Antibody data
- Antibody Data
- Antigen structure
- References [10]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 25-1178-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD117 (c-Kit) Monoclonal Antibody (104D2), PE-Cyanine7, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 104D2 monoclonal antibody reacts with human CD117, also known as c-Kit, Steel factor receptor and stem cell factor receptor. A member of the tyrosine kinase receptor family, this 145 kDa molecule is expressed by hematopoietic progenitor cell subsets and mast cells. The interaction of c-Kit and Steel factor promotes proliferation and differentiation of hematopoietic progenitor cells and mast cell differentiation. CD117 is also expressed by melanocytes and plays a role in signaling and activation of these cells. Applications Reported: This 104D2 antibody has been reported for use in flow cytometric analysis. Applications Tested: This 104D2 antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.125 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Light sensitivity: This tandem dye is sensitive photo-induced oxidation. Please protect this vial and stained samples from light. Fixation: Samples can be stored in IC Fixation Buffer (Product # 00-822-49) (100 µL cell sample + 100 µL IC Fixation Buffer) or 1-step Fix/Lyse Solution (Product # 00-5333-54) for up to 3 days in the dark at 4°C with minimal impact on brightness and FRET efficiency/compensation. Some generalizations regarding fluorophore performance after fixation can be made, but clone specific performance should be determined empirically. Excitation: 488-561 nm; Emission: 775 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 104D2
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Human oral mucosa cell atlas reveals a stromal-neutrophil axis regulating tissue immunity.
Coordinated alterations in RNA splicing and epigenetic regulation drive leukaemogenesis.
CD62L Is a Functional and Phenotypic Marker for Circulating Innate Lymphoid Cell Precursors.
Tracking of epigenetic changes during hematopoietic differentiation of induced pluripotent stem cells.
Chemically-Defined, Xeno-Free, Scalable Production of hPSC-Derived Definitive Endoderm Aggregates with Multi-Lineage Differentiation Potential.
Genetically engineered red cells expressing single domain camelid antibodies confer long-term protection against botulinum neurotoxin.
Modelling IRF8 Deficient Human Hematopoiesis and Dendritic Cell Development with Engineered iPS Cells.
Peritoneal carcinomatosis of colorectal cancer is characterized by structural and functional reorganization of the tumor microenvironment inducing senescence and proliferation arrest in cancer cells.
PPAR-α and glucocorticoid receptor synergize to promote erythroid progenitor self-renewal.
CD52 is a molecular target in advanced systemic mastocytosis.
Williams DW, Greenwell-Wild T, Brenchley L, Dutzan N, Overmiller A, Sawaya AP, Webb S, Martin D, NIDCD/NIDCR Genomics and Computational Biology Core, Hajishengallis G, Divaris K, Morasso M, Haniffa M, Moutsopoulos NM
Cell 2021 Jul 22;184(15):4090-4104.e15
Cell 2021 Jul 22;184(15):4090-4104.e15
Coordinated alterations in RNA splicing and epigenetic regulation drive leukaemogenesis.
Yoshimi A, Lin KT, Wiseman DH, Rahman MA, Pastore A, Wang B, Lee SC, Micol JB, Zhang XJ, de Botton S, Penard-Lacronique V, Stein EM, Cho H, Miles RE, Inoue D, Albrecht TR, Somervaille TCP, Batta K, Amaral F, Simeoni F, Wilks DP, Cargo C, Intlekofer AM, Levine RL, Dvinge H, Bradley RK, Wagner EJ, Krainer AR, Abdel-Wahab O
Nature 2019 Oct;574(7777):273-277
Nature 2019 Oct;574(7777):273-277
CD62L Is a Functional and Phenotypic Marker for Circulating Innate Lymphoid Cell Precursors.
Bar-Ephraim YE, Koning JJ, Burniol Ruiz E, Konijn T, Mourits VP, Lakeman KA, Boon L, Bögels M, van Maanen JP, Den Haan JMM, van Egmond M, Bouma G, Reijmers RM, Mebius RE
Journal of immunology (Baltimore, Md. : 1950) 2019 Jan 1;202(1):171-182
Journal of immunology (Baltimore, Md. : 1950) 2019 Jan 1;202(1):171-182
Tracking of epigenetic changes during hematopoietic differentiation of induced pluripotent stem cells.
Cypris O, Frobel J, Rai S, Franzen J, Sontag S, Goetzke R, Szymanski de Toledo MA, Zenke M, Wagner W
Clinical epigenetics 2019 Feb 4;11(1):19
Clinical epigenetics 2019 Feb 4;11(1):19
Chemically-Defined, Xeno-Free, Scalable Production of hPSC-Derived Definitive Endoderm Aggregates with Multi-Lineage Differentiation Potential.
Sahabian A, Sgodda M, Naujok O, Dettmer R, Dahlmann J, Manstein F, Cantz T, Zweigerdt R, Martin U, Olmer R
Cells 2019 Dec 4;8(12)
Cells 2019 Dec 4;8(12)
Genetically engineered red cells expressing single domain camelid antibodies confer long-term protection against botulinum neurotoxin.
Huang NJ, Pishesha N, Mukherjee J, Zhang S, Deshycka R, Sudaryo V, Dong M, Shoemaker CB, Lodish HF
Nature communications 2017 Sep 4;8(1):423
Nature communications 2017 Sep 4;8(1):423
Modelling IRF8 Deficient Human Hematopoiesis and Dendritic Cell Development with Engineered iPS Cells.
Sontag S, Förster M, Qin J, Wanek P, Mitzka S, Schüler HM, Koschmieder S, Rose-John S, Seré K, Zenke M
Stem cells (Dayton, Ohio) 2017 Apr;35(4):898-908
Stem cells (Dayton, Ohio) 2017 Apr;35(4):898-908
Peritoneal carcinomatosis of colorectal cancer is characterized by structural and functional reorganization of the tumor microenvironment inducing senescence and proliferation arrest in cancer cells.
Seebauer CT, Brunner S, Glockzin G, Piso P, Ruemmele P, Schlitt HJ, Geissler EK, Fichtner-Feigl S, Kesselring R
Oncoimmunology 2016;5(12):e1242543
Oncoimmunology 2016;5(12):e1242543
PPAR-α and glucocorticoid receptor synergize to promote erythroid progenitor self-renewal.
Lee HY, Gao X, Barrasa MI, Li H, Elmes RR, Peters LL, Lodish HF
Nature 2015 Jun 25;522(7557):474-7
Nature 2015 Jun 25;522(7557):474-7
CD52 is a molecular target in advanced systemic mastocytosis.
Hoermann G, Blatt K, Greiner G, Putz EM, Berger A, Herrmann H, Cerny-Reiterer S, Gleixner KV, Walz C, Hoetzenecker K, Müllauer L, Reiter A, Sotlar K, Sexl V, Valent P, Mayerhofer M
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2014 Aug;28(8):3540-51
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2014 Aug;28(8):3540-51
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of normal human peripheral blood cells with the Human Hematopoietic Lineage APC Cocktail (Product # 22-7776-72), Anti-Human CD34 FITC (Product # 11-0349-42) and Mouse IgG1 K Isotype Control PE-Cyanine7 (Product # 25-4714-80) (left) or Anti-Human CD117 (c-kit) PE-Cyanine7 (right). Lineage negative/low cells were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
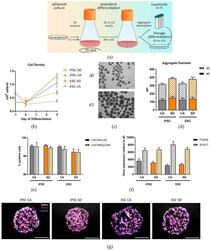
- Figure 1 Generation and characterization of DE aggregates in Erlenmeyer flasks. ( a ) Schematic overview of definitive endoderm differentiation in suspension. ( b ) Cell density analyzed at day 0 and day 3 of differentiation (n = 4-6). ( c ) Representative brightfield image of iPSC aggregates at day 0 and day 3 of differentiation (n = 4-6). ( d ) Aggregate diameters measured at day 0 and day 3 of differentiation. ( e ) Definitive endoderm efficiency quantification based on flow cytometry analysis of CXCR4/c-Kit and CXCR4/EpCAM double positive cells at day 3 of differentiation (n = 12-15). ( f ) qRT-PCR analysis of FOXA2 and SOX17 expression at day 3 of differentiation (n = 6-9). ( g ) Immunostaining of SOX17 (white) and FOXA2 (red), and nuclear stain DAPI (blue) of DE aggregates at day 3 of differentiation (n = 3). Scale bar, 100 mum. Each value of gene expression was first normalized to the reference genes and then to day 0 undifferentiated cells. Values are represented as the mean +- SEM. All n values correspond to independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
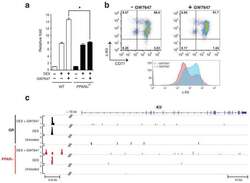
- Extended Figure 7 a, Real-time RT-PCR analysis of Kit gene expression in wild-type and PPARalpha -/- mouse BFU-E cells untreated or treated with DEX with or without addition of GW7647 (* p < 0.05; Student t test. Error bars represent mean +- S.D. from three independent experiments.); b , Human CD34+ cells were treated with or without GW7647 as described in the legend to Figure 2 . (top) At day 9 of culture, cell surface KIT and CD71 expression were analyzed by flow cytometry. (bottom) A representative histogram of KIT expression in cells treated or untreated with GW7647; c , ChIP-Seq occupancy signal map of GR and PPARalpha across the Kit locus in BFU-E cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
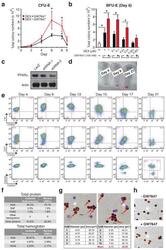
- Extended Figure 2 Human CD34 + Erythroid Differentiation System a , Total CFU-E colonies formed during day 0-9. CFU-E colony numbers were quantified by plating 1000 cells from various time points during day 0-9 of the human CD34 + erythroid culture on methylcellulose. CFU-E colonies were quantified after 12-14 days. Total CFU-E colony numbers in culture under conditions without GW7647 (black line) or with GW7647 (red line) were calculated using the total cell numbers at corresponding time points in Figure 2a. b , Human CD34+ cells were treated at day 1 with 100 nM GW7647 with or without DEX at the concentration indicated in the figure. At day 6, total cell numbers were counted and cells were collected for BFU-E colony assays. c , Protein expression of PPARalpha demonstrating shRNA knock-down efficiency via lentiviral transduction. LacZ shRNA is used as a control. shRNA-1 and -2 are both specific for PPARalpha . shRNA-2 has higher knock-down efficiency. d , Cell pellets of 1 million cells demonstrating hemoglobin accumulation during the differentiation process. e , Flow cytometry analyses of erythroid markers during the 21-day human CD34 + erythroid culture. (top row) c-kit vs. CD235a; (middle row) CD71 vs. CD235a. Note the sequential induction of c-kit, CD71 and CD235a, as well as the sequential down-regulation of c-kit and CD71, (bottom row) Enucleated reticulocytes are CD235a + Hoechst - , nuclei are CD235a - Hoechst + , and nucleated erythroblasts are CD235a + Hoechst + .
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry