Antibody data
- Antibody Data
- Antigen structure
- References [44]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [13]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 45-0048-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD4 Monoclonal Antibody (OKT4 (OKT-4)), PerCP-Cyanine5.5, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The OKT4 monoclonal antibody reacts with human CD4, a 59 kDa cell surface glycoprotein expressed by the majority of thymocytes, a subpopulation of mature T cells (T-helper cells) and in low levels on monocytes. CD4 is a receptor for the human immunodeficiency virus (HIV). The OKT4 antibody recognizes a different epitope than the RPA-T4 monoclonal antibody, and these antibodies do not cross-block binding to each other's respective epitopes. Applications Reported: This OKT4 (OKT-4) antibody has been reported for use in flow cytometric analysis. Applications Tested: This OKT4 (OKT-4) antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.25 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488 nm; Emission: 695 nm; Laser: Blue Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- OKT4 (OKT-4)
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Gallic acid induces T-helper-1-like T(reg) cells and strengthens immune checkpoint blockade efficacy.
Elevations of monocyte and neutrophils, and higher levels of granulocyte colony-stimulating factor in peripheral blood in lung cancer patients.
Repositioning liothyronine for cancer immunotherapy by blocking the interaction of immune checkpoint TIGIT/PVR.
Boosting with AIDSVAX B/E Enhances Env Constant Region 1 and 2 Antibody-Dependent Cellular Cytotoxicity Breadth and Potency.
Human Fc Receptor-like 3 Inhibits Regulatory T Cell Function and Binds Secretory IgA.
The Potential of Nanobody-Targeted Photodynamic Therapy to Trigger Immune Responses.
Efficient gene transfer into T lymphocytes by fiber-modified human adenovirus 5.
Schistosoma mansoni rSm29 Antigen Induces a Regulatory Phenotype on Dendritic Cells and Lymphocytes From Patients With Cutaneous Leishmaniasis.
Complement receptor CD46 co-stimulates optimal human CD8(+) T cell effector function via fatty acid metabolism.
Accumulation of T-helper 22 cells, interleukin-22 and myeloid-derived suppressor cells promotes gastric cancer progression in elderly patients.
IL-6 receptor blockade corrects defects of XIAP-deficient regulatory T cells.
Creation of an immunodeficient HLA-transgenic mouse (HUMAMICE) and functional validation of human immunity after transfer of HLA-matched human cells.
Targeted Disruption of TCF12 Reveals HEB as Essential in Human Mesodermal Specification and Hematopoiesis.
Dietary, nondigestible oligosaccharides and Bifidobacterium breve M-16V suppress allergic inflammation in intestine via targeting dendritic cell maturation.
Single-cell profiling reveals GPCR heterogeneity and functional patterning during neuroinflammation.
Heterogeneous Tumor-Immune Microenvironments among Differentially Growing Metastases in an Ovarian Cancer Patient.
Tax and Semaphorin 4D Released from Lymphocytes Infected with Human Lymphotropic Virus Type 1 and Their Effect on Neurite Growth.
HTLV-1 bZIP Factor Impairs Anti-viral Immunity by Inducing Co-inhibitory Molecule, T Cell Immunoglobulin and ITIM Domain (TIGIT).
PD-L1 expression and prognostic impact in glioblastoma.
Tumor-derived exosomes regulate expression of immune function-related genes in human T cell subsets.
Large-Scale Purification of r28M: A Bispecific scFv Antibody Targeting Human Melanoma Produced in Transgenic Cattle.
Dysbiotic bacteria translocate in progressive SIV infection.
A phase I dose-escalation clinical trial of a peptide-based human papillomavirus therapeutic vaccine with Candida skin test reagent as a novel vaccine adjuvant for treating women with biopsy-proven cervical intraepithelial neoplasia 2/3.
The Expression of Functional Vpx during Pathogenic SIVmac Infections of Rhesus Macaques Suppresses SAMHD1 in CD4+ Memory T Cells.
Expression of GM-CSF in T Cells Is Increased in Multiple Sclerosis and Suppressed by IFN-β Therapy.
The HIV-1 gp120 CD4-bound conformation is preferentially targeted by antibody-dependent cellular cytotoxicity-mediating antibodies in sera from HIV-1-infected individuals.
Elevated peripheral frequencies of Th22 cells: a novel potent participant in obesity and type 2 diabetes.
Increased peripheral proinflammatory T helper subsets contribute to cardiovascular complications in diabetic patients.
Aging and cytomegalovirus infection differentially and jointly affect distinct circulating T cell subsets in humans.
Ultradense array CGH and discovery of micro-copy number alterations and gene fusions in the cancer genome.
Genome-wide analysis of T cell responses during acute and latent simian varicella virus infections in rhesus macaques.
Memory T cells in latent Mycobacterium tuberculosis infection are directed against three antigenic islands and largely contained in a CXCR3+CCR6+ Th1 subset.
Transcriptional profiling of experimental CD8(+) lymphocyte depletion in rhesus macaques infected with simian immunodeficiency virus SIVmac239.
Assessment of immune interference, antagonism, and diversion following human immunization with biallelic blood-stage malaria viral-vectored vaccines and controlled malaria infection.
Reduced frequency of memory T cells and increased Th17 responses in patients with active tuberculosis.
CD4+CD8+ T cells represent a significant portion of the anti-HIV T cell response to acute HIV infection.
IL-17 and IFN-γ expression in lymphocytes from patients with active tuberculosis correlates with the severity of the disease.
Human CD8⁺ and CD4⁺ T cell memory to lymphocytic choriomeningitis virus infection.
SIV infection of rhesus macaques results in dysfunctional T- and B-cell responses to neo and recall Leishmania major vaccination.
Mechanisms underlying γδ T-cell subset perturbations in SIV-infected Asian rhesus macaques.
Disruption of intestinal CD4+ T cell homeostasis is a key marker of systemic CD4+ T cell activation in HIV-infected individuals.
Compromised gastrointestinal integrity in pigtail macaques is associated with increased microbial translocation, immune activation, and IL-17 production in the absence of SIV infection.
CD4-CCR5 interaction in intracellular compartments contributes to receptor expression at the cell surface.
Inhibition of gp160 and CD4 maturation in U937 cells after both defective and productive infections by human immunodeficiency virus type 1.
Deng B, Yang B, Chen J, Wang S, Zhang W, Guo Y, Han Y, Li H, Dang Y, Yuan Y, Dai X, Zang Y, Li Y, Li B
Journal for immunotherapy of cancer 2022 Jul;10(7)
Journal for immunotherapy of cancer 2022 Jul;10(7)
Elevations of monocyte and neutrophils, and higher levels of granulocyte colony-stimulating factor in peripheral blood in lung cancer patients.
Yin W, Lv J, Yao Y, Zhao Y, He Z, Wang Q, Cui L, Dai H
Thoracic cancer 2021 Oct;12(20):2680-2690
Thoracic cancer 2021 Oct;12(20):2680-2690
Repositioning liothyronine for cancer immunotherapy by blocking the interaction of immune checkpoint TIGIT/PVR.
Zhou X, Du J, Wang H, Chen C, Jiao L, Cheng X, Zhou X, Chen S, Gou S, Zhao W, Zhai W, Chen J, Gao Y
Cell communication and signaling : CCS 2020 Sep 7;18(1):142
Cell communication and signaling : CCS 2020 Sep 7;18(1):142
Boosting with AIDSVAX B/E Enhances Env Constant Region 1 and 2 Antibody-Dependent Cellular Cytotoxicity Breadth and Potency.
Easterhoff D, Pollara J, Luo K, Tolbert WD, Young B, Mielke D, Jha S, O'Connell RJ, Vasan S, Kim J, Michael NL, Excler JL, Robb ML, Rerks-Ngarm S, Kaewkungwal J, Pitisuttithum P, Nitayaphan S, Sinangil F, Tartaglia J, Phogat S, Kepler TB, Alam SM, Wiehe K, Saunders KO, Montefiori DC, Tomaras GD, Moody MA, Pazgier M, Haynes BF, Ferrari G
Journal of virology 2020 Jan 31;94(4)
Journal of virology 2020 Jan 31;94(4)
Human Fc Receptor-like 3 Inhibits Regulatory T Cell Function and Binds Secretory IgA.
Agarwal S, Kraus Z, Dement-Brown J, Alabi O, Starost K, Tolnay M
Cell reports 2020 Feb 4;30(5):1292-1299.e3
Cell reports 2020 Feb 4;30(5):1292-1299.e3
The Potential of Nanobody-Targeted Photodynamic Therapy to Trigger Immune Responses.
Beltrán Hernández I, Angelier ML, Del Buono D'Ondes T, Di Maggio A, Yu Y, Oliveira S
Cancers 2020 Apr 15;12(4)
Cancers 2020 Apr 15;12(4)
Efficient gene transfer into T lymphocytes by fiber-modified human adenovirus 5.
Lv Y, Xiao FJ, Wang Y, Zou XH, Wang H, Wang HY, Wang LS, Lu ZZ
BMC biotechnology 2019 Apr 24;19(1):23
BMC biotechnology 2019 Apr 24;19(1):23
Schistosoma mansoni rSm29 Antigen Induces a Regulatory Phenotype on Dendritic Cells and Lymphocytes From Patients With Cutaneous Leishmaniasis.
Lopes DM, Oliveira SC, Page B, Carvalho LP, Carvalho EM, Cardoso LS
Frontiers in immunology 2018;9:3122
Frontiers in immunology 2018;9:3122
Complement receptor CD46 co-stimulates optimal human CD8(+) T cell effector function via fatty acid metabolism.
Arbore G, West EE, Rahman J, Le Friec G, Niyonzima N, Pirooznia M, Tunc I, Pavlidis P, Powell N, Li Y, Liu P, Servais A, Couzi L, Fremeaux-Bacchi V, Placais L, Ferraro A, Walsh PR, Kavanagh D, Afzali B, Lavender P, Lachmann HJ, Kemper C
Nature communications 2018 Oct 10;9(1):4186
Nature communications 2018 Oct 10;9(1):4186
Accumulation of T-helper 22 cells, interleukin-22 and myeloid-derived suppressor cells promotes gastric cancer progression in elderly patients.
Chen X, Wang Y, Wang J, Wen J, Jia X, Wang X, Zhang H
Oncology letters 2018 Jul;16(1):253-261
Oncology letters 2018 Jul;16(1):253-261
IL-6 receptor blockade corrects defects of XIAP-deficient regulatory T cells.
Hsieh WC, Hsu TS, Chang YJ, Lai MZ
Nature communications 2018 Jan 31;9(1):463
Nature communications 2018 Jan 31;9(1):463
Creation of an immunodeficient HLA-transgenic mouse (HUMAMICE) and functional validation of human immunity after transfer of HLA-matched human cells.
Zeng Y, Liu B, Rubio MT, Wang X, Ojcius DM, Tang R, Durrbach A, Ru Z, Zhou Y, Lone YC
PloS one 2017;12(4):e0173754
PloS one 2017;12(4):e0173754
Targeted Disruption of TCF12 Reveals HEB as Essential in Human Mesodermal Specification and Hematopoiesis.
Li Y, Brauer PM, Singh J, Xhiku S, Yoganathan K, Zúñiga-Pflücker JC, Anderson MK
Stem cell reports 2017 Sep 12;9(3):779-795
Stem cell reports 2017 Sep 12;9(3):779-795
Dietary, nondigestible oligosaccharides and Bifidobacterium breve M-16V suppress allergic inflammation in intestine via targeting dendritic cell maturation.
de Kivit S, Kostadinova AI, Kerperien J, Morgan ME, Muruzabal VA, Hofman GA, Knippels LMJ, Kraneveld AD, Garssen J, Willemsen LEM
Journal of leukocyte biology 2017 Jul;102(1):105-115
Journal of leukocyte biology 2017 Jul;102(1):105-115
Single-cell profiling reveals GPCR heterogeneity and functional patterning during neuroinflammation.
Tischner D, Grimm M, Kaur H, Staudenraus D, Carvalho J, Looso M, Günther S, Wanke F, Moos S, Siller N, Breuer J, Schwab N, Zipp F, Waisman A, Kurschus FC, Offermanns S, Wettschureck N
JCI insight 2017 Aug 3;2(15)
JCI insight 2017 Aug 3;2(15)
Heterogeneous Tumor-Immune Microenvironments among Differentially Growing Metastases in an Ovarian Cancer Patient.
Jiménez-Sánchez A, Memon D, Pourpe S, Veeraraghavan H, Li Y, Vargas HA, Gill MB, Park KJ, Zivanovic O, Konner J, Ricca J, Zamarin D, Walther T, Aghajanian C, Wolchok JD, Sala E, Merghoub T, Snyder A, Miller ML
Cell 2017 Aug 24;170(5):927-938.e20
Cell 2017 Aug 24;170(5):927-938.e20
Tax and Semaphorin 4D Released from Lymphocytes Infected with Human Lymphotropic Virus Type 1 and Their Effect on Neurite Growth.
Quintremil S, Alberti C, Rivera M, Medina F, Puente J, Cartier L, Ramírez E, Tanaka Y, Valenzuela MA
AIDS research and human retroviruses 2016 Jan;32(1):68-79
AIDS research and human retroviruses 2016 Jan;32(1):68-79
HTLV-1 bZIP Factor Impairs Anti-viral Immunity by Inducing Co-inhibitory Molecule, T Cell Immunoglobulin and ITIM Domain (TIGIT).
Yasuma K, Yasunaga J, Takemoto K, Sugata K, Mitobe Y, Takenouchi N, Nakagawa M, Suzuki Y, Matsuoka M
PLoS pathogens 2016 Jan;12(1):e1005372
PLoS pathogens 2016 Jan;12(1):e1005372
PD-L1 expression and prognostic impact in glioblastoma.
Nduom EK, Wei J, Yaghi NK, Huang N, Kong LY, Gabrusiewicz K, Ling X, Zhou S, Ivan C, Chen JQ, Burks JK, Fuller GN, Calin GA, Conrad CA, Creasy C, Ritthipichai K, Radvanyi L, Heimberger AB
Neuro-oncology 2016 Feb;18(2):195-205
Neuro-oncology 2016 Feb;18(2):195-205
Tumor-derived exosomes regulate expression of immune function-related genes in human T cell subsets.
Muller L, Mitsuhashi M, Simms P, Gooding WE, Whiteside TL
Scientific reports 2016 Feb 4;6:20254
Scientific reports 2016 Feb 4;6:20254
Large-Scale Purification of r28M: A Bispecific scFv Antibody Targeting Human Melanoma Produced in Transgenic Cattle.
Spiesberger K, Paulfranz F, Egger A, Reiser J, Vogl C, Rudolf-Scholik J, Mayrhofer C, Grosse-Hovest L, Brem G
PloS one 2015;10(10):e0140471
PloS one 2015;10(10):e0140471
Dysbiotic bacteria translocate in progressive SIV infection.
Klase Z, Ortiz A, Deleage C, Mudd JC, Quiñones M, Schwartzman E, Klatt NR, Canary L, Estes JD, Brenchley JM
Mucosal immunology 2015 Sep;8(5):1009-20
Mucosal immunology 2015 Sep;8(5):1009-20
A phase I dose-escalation clinical trial of a peptide-based human papillomavirus therapeutic vaccine with Candida skin test reagent as a novel vaccine adjuvant for treating women with biopsy-proven cervical intraepithelial neoplasia 2/3.
Greenfield WW, Stratton SL, Myrick RS, Vaughn R, Donnalley LM, Coleman HN, Mercado M, Moerman-Herzog AM, Spencer HJ, Andrews-Collins NR, Hitt WC, Low GM, Manning NA, McKelvey SS, Smith D, Smith MV, Phillips AM, Quick CM, Jeffus SK, Hutchins LF, Nakagawa M
Oncoimmunology 2015 Oct;4(10):e1031439
Oncoimmunology 2015 Oct;4(10):e1031439
The Expression of Functional Vpx during Pathogenic SIVmac Infections of Rhesus Macaques Suppresses SAMHD1 in CD4+ Memory T Cells.
Shingai M, Welbourn S, Brenchley JM, Acharya P, Miyagi E, Plishka RJ, Buckler-White A, Kwong PD, Nishimura Y, Strebel K, Martin MA
PLoS pathogens 2015 May;11(5):e1004928
PLoS pathogens 2015 May;11(5):e1004928
Expression of GM-CSF in T Cells Is Increased in Multiple Sclerosis and Suppressed by IFN-β Therapy.
Rasouli J, Ciric B, Imitola J, Gonnella P, Hwang D, Mahajan K, Mari ER, Safavi F, Leist TP, Zhang GX, Rostami A
Journal of immunology (Baltimore, Md. : 1950) 2015 Jun 1;194(11):5085-93
Journal of immunology (Baltimore, Md. : 1950) 2015 Jun 1;194(11):5085-93
The HIV-1 gp120 CD4-bound conformation is preferentially targeted by antibody-dependent cellular cytotoxicity-mediating antibodies in sera from HIV-1-infected individuals.
Veillette M, Coutu M, Richard J, Batraville LA, Dagher O, Bernard N, Tremblay C, Kaufmann DE, Roger M, Finzi A
Journal of virology 2015 Jan;89(1):545-51
Journal of virology 2015 Jan;89(1):545-51
Elevated peripheral frequencies of Th22 cells: a novel potent participant in obesity and type 2 diabetes.
Zhao R, Tang D, Yi S, Li W, Wu C, Lu Y, Hou X, Song J, Lin P, Chen L, Sun L
PloS one 2014;9(1):e85770
PloS one 2014;9(1):e85770
Increased peripheral proinflammatory T helper subsets contribute to cardiovascular complications in diabetic patients.
Zhao RX, Li WJ, Lu YR, Qin J, Wu CL, Tian M, He TY, Yi SN, Tang DQ, Sun L, Chen L
Mediators of inflammation 2014;2014:596967
Mediators of inflammation 2014;2014:596967
Aging and cytomegalovirus infection differentially and jointly affect distinct circulating T cell subsets in humans.
Wertheimer AM, Bennett MS, Park B, Uhrlaub JL, Martinez C, Pulko V, Currier NL, Nikolich-Žugich D, Kaye J, Nikolich-Žugich J
Journal of immunology (Baltimore, Md. : 1950) 2014 Mar 1;192(5):2143-55
Journal of immunology (Baltimore, Md. : 1950) 2014 Mar 1;192(5):2143-55
Ultradense array CGH and discovery of micro-copy number alterations and gene fusions in the cancer genome.
Przybytkowski E, Aguilar-Mahecha A, Nabavi S, Tonellato PJ, Basik M
Methods in molecular biology (Clifton, N.J.) 2013;973:15-38
Methods in molecular biology (Clifton, N.J.) 2013;973:15-38
Genome-wide analysis of T cell responses during acute and latent simian varicella virus infections in rhesus macaques.
Haberthur K, Kraft A, Arnold N, Park B, Meyer C, Asquith M, Dewane J, Messaoudi I
Journal of virology 2013 Nov;87(21):11751-61
Journal of virology 2013 Nov;87(21):11751-61
Memory T cells in latent Mycobacterium tuberculosis infection are directed against three antigenic islands and largely contained in a CXCR3+CCR6+ Th1 subset.
Lindestam Arlehamn CS, Gerasimova A, Mele F, Henderson R, Swann J, Greenbaum JA, Kim Y, Sidney J, James EA, Taplitz R, McKinney DM, Kwok WW, Grey H, Sallusto F, Peters B, Sette A
PLoS pathogens 2013 Jan;9(1):e1003130
PLoS pathogens 2013 Jan;9(1):e1003130
Transcriptional profiling of experimental CD8(+) lymphocyte depletion in rhesus macaques infected with simian immunodeficiency virus SIVmac239.
Bosinger SE, Jochems SP, Folkner KA, Hayes TL, Klatt NR, Silvestri G
Journal of virology 2013 Jan;87(1):433-43
Journal of virology 2013 Jan;87(1):433-43
Assessment of immune interference, antagonism, and diversion following human immunization with biallelic blood-stage malaria viral-vectored vaccines and controlled malaria infection.
Elias SC, Collins KA, Halstead FD, Choudhary P, Bliss CM, Ewer KJ, Sheehy SH, Duncan CJ, Biswas S, Hill AV, Draper SJ
Journal of immunology (Baltimore, Md. : 1950) 2013 Feb 1;190(3):1135-47
Journal of immunology (Baltimore, Md. : 1950) 2013 Feb 1;190(3):1135-47
Reduced frequency of memory T cells and increased Th17 responses in patients with active tuberculosis.
Marín ND, París SC, Rojas M, García LF
Clinical and vaccine immunology : CVI 2012 Oct;19(10):1667-76
Clinical and vaccine immunology : CVI 2012 Oct;19(10):1667-76
CD4+CD8+ T cells represent a significant portion of the anti-HIV T cell response to acute HIV infection.
Frahm MA, Picking RA, Kuruc JD, McGee KS, Gay CL, Eron JJ, Hicks CB, Tomaras GD, Ferrari G
Journal of immunology (Baltimore, Md. : 1950) 2012 May 1;188(9):4289-96
Journal of immunology (Baltimore, Md. : 1950) 2012 May 1;188(9):4289-96
IL-17 and IFN-γ expression in lymphocytes from patients with active tuberculosis correlates with the severity of the disease.
Jurado JO, Pasquinelli V, Alvarez IB, Peña D, Rovetta AI, Tateosian NL, Romeo HE, Musella RM, Palmero D, Chuluyán HE, García VE
Journal of leukocyte biology 2012 Jun;91(6):991-1002
Journal of leukocyte biology 2012 Jun;91(6):991-1002
Human CD8⁺ and CD4⁺ T cell memory to lymphocytic choriomeningitis virus infection.
Kotturi MF, Swann JA, Peters B, Arlehamn CL, Sidney J, Kolla RV, James EA, Akondy RS, Ahmed R, Kwok WW, Buchmeier MJ, Sette A
Journal of virology 2011 Nov;85(22):11770-80
Journal of virology 2011 Nov;85(22):11770-80
SIV infection of rhesus macaques results in dysfunctional T- and B-cell responses to neo and recall Leishmania major vaccination.
Klatt NR, Vinton CL, Lynch RM, Canary LA, Ho J, Darrah PA, Estes JD, Seder RA, Moir SL, Brenchley JM
Blood 2011 Nov 24;118(22):5803-12
Blood 2011 Nov 24;118(22):5803-12
Mechanisms underlying γδ T-cell subset perturbations in SIV-infected Asian rhesus macaques.
Harris LD, Klatt NR, Vinton C, Briant JA, Tabb B, Ladell K, Lifson J, Estes JD, Price DA, Hirsch VM, Brenchley JM
Blood 2010 Nov 18;116(20):4148-57
Blood 2010 Nov 18;116(20):4148-57
Disruption of intestinal CD4+ T cell homeostasis is a key marker of systemic CD4+ T cell activation in HIV-infected individuals.
Gordon SN, Cervasi B, Odorizzi P, Silverman R, Aberra F, Ginsberg G, Estes JD, Paiardini M, Frank I, Silvestri G
Journal of immunology (Baltimore, Md. : 1950) 2010 Nov 1;185(9):5169-79
Journal of immunology (Baltimore, Md. : 1950) 2010 Nov 1;185(9):5169-79
Compromised gastrointestinal integrity in pigtail macaques is associated with increased microbial translocation, immune activation, and IL-17 production in the absence of SIV infection.
Klatt NR, Harris LD, Vinton CL, Sung H, Briant JA, Tabb B, Morcock D, McGinty JW, Lifson JD, Lafont BA, Martin MA, Levine AD, Estes JD, Brenchley JM
Mucosal immunology 2010 Jul;3(4):387-98
Mucosal immunology 2010 Jul;3(4):387-98
CD4-CCR5 interaction in intracellular compartments contributes to receptor expression at the cell surface.
Achour L, Scott MG, Shirvani H, Thuret A, Bismuth G, Labbé-Jullié C, Marullo S
Blood 2009 Feb 26;113(9):1938-47
Blood 2009 Feb 26;113(9):1938-47
Inhibition of gp160 and CD4 maturation in U937 cells after both defective and productive infections by human immunodeficiency virus type 1.
Bour S, Boulerice F, Wainberg MA
Journal of virology 1991 Dec;65(12):6387-96
Journal of virology 1991 Dec;65(12):6387-96
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
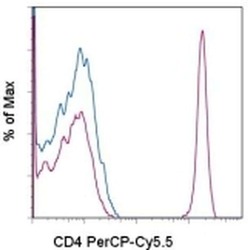
- Staining of normal human peripheral blood cells with Mouse IgG2b K Isotype Control PerCP-Cyanine5-5 (Product # 45-4732-82) (blue histogram) or Anti-Human CD4 PerCP-Cyanine5-5 (purple histogram). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
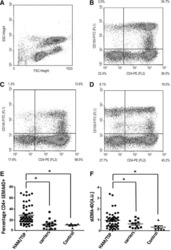
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
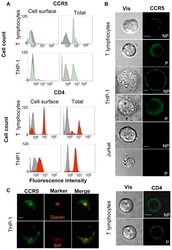
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
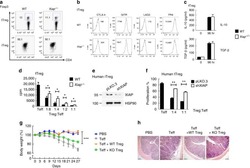
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
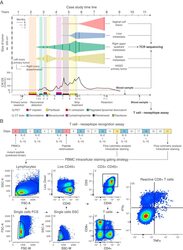
- Experimental details
- Figure S6 PBMCs Sample Timeline and T Cell-Neoepitope Recognition Assay, Related to Figures 4 and 5 and Table S5 (A) Blood samples obtained from the patient 550 and 978 days after resection were used for TCR sequencing and T cell - neoepitope recognition assays respectively. (B) Experimental setup and flow cytometry gating strategy for the T cell -neoepitope recognition assays (intracellular cytokine staining assay) with surface staining of CD3, CD4, CD8, CD45, and intracellular staining of IL-4, IFN-gamma, TNF-alpha. PBMC = peripheral blood mononuclear cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 Effects of TEX on protein expression and functions of T cells. In ( a ) down-regulation of CD69 protein expression on the surface of responder CD4 + Tconv after co-incubation with TEX. Activated CD4 + Tconv were co-incubated with TEX (10 ug protein) produced by the PCI-13 cells or with PBS for 12 h. The CD69 expression levels on CD4 + Tconv were then determined by flow cytometry (MFI) and were converted into MESF units based on calibration curves established with fluorescent calibration beads. The bar graphs show data (mean values +- SD) from 3 independent experiments performed with CD4 + Tconv obtained from different normal donors. The asterisks indicate p values at p < 0.0005. In ( b ) changes in expression levels of CD39 protein on the surface of resting CD4 + CD39 + Treg co-incubated with TEX produced by the PCI-13 cell line or DEX. The exosomes were used at the concentration of 10 ng protein/ assay. Exogenous ATP was added as described in Methods. Flow cytometry ( right ) shows up-regulation of MFI for CD39 in a representative experiment, and the bar graph summarizes results of three experiments performed with Treg obtained from different donors. In ( c ), Production levels of 5' AMP, adenosine and inosine by resting CD4 + CD39 + Treg co-incubated with TEX produced by the PCI-13 cell line. The data are from one of two experiments performed in the presence of exogenous ATP. The analyte levels were measured by mass spectrometry as described in Methods.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
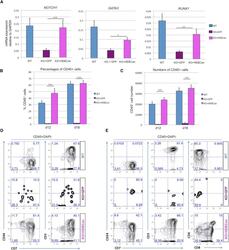
- Figure 7 HEBCan Rescues Hematopoiesis and T Cell Development in HEB -/- hESCs in OP9-DL4 Co-cultures (A) qRT-PCR analysis for the expression of hematopoietic genes in CD34 + cells sorted from WT, KO + GFP, and KO + HEBCan day-8 (d8) EBs. mRNA levels are shown relative to GAPDH. (B and C) Percentages (B) and numbers (C) of CD45 + cells in d12 and d18 OP9-DL4 co-cultures. (D and E) Flow-cytometric analysis of T cell development from WT, KO + GFP, and KO + HEBCan d8 EB-derived CD34 + cells at d12 (D) and d18 (E) of OP9-DL4 co-culture. Cells are gated on the CD45 + DAPI - population. Error bars represent mean +- SD (n = 3 independent experiments). * p < 0.05, ** p < 0.01, *** p < 0.005 by Student's t test. Plots in (B), (D), and (E) are representative of three independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
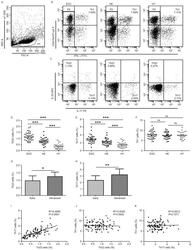
- Figure 1. Flow cytometric analysis was used to determine the distribution of Th22, Th17 and Th1 cells in EGC, HE and HY ( Fig. 1 ). Flow cytometric analysis of Th22, Th17 and Th1 cells in peripheral whole blood from EGC (n=39), HE (n=32) and HY (n=31). (A) Lymphocytes were gated in P1 using flow cytometry. CD4 + IFN-gamma - lymphocytes were gated in P2 using flow cytometry, and representative results of flow cytometric analyses for (B) Th1 (CD4 + IFN-gamma + ), (C) Th22 (CD4 + IFN-gamma - IL-17 - IL-22 + ) and Th17 (CD4 + IFN-gamma - IL-17 + IL-22 - ) cells in the three groups of subjects are presented. The number of cells stained in EGC, HE and HY in P2 were 2,654, 4,696 and 5,185, respectively. The proportion of (D) Th22, (E) Th17 and (F) Th1 cells in the three groups of subjects. The proportion of (G) Th22 and (H) Th17 cells in peripheral whole blood derived from patients with early (n=13) or advanced (n=26) gastric cancer. The association between the proportion of (I) Th22 and Th17 cells, (J) Th22 and Th1 cells, and (K) Th17 and Th1 cells, in peripheral whole blood of all subjects. *P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
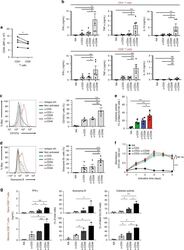
- Fig. 1 CD46 co-stimulation provides superior support for CTL activity. a CD46 expression on the surface of resting human CD4 + and CD8 + T cells assessed by FACS analysis ( n = 4, gating strategy in Supplementary Fig. 7a ). b Comparison of IFN-gamma, TNF-alpha and IL-10 secretion by CD3 + CD46-activated T cells. Purified CD4 + and CD8 + T cells from healthy donors were left non-activated (NA) or stimulated with immobilized antibodies to CD3, CD3 + CD28 or CD3 + CD46 and cytokines measured 60 h post activation ( n = 5). c, d Degranulation (CD107a staining, ( c )) and granzyme B expression ( d ) by CD8 + T cells upon CD46 co-stimulation. CD8 + T cells were stimulated as in ( a ) and CD107a and granzyme B expression assessed with left panels showing representative cytometry images and right panels corresponding quantifications ( n = 3, gating strategy in Supplementary Fig. 7b ). e Killing activity of CD46-activated CD8 + T cells. T cells were stimulated as depicted for 24 h and cytotoxic activity of differently activated CD8 + T cells towards DU145 target cells assessed 24 h post co-culture of T cells and DU145 cells ( n = 4, gating strategy in Supplementary Fig. 7c ). f Effect of CD46 co-stimulation on CD8 + T-cell proliferation. Purified T cells were activated as indicated for 5 days and cell proliferation measured each day ( n = 4) (black circles, non-activated cells; green, blue, and red circles, CD3, CD3 + CD28 or CD3 + CD46-activated cells, res
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry