Antibody data
- Antibody Data
- Antigen structure
- References [15]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [9]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 17-9715-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-AKT1 (Ser473) Monoclonal Antibody (SDRNR), APC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: This SDRNR monoclonal antibody recognizes human and mouse AKT (also known as Protein Kinase B (PKB)) when phosphorylated on S473. AKT is a serine/threonine protein kinase that plays a key role in multiple cellular processes including metabolism, proliferation, apoptosis/survival, and migration. There are three homologous isoforms of AKT: AKT1, AKT2, and AKT3. AKT is activated by binding of its pleckstrin homology (PH) domain to membrane phospholipids and by phosphorylation. Phosphorylation of AKT at T308 by PDK1 and at S473 is required for full activation of this kinase. AKT promotes cell survival by inhibiting apoptosis via phosphorylation and inactivation of several targets including Bad, Foxo1, c-Raf, and caspase-9. Deregulation of AKT has been implicated as a major contributing factor in many types of cancer. AKT is negatively regulated by the phosphatase PTEN as well as by the chemical inhibitor LY294002. Specificity of this SDRNR clone was determined by ELISA, flow cytometry, and western blotting. Applications Reported: This SDRNR antibody has been reported for use in intracellular staining followed by flow cytometric analysis. Applications Tested: This SDRNR antibody has been pre-titrated and tested by intracellular staining followed by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.5 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Staining Protocol: All protocols work well for this monoclonal antibody. Use of Protocol A: Two-step protocol: intracellular (cytoplasmic) proteins allows for the greatest flexibility for detection of surface and intracellular (cytoplasmic) proteins. Use of Protocol B: One-step protocol: intracellular (nuclear) proteins is recommended for staining of transcription factors in conjunction with surface and phosphorylated intracellular (cytoplasmic) proteins. Protocol C: Two-step protocol: Fixation/Methanol allows for the greatest discrimination of phospho-specific signaling between unstimulated and stimulated samples, but with limitations on the ability to stain specific surface proteins (refer to "Clone Performance Following Fixation/Permeabilization" located in the Best Protocols Section under the Resources tab online). All Protocols can be found in the Flow Cytometry Protocols: "Staining Intracellular Antigens for Flow Cytometry Protocol" located in the Best Protocols Section under the Resources tab online. Excitation: 633-647 nm; Emission: 660 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human, Mouse
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- SDRNR
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Combinatorial immunotherapy of N-803 (IL-15 superagonist) and dinutuximab with ex vivo expanded natural killer cells significantly enhances in vitro cytotoxicity against GD2(+) pediatric solid tumors and in vivo survival of xenografted immunodeficient NSG mice.
Novel cytokine-antibody fusion protein, N-820, to enhance the functions of ex vivo expanded natural killer cells against Burkitt lymphoma.
AKT but not MYC promotes reactive oxygen species-mediated cell death in oxidative culture.
IL-15 negatively regulates curdlan-induced IL-23 production by human monocyte-derived dendritic cells and subsequent Th17 response.
Fc Receptor-Like 1 as a Promising Target for Immunotherapeutic Interventions of B-Cell-Related Disorders.
OX40 expression in neutrophils promotes hepatic ischemia/reperfusion injury.
Crosstalks between mTORC1 and mTORC2 variagate cytokine signaling to control NK maturation and effector function.
MicroRNA-126 deficiency enhanced the activation and function of CD4(+) T cells by elevating IRS-1 pathway.
Autophagy orchestrates the regulatory program of tumor-associated myeloid-derived suppressor cells.
Store-Operated Ca(2+) Entry Controls Clonal Expansion of T Cells through Metabolic Reprogramming.
Deletion of PIKfyve alters alveolar macrophage populations and exacerbates allergic inflammation in mice.
MiR-128-2 inhibits common lymphoid progenitors from developing into progenitor B cells.
Protein kinase B controls transcriptional programs that direct cytotoxic T cell fate but is dispensable for T cell metabolism.
T cell receptor signaling controls Foxp3 expression via PI3K, Akt, and mTOR.
Protein kinase B (Akt) regulation and function in T lymphocytes.
Chu Y, Nayyar G, Jiang S, Rosenblum JM, Soon-Shiong P, Safrit JT, Lee DA, Cairo MS
Journal for immunotherapy of cancer 2021 Jul;9(7)
Journal for immunotherapy of cancer 2021 Jul;9(7)
Novel cytokine-antibody fusion protein, N-820, to enhance the functions of ex vivo expanded natural killer cells against Burkitt lymphoma.
Chu Y, Nayyar G, Kham Su N, Rosenblum JM, Soon-Shiong P, Lee J, Safrit JT, Barth M, Lee D, Cairo MS
Journal for immunotherapy of cancer 2020 Oct;8(2)
Journal for immunotherapy of cancer 2020 Oct;8(2)
AKT but not MYC promotes reactive oxygen species-mediated cell death in oxidative culture.
Zheng D, Sussman JH, Jeon MP, Parrish ST, MacMullan MA, Delfarah A, Graham NA
Journal of cell science 2020 Apr 9;133(7)
Journal of cell science 2020 Apr 9;133(7)
IL-15 negatively regulates curdlan-induced IL-23 production by human monocyte-derived dendritic cells and subsequent Th17 response.
Eken A, Okus Z, Erdem S, Azizoglu ZB, Haliloglu Y, Bicer A, Gur TN, Yilmaz E, Karakukcu M, Altuntas HD, Canatan H
Northern clinics of Istanbul 2019;6(4):379-387
Northern clinics of Istanbul 2019;6(4):379-387
Fc Receptor-Like 1 as a Promising Target for Immunotherapeutic Interventions of B-Cell-Related Disorders.
Yousefi Z, Sharifzadeh S, Yar-Ahmadi V, Andalib A, Eskandari N
Biomarker insights 2019;14:1177271919882351
Biomarker insights 2019;14:1177271919882351
OX40 expression in neutrophils promotes hepatic ischemia/reperfusion injury.
Jin H, Zhang C, Sun C, Zhao X, Tian D, Shi W, Tian Y, Liu K, Sun G, Xu H, Zhang D
JCI insight 2019 Nov 1;4(21)
JCI insight 2019 Nov 1;4(21)
Crosstalks between mTORC1 and mTORC2 variagate cytokine signaling to control NK maturation and effector function.
Wang F, Meng M, Mo B, Yang Y, Ji Y, Huang P, Lai W, Pan X, You T, Luo H, Guan X, Deng Y, Yuan S, Chu J, Namaka M, Hughes T, Ye L, Yu J, Li X, Deng Y
Nature communications 2018 Nov 19;9(1):4874
Nature communications 2018 Nov 19;9(1):4874
MicroRNA-126 deficiency enhanced the activation and function of CD4(+) T cells by elevating IRS-1 pathway.
Chu F, Hu Y, Zhou Y, Guo M, Lu J, Zheng W, Xu H, Zhao J, Xu L
Clinical and experimental immunology 2018 Feb;191(2):166-179
Clinical and experimental immunology 2018 Feb;191(2):166-179
Autophagy orchestrates the regulatory program of tumor-associated myeloid-derived suppressor cells.
Alissafi T, Hatzioannou A, Mintzas K, Barouni RM, Banos A, Sormendi S, Polyzos A, Xilouri M, Wielockx B, Gogas H, Verginis P
The Journal of clinical investigation 2018 Aug 31;128(9):3840-3852
The Journal of clinical investigation 2018 Aug 31;128(9):3840-3852
Store-Operated Ca(2+) Entry Controls Clonal Expansion of T Cells through Metabolic Reprogramming.
Vaeth M, Maus M, Klein-Hessling S, Freinkman E, Yang J, Eckstein M, Cameron S, Turvey SE, Serfling E, Berberich-Siebelt F, Possemato R, Feske S
Immunity 2017 Oct 17;47(4):664-679.e6
Immunity 2017 Oct 17;47(4):664-679.e6
Deletion of PIKfyve alters alveolar macrophage populations and exacerbates allergic inflammation in mice.
Kawasaki T, Ito K, Miyata H, Akira S, Kawai T
The EMBO journal 2017 Jun 14;36(12):1707-1718
The EMBO journal 2017 Jun 14;36(12):1707-1718
MiR-128-2 inhibits common lymphoid progenitors from developing into progenitor B cells.
Yang Y, Xu J, Chen H, Fei X, Tang Y, Yan Y, Zhang H, Zhang J
Oncotarget 2016 Apr 5;7(14):17520-31
Oncotarget 2016 Apr 5;7(14):17520-31
Protein kinase B controls transcriptional programs that direct cytotoxic T cell fate but is dispensable for T cell metabolism.
Macintyre AN, Finlay D, Preston G, Sinclair LV, Waugh CM, Tamas P, Feijoo C, Okkenhaug K, Cantrell DA
Immunity 2011 Feb 25;34(2):224-36
Immunity 2011 Feb 25;34(2):224-36
T cell receptor signaling controls Foxp3 expression via PI3K, Akt, and mTOR.
Sauer S, Bruno L, Hertweck A, Finlay D, Leleu M, Spivakov M, Knight ZA, Cobb BS, Cantrell D, O'Connor E, Shokat KM, Fisher AG, Merkenschlager M
Proceedings of the National Academy of Sciences of the United States of America 2008 Jun 3;105(22):7797-802
Proceedings of the National Academy of Sciences of the United States of America 2008 Jun 3;105(22):7797-802
Protein kinase B (Akt) regulation and function in T lymphocytes.
Cantrell D
Seminars in immunology 2002 Feb;14(1):19-26
Seminars in immunology 2002 Feb;14(1):19-26
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
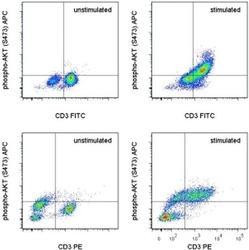
- Experimental details
- TOP: Normal human peripheral blood cells were unstimulated (left) or stimulated with Anti-Human CD3 Functional Grade Purified (Product # 16-0037-81) and Anti-Human CD28 Functional Grade Purified (Product # 16-0289-81) for 48 hours (right). The cells were then intracellularly stained with Anti-Human CD3 FITC (Product # 11-0036-42) and Anti-Human/Mouse phospho-AKT (S473) APC using the Intracellular Fixation & Permeabilization Buffer Set (Product # 88-8824-00) and protocol. Cells in the lymphocyte gate were used for analysis. BOTTOM: Mouse splenocytes were unstimulated (left) or stimulated with Anti-Mouse CD3e Functional Grade Purified (Product # 16-0031-82) and Anti-Mouse CD28 Functional Grade Purified (Product # 16-0281-82) for 48 hours (right). The cells were then intracellularly stained with Anti-Mouse CD3e PE (Product # 12-0031-82) and Anti-Human/Mouse phospho-AKT (S473) APC using the Intracellular Fixation & Permeabilization Buffer Set (Product # 88-8824-00) and protocol. Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
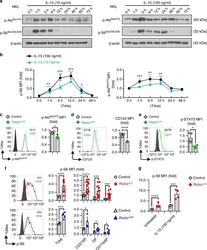
- Fig. 4 Crosstalk between mTORC1 and mTORC2 during NK cell activation. a Immunoblotting to detect the phosphorylation (p-) of S6 at ser235/236 (p-S6 ser235/236 ) and Akt at Ser473 (p-Akt ser473 ) in NKL cells after stimulation with IL-15 (10 ng/ml) (left) or IL-15 (100 ng/ml) (right) for the indicated time periods. beta-Actin was used as an internal control. The data shown are representative of three independent experiments. Uncropped gels are shown in Supplementary Fig. 8 . b Intracellular flow cytometric analysis of p-S6 ser235/236 and p-Akt ser473 in murine splenic NK cells (CD3 - CD19 - NK1.1 + ) after stimulation with IL-15 (100 or 10 ng/ml) for the indicated time periods. The MFI of p-S6 ser235/236 or p-Akt ser473 at each time point was normalized to the baseline reading at the 0-h time point for each individual mouse. Four independent individual mice were used. c - e Flow cytometric analysis and cumulative results for p-Akt Ser473 ( c ), CD122 ( d ), and p-STAT5 Y694 ( e ) in splenic NK cells (CD3 - CD19 - NK1.1 + ) from control versus Raptor NK mice. f Intracellular flow cytometric analysis and cumulative results for p-S6 ser235/236 in total splenic NK cells (CD3 - CD19 - NK1.1 + ) and the indicated subpopulations thereof from control versus Rictor / (top) or control versus Rictor NK (bottom) mice. g Statistical quantification of p-S6 ser235/236 (MFI relative to the untreated control littermates) in splenic NK cells (CD3 - CD19 - NK1.1 + ) wit
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
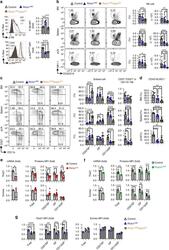
- Fig. 5 mTORC2 and mTORC1 promote NK cell maturation by controlling the expression of Tbx21 and Eomes in a cooperative and nonredundant manner. a Intracellular flow cytometric analysis and cumulative results for the phosphorylation (p-) of S6 at ser235/236 (p-S6 ser235/236 ) (top) and Akt at Ser473 (p-Akt Ser473 ) (bottom) in splenic NK cells (CD3 - CD19 - NK1.1 + ) from mice of the indicated genotype. b Cumulative ratio and enumeration of NK cells (CD3 - CD19 - NK1.1 + ) in the BM, spleen, and peripheral lymph nodes (pLNs) from mice of the indicated genotype. c Flow cytometric analysis and cumulative frequencies of subpopulations of NK cells (CD3 - CD19 - NK1.1 + NKp46 + ) in the BM, spleen, and pLNs (left) and the calculated ratio of CD27 - versus CD27 + cells among CD11b + NK cells (right). d The cumulative frequencies depicting the CD43 + KLRG1 + subset of NK cells (CD3 - CD19 - NK1.1 + NKp46 + ) in the BM, spleen, and pLNs from mice of the indicated genotype. e , f Tbx21 and Eomes mRNA and protein expression in control versus Rictor / mice ( e ) and control versus Raptor NK mice ( f ), as assessed by quantitative RT-PCR and flow cytometry, respectively. Purified splenic NK cells were used for quantitative RT-PCR (left). Cumulative data for Tbx21 and Eomes protein expression in total splenic NK cells (CD3 - CD19 - NK1.1 + NKp46 + ) and the indicated subpopulations thereof were analyzed by flow cytometry (right). g Cumulative data for Tbx21 and Eomes pr
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
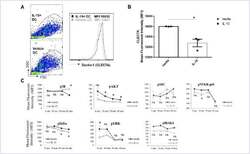
- FIGURE 3 IL-15 exposure downregulates surface Dectin-1 receptor expression and impacts signalling in DCs (A) After five days of culture in with IL-4/GM-CSF or IL-4/GM-CSF/IL-15 DCs were surface stained for Clec7 (Dectin-1) and a representative flow graph and (B) quantification of mean fluorescent intensity (MFI) as bar graph are presented. The experiment was run in triplicate wells and a representative result from a single donor was shown, the experiment was repeated with four different donors. (C) DCs from ""A"" were stimulated with curdlan (50 ug/ml) and phosphorylation of p38, AKT, SRC, NFKB p65, IkBa, ERK1/2 and IRAK4 was measured by phospho-flow assay. A representative result from PBMCs of a single donor was charted. *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
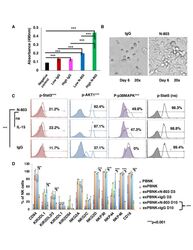
- Figure 1 N-803 increased the viability and proliferation of exPBNK with enhanced p-Stat3, p-Stat5, pAKT, p-p38MAPK and NK activating receptors. PBMNCs were stimulated with irradiated genetically modified K562-mbIL21-41BBL cells for 2-3 weeks. (A) Purified exPBNK cells were cultured in complete medium with 0.35 ng/mL (low) or 3.5 ng/mL (high) N-803 or molar equivalent dose of IgG for 3 days. NK viability and proliferation were monitored by MTS assays. The amount of 490 nm absorbance is directly proportional to the number of living exPBNK cells in the culture. The exPBNK cells with N-803 at 0.35 ng/mL or 3.5 ng/mL have significantly higher viability as compared with IgG or medium controls (p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
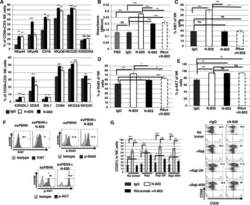
- Figure 1 N-820 enhanced the expression of NK activating receptors and proliferation of exPBNK with enhanced Ki67, p-Stat5, and CD25 levels. The percentages of NK activating and inhibitory receptors on exPBNK surface were monitored by flow cytometry analysis at day 3 (A). The NK proliferation was monitored by CellTiter 96 AQueous One solution cell proliferation assay (Promega) according to the manufacturer's instruction (B). Intracellular Ki67 (C), Phosphorylated STAT5 (p-STAT5) (D) and phosphorylated Akt (p-AKT) (E) were monitored by flow cytometry analysis at day 3. Ki67 (p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4. The effect of FCRL1 knockdown the extent of the expression of PI3K /p-AKT pathway in the Ramos and Daudi cells.(a) The real-time PCR approach revealed a significant decrease in theextent of the PI3K gene expression in the BL cellsafter 3 days of the infection procedure. The extent of the expression ofthe p-AKT protein was measured by (b) cytometry and (c) using the FlowJosoftware on day 4 of infection procedure. Shaded-matched representsmatched isotype control antibody, blue represents uninfected cells,green represents control cells, and red represents treated cells. Dataare shown as mean +- standard deviation. BL indicates Burkitt lymphoma; FCRL, Fc receptor-like; PCR, polymerasechain reaction.
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry