produced by the PCI-13 cell line. The data are from one of two experiments performed in the presence of exogenous ATP. The analyte levels were measured by mass spectrometry as described in Methods.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
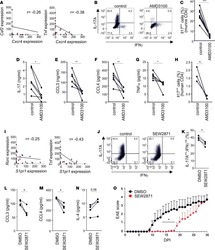
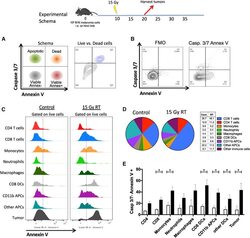
- Figure 1. PS Expression on Immune Subsets in the Tumor Increases after RT C57BL/6 mice were injected with 100,000 B16F10 melanoma cells intradermally on the hindlimb. 10 days later, tumor-bearing hindlimbs received 15 Gy RT. 10 days thereafter, tumors were excised and analyzed by flow cytometry. (A) Schema and representative plots showing caspase-3/7 activity and PS expression on live (blue) versus dead (gray) tumor cells determined by a viability dye. (B) Representative gating strategy used to measure PS expression using viable CD8+ T cells from the tumor as an example. Gating was based on caspase-3/7 and annexin V fluorescence minus one (FMO). (C) Relative expression of PS on viable immune cell subsets in the tumors of mice with and without tumor-directed RT. (D) Frequencies of immune cells as a percentage of live CD45+ cells in the tumor with and without tumor-directed RT. The tables next to the figure legend lists the percentages shown in the pie charts. (E) Quantification of annexin V+ immune and tumor cells +-SEM (3-5 mice per group) from control tumors (white bars) and RT tumors (black bars). *p < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
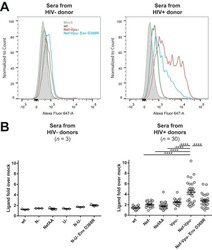
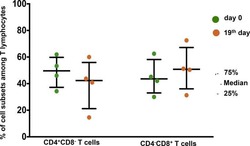
- Figure 2. EV quantification and cell origin in association with HIV. Purified plasma EVs fromuninfected (Control, n = 8) and HIV+ patients (n = 17) were stained with the lipophilic fluorescent tracer dye DiD to count total vesicles ( Figure 2A and B ), then labeled with antibodies directed against receptors CD45 ( Figure 2C and D ), CD4 ( Figure 2E and F ), and CD8 ( Figure 2 G and H ) to evaluate cell origin by cytofluorometry. Asterisks denote significant difference (** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
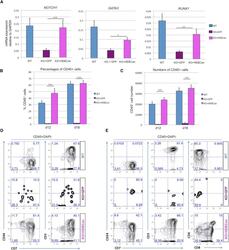
- Figure 7 HEBCan Rescues Hematopoiesis and T Cell Development in HEB -/- hESCs in OP9-DL4 Co-cultures (A) qRT-PCR analysis for the expression of hematopoietic genes in CD34 + cells sorted from WT, KO + GFP, and KO + HEBCan day-8 (d8) EBs. mRNA levels are shown relative to GAPDH. (B and C) Percentages (B) and numbers (C) of CD45 + cells in d12 and d18 OP9-DL4 co-cultures. (D and E) Flow-cytometric analysis of T cell development from WT, KO + GFP, and KO + HEBCan d8 EB-derived CD34 + cells at d12 (D) and d18 (E) of OP9-DL4 co-culture. Cells are gated on the CD45 + DAPI - population. Error bars represent mean +- SD (n = 3 independent experiments). * p < 0.05, ** p < 0.01, *** p < 0.005 by Student's t test. Plots in (B), (D), and (E) are representative of three independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
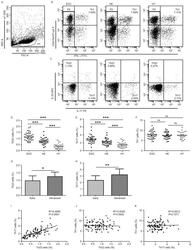
- Figure 1. Flow cytometric analysis was used to determine the distribution of Th22, Th17 and Th1 cells in EGC, HE and HY ( Fig. 1 ). Flow cytometric analysis of Th22, Th17 and Th1 cells in peripheral whole blood from EGC (n=39), HE (n=32) and HY (n=31). (A) Lymphocytes were gated in P1 using flow cytometry. CD4 + IFN-gamma - lymphocytes were gated in P2 using flow cytometry, and representative results of flow cytometric analyses for (B) Th1 (CD4 + IFN-gamma + ), (C) Th22 (CD4 + IFN-gamma - IL-17 - IL-22 + ) and Th17 (CD4 + IFN-gamma - IL-17 + IL-22 - ) cells in the three groups of subjects are presented. The number of cells stained in EGC, HE and HY in P2 were 2,654, 4,696 and 5,185, respectively. The proportion of (D) Th22, (E) Th17 and (F) Th1 cells in the three groups of subjects. The proportion of (G) Th22 and (H) Th17 cells in peripheral whole blood derived from patients with early (n=13) or advanced (n=26) gastric cancer. The association between the proportion of (I) Th22 and Th17 cells, (J) Th22 and Th1 cells, and (K) Th17 and Th1 cells, in peripheral whole blood of all subjects. *P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
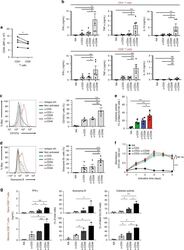
- Fig. 1 CD46 co-stimulation provides superior support for CTL activity. a CD46 expression on the surface of resting human CD4 + and CD8 + T cells assessed by FACS analysis ( n = 4, gating strategy in Supplementary Fig. 7a ). b Comparison of IFN-gamma, TNF-alpha and IL-10 secretion by CD3 + CD46-activated T cells. Purified CD4 + and CD8 + T cells from healthy donors were left non-activated (NA) or stimulated with immobilized antibodies to CD3, CD3 + CD28 or CD3 + CD46 and cytokines measured 60 h post activation ( n = 5). c, d Degranulation (CD107a staining, ( c )) and granzyme B expression ( d ) by CD8 + T cells upon CD46 co-stimulation. CD8 + T cells were stimulated as in ( a ) and CD107a and granzyme B expression assessed with left panels showing representative cytometry images and right panels corresponding quantifications ( n = 3, gating strategy in Supplementary Fig. 7b ). e Killing activity of CD46-activated CD8 + T cells. T cells were stimulated as depicted for 24 h and cytotoxic activity of differently activated CD8 + T cells towards DU145 target cells assessed 24 h post co-culture of T cells and DU145 cells ( n = 4, gating strategy in Supplementary Fig. 7c ). f Effect of CD46 co-stimulation on CD8 + T-cell proliferation. Purified T cells were activated as indicated for 5 days and cell proliferation measured each day ( n = 4) (black circles, non-activated cells; green, blue, and red circles, CD3, CD3 + CD28 or CD3 + CD46-activated cells, res
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
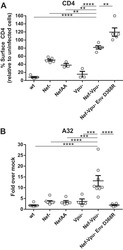
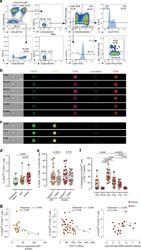
- Fig. 1 CD20 dim CD4 + T cells involve memory phenotypes with more activation than CD20 - cells. PBMC from uninfected donors, ART-suppressed patients and viremic patients were stained with CD20, activation markers (HLA-DR, CD69, CD25), and T-cell differentiation markers (CCR7, CD45RO, CD27, CD95). a Gating strategy used to identify CD20 dim CD4 + T cells. Previous sequential gates are represented in Supplementary Fig. 9 . Viable CD4 + T cells (identified by CD3 and CD4 expression) were first selected for the analysis of CD20 expression, defined by its Fluorescence Minus One (FMO) control. T-cell memory subsets were selected as follows: CD4 + T NA (CCR7 + , CD45RO - , CD27 + , CD95 - ), CD4 + T SCM (CCR7 + , CD45RO - , CD27 + , CD95 + ); CD4 + T CM (CCR7 + , CD45RO + ), CD4 + T TM (CCR7 - , CD45RO + , CD27 + ); CD4 + T EM (CCR7 - , CD45RO + , CD27 - ), CD4 + T TD (CCR7 - , CD45RO - ). b , c Representative bright-field and pseudo-color fluorescence images of CD20 dim CD4 + T cells ( b ) and B cells ( c ) from two ART-suppressed patients (#9 and #22) using the Amnis imaging flow cytometer technology. Scale bar 10 um. d Percentage of CD20 dim expression within CD4 + T cells in uninfected controls and the two patient cohorts. Mann-Whitney comparison was used to compare n = 6 uninfected controls, n = 21 ART-suppressed patients, n = 20 viremic patients. e Expression of the activation markers CD25, CD69 or HLA-DR in CD20 dim and CD20 - CD4 + T cells in different cohorts of HIV + patie
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
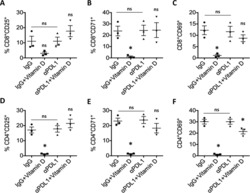
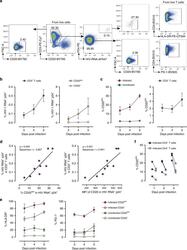
- Fig. 5 Ex vivo infection upregulates CD20 expression. Unstimulated PBMCs from three uninfected donors were infected with HIV strain NL4.3. Infection was monitored by simultaneous staining of HIV RNA using the RNA FISH-flow assay and the viral protein p24 at days 3, 4, and 5. a Gating strategy used to monitor HIV infection and expression of CD20 dim . Previous sequential gates are represented in Supplementary Fig. 9 . b Percentage of productively HIV-infected cells in CD3 + T cells (left panel) and in CD20 dim or CD20 - CD3 + T cells (right panel). c Expression of CD20 dim in infected and uninfected CD3 + T cells (left panel) and in the total CD3 + T cell population (right panel). d Correlation between the proportion of infected cells within the CD3 + T cell population and CD20 expression (left panel) and the mean fluorescence intensity (MFI) of CD20 (right panel). e Percentage of HLA-DR and PD-1 in infected and uninfected cells expressing or not expressing CD20 dim . f Proportion of CD20 dim in infected cells expressing the CD4 cell receptor versus infected cells with marked downregulation of the CD4 receptor. In all panels, the mean and SEM value of three independent experiments is represented. In panel ( d ), Spearman's nonparametric correlation coefficients and associated p values are shown. Data underlying this Figure are provided as Source Data file
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
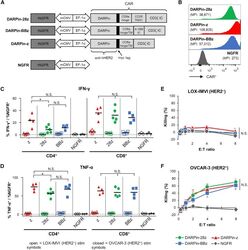
- Figure 1 Anti-HER2 DARPin-Targeted CAR-T Cells Demonstrated Similarl Efficacy In Vitro (A) Schematics of the dual-promoter lentiviral (LV) gene cassettes used to generate anti-HER2 DARPin-targeted first- or second-generation CAR-T cells (structural details are as indicated; TM, transmembrane; IC, intracellular) or CAR-negative control NGFR-T cells. In all cases, truncated NGFR (tNGFR) is included as a transduction marker. (B) Expression of CARs on the surface of engineered (NGFR + ) T cells as determined by flow cytometry (upstream gating strategy: lymphocytes - singlets - NGFR + ). Mean fluorescence intensity (MFI) for CAR expression is indicated in brackets. Representative results have been replicated in 2-4 additional independent experiments. (C and D) Production of IFN-gamma (C) and TNF-alpha (D) upon CAR-T cell stimulation with HER2 + (OVCAR-3; closed symbols) or HER2 - (LOX-IMVI; open symbols) human tumor cell lines was measured by intracellular cytokine staining (ICS) and subsequent flow cytometry (upstream gating strategy: lymphocytes - singlets - CD4 + or CD8 + T cells). Percent cytokine production was normalized for transduction (transduction ranges observed: DARPin-28z, 39%-60%; DARPin-BBz, 33%-52%; DARPin-z, 25%-63%; NGFR, 63%-86%). Each point indicates data from a single independent experiment (n = 3-5 per LV construct); black lines indicate mean values. (E and F) Cytotoxicity across various effector:target (E:T) ratios with LOX-IMVI (E) or OVCAR-3 (F) tumor cell
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
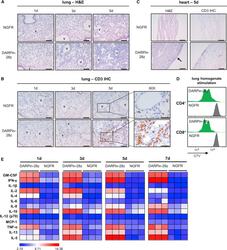
- Figure 3 DAPRin-28z-T Cells Activated in the Lungs and Heart, Resulting in a Systemic Cytokine Storm OVCAR-3 tumor-bearing NRG mice were treated with 6 x 10 6 effective DARPin-28z-T cells or a matched number of NGFR-T cells. (A-C) Mice were sacrificed at 1, 3, or 5 days post-ACT1 for total body perfusion, fixation, necropsy, and histological analysis. (A) Hematoxylin and eosin (H&E) staining of the lungs at 20x magnification (scale bars, 100 mum). V, vasculature. (B) Immunohistochemistry (IHC) for human CD3 in the lungs at 20x magnification (scale bars, 100 mum) or 60x magnification (zoom-in; scale bars, 50 mum). (C) H&E or CD3 IHC staining of the heart at 20x magnification (scale bars, 100 mum); arrow indicates aberrant region of inflammation along the right heart wall. Representative images from n = 2-3 mice are shown. Findings have been recapitulated in 1-2 additional independent experiments. (D) DARPin-28z- or NGFR-T cells were co-cultured with tumor-free NRG mouse lung homogenates ex vivo . T cell proliferation was measured by flow cytometry using CellTrace Violet (CTV) dye. Data are representative of two independent experiments. (E) Mice were bled at 1, 3, 5, or 7 days post-ACT1 for multiplex analysis of human serum cytokine content; a globally normalized heatmap of log2-transformed human cytokine fluorescence readings is shown. Each square represents data from one mouse. Colorimetric scale bar indicates minimum, average, and maximum values on map. Absolute values are d
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
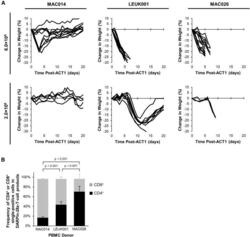
- Figure 4 Differential In Vivo Toxicity of DARPin-28z-T Cells Manufactured from Unique PBMC Donors Correlated with the Frequency of CD4+ T Cells in the Adoptive Transfer Product (A) OVCAR-3 tumor-bearing NRG mice were treated with 6.0 x 10 6 or 1.7-2.0 x 10 6 DARPin-28z-T cells produced from MAC026, LEUK001, or MAC014 PBMCs. Mice were monitored over time for changes in weight. Data were pooled from n = x independent experiments. For 6.0 x 10 6 cells, MAC014, 2; LEUK001, 3; and MAC026, 4. For 2.0 x 10 6 cells, MAC014, 1; LEUK001, 2; and MAC026, 1. Each line indicates data from one animal; curves end, indicating when mice succumbed to toxicity. (B) Composition of CD4 + or CD8 + cells in DARPin-28z-T cell products (days 13-14 post-activation) manufactured using starting PBMCs from donors as indicated and determined using flow cytometry (upstream gating strategy: lymphocytes - singlets - NGFR + ). Error bars represent SD. Data from n = x independent experiments; MAC014, 5 (2 unique PBMC preparations); LEUK001, 6 (1 PBMC preparation); and MAC026, 12 (5 unique PBMC preparations).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Enhanced proliferation and IFNgamma release of CD4+ T cells induced by moDCs stimulated with supernatant of NB-PDT treated tumor cells. A431 cells were treated with NB-PDT, the supernatant was collected 24 h later and incubated with immature moDCs for another 24 h. moDCs were then co- incubated with allogeneic CFSE-labeled CD4+ T cells in a 1:10 ratio. After 6 days, CD4+ T cell proliferation was measured with flow cytometry and IFNgamma release was assessed by ELISA. ( a ), Percentage of CD4+ T cells with weak CFSE signal, thus proliferating cells (n = 4). ( b ), Quantification of released IFNgamma by CD4+ T cells (n = 3). Each combination of allogeneic donors is represented by a different symbol and color. ctr, unstimulated DCs; LPS, LPS-stimulated DCs; NT, untreated tumor cells; LD50, mild cytotoxic NB-PDT; LD100, highly cytotoxic NB-PDT; Light, only light control; NB-PS, only NB-PS conjugate control. Significance is displayed as * p
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry