MA1-066
antibody from Invitrogen Antibodies
Targeting: IGF2R
CD222, CI-M6PR, CI-MPR, CIMPR, M6P-R, MPR1, MPR300, MPRI
 Western blot
Western blot Immunocytochemistry
Immunocytochemistry Immunoprecipitation
Immunoprecipitation Immunohistochemistry
Immunohistochemistry Flow cytometry
Flow cytometry Other assay
Other assayAntibody data
- Antibody Data
- Antigen structure
- References [46]
- Comments [0]
- Validations
- Immunocytochemistry [10]
- Other assay [15]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA1-066 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IGF2R Monoclonal Antibody (2G11)
- Antibody type
- Monoclonal
- Antigen
- Purifed from natural sources
- Description
- MA1-066 detects mannose 6-phospate receptor (MPR) from human, rat, monkey and bovine tissues. This antibody does not detect Chinese hamster ovary cell MPR. MA1-066 has been successfully used in Western blot, immunofluorescence, EM immunocytochemistry and immunoprecipitation procedures. By Western blot, this antibody detects a ~300 kDa protein representing MPR in HeLa cell extract under non-reducing conditions. Immunofluorescence staining of MPR in HeLa cells with MA1-066 results in perinuclear staining. MA1-066 immunogen is purified bovine ~300 kDa Cl-MPR. This antibody is shown to recognize an epitope in the extracellular domain of MPR.
- Reactivity
- Human, Rat, Bovine
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 2G11
- Vial size
- 100 μg
- Concentration
- 1 mg/mL
- Storage
- -20°C, Avoid Freeze/Thaw Cycles
Submitted references Coronin 1C restricts endosomal branched actin to organize ER contact and endosome fission.
Fate of the Fc fusion protein aflibercept in retinal endothelial cells: competition of recycling and degradation.
A Novel Class of ER Membrane Proteins Regulates ER-Associated Endosome Fission.
Amino acids stimulate the endosome-to-Golgi trafficking through Ragulator and small GTPase Arl5.
Caspase-mediated proteolysis of the sorting nexin 2 disrupts retromer assembly and potentiates Met/hepatocyte growth factor receptor signaling.
Sequence-dependent cargo recognition by SNX-BARs mediates retromer-independent transport of CI-MPR.
A Trematode Parasite Derived Growth Factor Binds and Exerts Influences on Host Immune Functions via Host Cytokine Receptor Complexes.
Excess sphingomyelin disturbs ATG9A trafficking and autophagosome closure.
Human Copper Chaperone Atox1 Translocates to the Nucleus but does not Bind DNA In Vitro.
Human Copper Chaperone Atox1 Translocates to the Nucleus but does not Bind DNA In Vitro.
Syntaxin 5-dependent retrograde transport to the trans-Golgi network is required for adeno-associated virus transduction.
Chaperonin-containing TCP-1 complex directly binds to the cytoplasmic domain of the LOX-1 receptor.
EGFR-dependent phosphorylation of leucine-rich repeat kinase LRRK1 is important for proper endosomal trafficking of EGFR.
Endo-lysosomal vesicles positive for Rab7 and LAMP1 are terminal vesicles for the transport of dextran.
Intracellular degradation of low-density lipoprotein probed with two-color fluorescence microscopy.
A spatio-temporal analysis of matrix protein and nucleocapsid trafficking during vesicular stomatitis virus uncoating.
Myosin Vc is a molecular motor that functions in secretory granule trafficking.
EHD3 regulates early-endosome-to-Golgi transport and preserves Golgi morphology.
A syntaxin 10-SNARE complex distinguishes two distinct transport routes from endosomes to the trans-Golgi in human cells.
Modulatory role of phosphoinositide 3-kinase in gastric acid secretion.
Identification of compartments involved in mammalian subcellular trafficking pathways by indirect immunofluorescence.
Cholesterol accumulation sequesters Rab9 and disrupts late endosome function in NPC1-deficient cells.
The DnaJ-domain protein RME-8 functions in endosomal trafficking.
Lowe syndrome protein OCRL1 interacts with clathrin and regulates protein trafficking between endosomes and the trans-Golgi network.
A novel GTPase-activating protein for ARF6 directly interacts with clathrin and regulates clathrin-dependent endocytosis.
Defective acidification of intracellular organelles results in aberrant secretion of cathepsin D in cancer cells.
A novel kinesin-like protein, KIF1Bbeta3 is involved in the movement of lysosomes to the cell periphery in non-neuronal cells.
High-pressure freezing of isolated gastric glands provides new insight into the fine structure and subcellular localization of H+/K+-ATPase in gastric parietal cells.
High-pressure freezing of isolated gastric glands provides new insight into the fine structure and subcellular localization of H+/K+-ATPase in gastric parietal cells.
Rab9 GTPase regulates late endosome size and requires effector interaction for its stability.
Intracellular processing and activation of membrane type 1 matrix metalloprotease depends on its partitioning into lipid domains.
Distribution and trafficking of MPR300 is normal in cells with cholesterol accumulated in late endocytic compartments: evidence for early endosome-to-TGN trafficking of MPR300.
Mechanism of constitutive export from the golgi: bulk flow via the formation, protrusion, and en bloc cleavage of large trans-golgi network tubular domains.
A role for GRIP domain proteins and/or their ligands in structure and function of the trans Golgi network.
Accumulation of tyrosinase in the endolysosomal compartment is induced by U18666A.
Overexpression of an ADP-ribosylation factor-guanine nucleotide exchange factor, BIG2, uncouples brefeldin A-induced adaptor protein-1 coat dissociation and membrane tubulation.
Human myosin-Vc is a novel class V myosin expressed in epithelial cells.
Sorting of mannose 6-phosphate receptors mediated by the GGAs.
Functionally distinct pools of actin in secretory cells.
Functionally distinct pools of actin in secretory cells.
Expression of rab11a N124I in gastric parietal cells inhibits stimulatory recruitment of the H+-K+-ATPase.
Expression of rab11a N124I in gastric parietal cells inhibits stimulatory recruitment of the H+-K+-ATPase.
Receptor extracellular domains may contain trafficking information. Studies of the 300-kDa mannose 6-phosphate receptor.
Characterization of the beta-subunit of the H(+)-K(+)-ATPase using an inhibitory monoclonal antibody.
Characterization of the beta-subunit of the H(+)-K(+)-ATPase using an inhibitory monoclonal antibody.
Serum form of the rat insulin-like growth factor II/mannose 6-phosphate receptor is truncated in the carboxyl-terminal domain.
Striepen JF, Voeltz GK
The Journal of cell biology 2022 Aug 1;221(8)
The Journal of cell biology 2022 Aug 1;221(8)
Fate of the Fc fusion protein aflibercept in retinal endothelial cells: competition of recycling and degradation.
Deissler HL, Lang GK, Lang GE
Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie 2019 Jan;257(1):83-94
Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie 2019 Jan;257(1):83-94
A Novel Class of ER Membrane Proteins Regulates ER-Associated Endosome Fission.
Hoyer MJ, Chitwood PJ, Ebmeier CC, Striepen JF, Qi RZ, Old WM, Voeltz GK
Cell 2018 Sep 20;175(1):254-265.e14
Cell 2018 Sep 20;175(1):254-265.e14
Amino acids stimulate the endosome-to-Golgi trafficking through Ragulator and small GTPase Arl5.
Shi M, Chen B, Mahajan D, Boh BK, Zhou Y, Dutta B, Tie HC, Sze SK, Wu G, Lu L
Nature communications 2018 Nov 26;9(1):4987
Nature communications 2018 Nov 26;9(1):4987
Caspase-mediated proteolysis of the sorting nexin 2 disrupts retromer assembly and potentiates Met/hepatocyte growth factor receptor signaling.
Duclos CM, Champagne A, Carrier JC, Saucier C, Lavoie CL, Denault JB
Cell death discovery 2017;3:16100
Cell death discovery 2017;3:16100
Sequence-dependent cargo recognition by SNX-BARs mediates retromer-independent transport of CI-MPR.
Simonetti B, Danson CM, Heesom KJ, Cullen PJ
The Journal of cell biology 2017 Nov 6;216(11):3695-3712
The Journal of cell biology 2017 Nov 6;216(11):3695-3712
A Trematode Parasite Derived Growth Factor Binds and Exerts Influences on Host Immune Functions via Host Cytokine Receptor Complexes.
Sulaiman AA, Zolnierczyk K, Japa O, Owen JP, Maddison BC, Emes RD, Hodgkinson JE, Gough KC, Flynn RJ
PLoS pathogens 2016 Nov;12(11):e1005991
PLoS pathogens 2016 Nov;12(11):e1005991
Excess sphingomyelin disturbs ATG9A trafficking and autophagosome closure.
Corcelle-Termeau E, Vindeløv SD, Hämälistö S, Mograbi B, Keldsbo A, Bräsen JH, Favaro E, Adam D, Szyniarowski P, Hofman P, Krautwald S, Farkas T, Petersen NH, Rohde M, Linkermann A, Jäättelä M
Autophagy 2016 May 3;12(5):833-49
Autophagy 2016 May 3;12(5):833-49
Human Copper Chaperone Atox1 Translocates to the Nucleus but does not Bind DNA In Vitro.
Kahra D, Mondol T, Niemiec MS, Wittung-Stafshede P
Protein and peptide letters 2015;22(6):532-8
Protein and peptide letters 2015;22(6):532-8
Human Copper Chaperone Atox1 Translocates to the Nucleus but does not Bind DNA In Vitro.
Kahra D, Mondol T, Niemiec MS, Wittung-Stafshede P
Protein and peptide letters 2015;22(6):532-8
Protein and peptide letters 2015;22(6):532-8
Syntaxin 5-dependent retrograde transport to the trans-Golgi network is required for adeno-associated virus transduction.
Nonnenmacher ME, Cintrat JC, Gillet D, Weber T
Journal of virology 2015 Feb;89(3):1673-87
Journal of virology 2015 Feb;89(3):1673-87
Chaperonin-containing TCP-1 complex directly binds to the cytoplasmic domain of the LOX-1 receptor.
Bakthavatsalam D, Soung RH, Tweardy DJ, Chiu W, Dixon RA, Woodside DG
FEBS letters 2014 Jun 13;588(13):2133-40
FEBS letters 2014 Jun 13;588(13):2133-40
EGFR-dependent phosphorylation of leucine-rich repeat kinase LRRK1 is important for proper endosomal trafficking of EGFR.
Ishikawa K, Nara A, Matsumoto K, Hanafusa H
Molecular biology of the cell 2012 Apr;23(7):1294-306
Molecular biology of the cell 2012 Apr;23(7):1294-306
Endo-lysosomal vesicles positive for Rab7 and LAMP1 are terminal vesicles for the transport of dextran.
Humphries WH 4th, Szymanski CJ, Payne CK
PloS one 2011;6(10):e26626
PloS one 2011;6(10):e26626
Intracellular degradation of low-density lipoprotein probed with two-color fluorescence microscopy.
Humphries WH 4th, Fay NC, Payne CK
Integrative biology : quantitative biosciences from nano to macro 2010 Oct;2(10):536-44
Integrative biology : quantitative biosciences from nano to macro 2010 Oct;2(10):536-44
A spatio-temporal analysis of matrix protein and nucleocapsid trafficking during vesicular stomatitis virus uncoating.
Mire CE, White JM, Whitt MA
PLoS pathogens 2010 Jul 15;6(7):e1000994
PLoS pathogens 2010 Jul 15;6(7):e1000994
Myosin Vc is a molecular motor that functions in secretory granule trafficking.
Jacobs DT, Weigert R, Grode KD, Donaldson JG, Cheney RE
Molecular biology of the cell 2009 Nov;20(21):4471-88
Molecular biology of the cell 2009 Nov;20(21):4471-88
EHD3 regulates early-endosome-to-Golgi transport and preserves Golgi morphology.
Naslavsky N, McKenzie J, Altan-Bonnet N, Sheff D, Caplan S
Journal of cell science 2009 Feb 1;122(Pt 3):389-400
Journal of cell science 2009 Feb 1;122(Pt 3):389-400
A syntaxin 10-SNARE complex distinguishes two distinct transport routes from endosomes to the trans-Golgi in human cells.
Ganley IG, Espinosa E, Pfeffer SR
The Journal of cell biology 2008 Jan 14;180(1):159-72
The Journal of cell biology 2008 Jan 14;180(1):159-72
Modulatory role of phosphoinositide 3-kinase in gastric acid secretion.
Mettler SE, Ghayouri S, Christensen GP, Forte JG
American journal of physiology. Gastrointestinal and liver physiology 2007 Sep;293(3):G532-43
American journal of physiology. Gastrointestinal and liver physiology 2007 Sep;293(3):G532-43
Identification of compartments involved in mammalian subcellular trafficking pathways by indirect immunofluorescence.
Doody A, Putnam D
Methods in molecular medicine 2006;127:127-36
Methods in molecular medicine 2006;127:127-36
Cholesterol accumulation sequesters Rab9 and disrupts late endosome function in NPC1-deficient cells.
Ganley IG, Pfeffer SR
The Journal of biological chemistry 2006 Jun 30;281(26):17890-9
The Journal of biological chemistry 2006 Jun 30;281(26):17890-9
The DnaJ-domain protein RME-8 functions in endosomal trafficking.
Girard M, Poupon V, Blondeau F, McPherson PS
The Journal of biological chemistry 2005 Dec 2;280(48):40135-43
The Journal of biological chemistry 2005 Dec 2;280(48):40135-43
Lowe syndrome protein OCRL1 interacts with clathrin and regulates protein trafficking between endosomes and the trans-Golgi network.
Choudhury R, Diao A, Zhang F, Eisenberg E, Saint-Pol A, Williams C, Konstantakopoulos A, Lucocq J, Johannes L, Rabouille C, Greene LE, Lowe M
Molecular biology of the cell 2005 Aug;16(8):3467-79
Molecular biology of the cell 2005 Aug;16(8):3467-79
A novel GTPase-activating protein for ARF6 directly interacts with clathrin and regulates clathrin-dependent endocytosis.
Tanabe K, Torii T, Natsume W, Braesch-Andersen S, Watanabe T, Satake M
Molecular biology of the cell 2005 Apr;16(4):1617-28
Molecular biology of the cell 2005 Apr;16(4):1617-28
Defective acidification of intracellular organelles results in aberrant secretion of cathepsin D in cancer cells.
Kokkonen N, Rivinoja A, Kauppila A, Suokas M, Kellokumpu I, Kellokumpu S
The Journal of biological chemistry 2004 Sep 17;279(38):39982-8
The Journal of biological chemistry 2004 Sep 17;279(38):39982-8
A novel kinesin-like protein, KIF1Bbeta3 is involved in the movement of lysosomes to the cell periphery in non-neuronal cells.
Matsushita M, Tanaka S, Nakamura N, Inoue H, Kanazawa H
Traffic (Copenhagen, Denmark) 2004 Mar;5(3):140-51
Traffic (Copenhagen, Denmark) 2004 Mar;5(3):140-51
High-pressure freezing of isolated gastric glands provides new insight into the fine structure and subcellular localization of H+/K+-ATPase in gastric parietal cells.
Sawaguchi A, McDonald KL, Forte JG
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2004 Jan;52(1):77-86
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2004 Jan;52(1):77-86
High-pressure freezing of isolated gastric glands provides new insight into the fine structure and subcellular localization of H+/K+-ATPase in gastric parietal cells.
Sawaguchi A, McDonald KL, Forte JG
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2004 Jan;52(1):77-86
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2004 Jan;52(1):77-86
Rab9 GTPase regulates late endosome size and requires effector interaction for its stability.
Ganley IG, Carroll K, Bittova L, Pfeffer S
Molecular biology of the cell 2004 Dec;15(12):5420-30
Molecular biology of the cell 2004 Dec;15(12):5420-30
Intracellular processing and activation of membrane type 1 matrix metalloprotease depends on its partitioning into lipid domains.
Mazzone M, Baldassarre M, Beznoussenko G, Giacchetti G, Cao J, Zucker S, Luini A, Buccione R
Journal of cell science 2004 Dec 15;117(Pt 26):6275-87
Journal of cell science 2004 Dec 15;117(Pt 26):6275-87
Distribution and trafficking of MPR300 is normal in cells with cholesterol accumulated in late endocytic compartments: evidence for early endosome-to-TGN trafficking of MPR300.
Umeda A, Fujita H, Kuronita T, Hirosako K, Himeno M, Tanaka Y
Journal of lipid research 2003 Oct;44(10):1821-32
Journal of lipid research 2003 Oct;44(10):1821-32
Mechanism of constitutive export from the golgi: bulk flow via the formation, protrusion, and en bloc cleavage of large trans-golgi network tubular domains.
Polishchuk EV, Di Pentima A, Luini A, Polishchuk RS
Molecular biology of the cell 2003 Nov;14(11):4470-85
Molecular biology of the cell 2003 Nov;14(11):4470-85
A role for GRIP domain proteins and/or their ligands in structure and function of the trans Golgi network.
Yoshino A, Bieler BM, Harper DC, Cowan DA, Sutterwala S, Gay DM, Cole NB, McCaffery JM, Marks MS
Journal of cell science 2003 Nov 1;116(Pt 21):4441-54
Journal of cell science 2003 Nov 1;116(Pt 21):4441-54
Accumulation of tyrosinase in the endolysosomal compartment is induced by U18666A.
Hall AM, Krishnamoorthy L, Orlow SJ
Pigment cell research 2003 Apr;16(2):149-58
Pigment cell research 2003 Apr;16(2):149-58
Overexpression of an ADP-ribosylation factor-guanine nucleotide exchange factor, BIG2, uncouples brefeldin A-induced adaptor protein-1 coat dissociation and membrane tubulation.
Shinotsuka C, Yoshida Y, Kawamoto K, Takatsu H, Nakayama K
The Journal of biological chemistry 2002 Mar 15;277(11):9468-73
The Journal of biological chemistry 2002 Mar 15;277(11):9468-73
Human myosin-Vc is a novel class V myosin expressed in epithelial cells.
Rodriguez OC, Cheney RE
Journal of cell science 2002 Mar 1;115(Pt 5):991-1004
Journal of cell science 2002 Mar 1;115(Pt 5):991-1004
Sorting of mannose 6-phosphate receptors mediated by the GGAs.
Puertollano R, Aguilar RC, Gorshkova I, Crouch RJ, Bonifacino JS
Science (New York, N.Y.) 2001 Jun 1;292(5522):1712-6
Science (New York, N.Y.) 2001 Jun 1;292(5522):1712-6
Functionally distinct pools of actin in secretory cells.
Ammar DA, Nguyen PN, Forte JG
American journal of physiology. Cell physiology 2001 Aug;281(2):C407-17
American journal of physiology. Cell physiology 2001 Aug;281(2):C407-17
Functionally distinct pools of actin in secretory cells.
Ammar DA, Nguyen PN, Forte JG
American journal of physiology. Cell physiology 2001 Aug;281(2):C407-17
American journal of physiology. Cell physiology 2001 Aug;281(2):C407-17
Expression of rab11a N124I in gastric parietal cells inhibits stimulatory recruitment of the H+-K+-ATPase.
Duman JG, Tyagarajan K, Kolsi MS, Moore HP, Forte JG
The American journal of physiology 1999 Sep;277(3):C361-72
The American journal of physiology 1999 Sep;277(3):C361-72
Expression of rab11a N124I in gastric parietal cells inhibits stimulatory recruitment of the H+-K+-ATPase.
Duman JG, Tyagarajan K, Kolsi MS, Moore HP, Forte JG
The American journal of physiology 1999 Sep;277(3 Pt 1):C361-72
The American journal of physiology 1999 Sep;277(3 Pt 1):C361-72
Receptor extracellular domains may contain trafficking information. Studies of the 300-kDa mannose 6-phosphate receptor.
Dintzis SM, Velculescu VE, Pfeffer SR
The Journal of biological chemistry 1994 Apr 22;269(16):12159-66
The Journal of biological chemistry 1994 Apr 22;269(16):12159-66
Characterization of the beta-subunit of the H(+)-K(+)-ATPase using an inhibitory monoclonal antibody.
Chow DC, Forte JG
The American journal of physiology 1993 Dec;265(6 Pt 1):C1562-70
The American journal of physiology 1993 Dec;265(6 Pt 1):C1562-70
Characterization of the beta-subunit of the H(+)-K(+)-ATPase using an inhibitory monoclonal antibody.
Chow DC, Forte JG
The American journal of physiology 1993 Dec;265(6 Pt 1):C1562-70
The American journal of physiology 1993 Dec;265(6 Pt 1):C1562-70
Serum form of the rat insulin-like growth factor II/mannose 6-phosphate receptor is truncated in the carboxyl-terminal domain.
MacDonald RG, Tepper MA, Clairmont KB, Perregaux SB, Czech MP
The Journal of biological chemistry 1989 Feb 25;264(6):3256-61
The Journal of biological chemistry 1989 Feb 25;264(6):3256-61
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
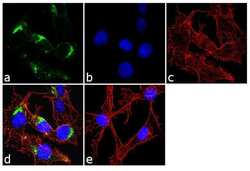
- Immunofluorescence analysis of Mannose 6 Phosphate Receptor/CD222 was done on 70% confluent log phase HepG2 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Mannose 6 Phosphate Receptor/CD222 (2G11) Mouse Monoclonal Antibody (Product # MA1-066) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d is a merged image showing cytoplasmic localization. Panel e is a no primary antibody control. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
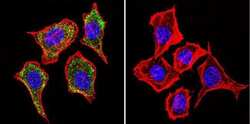
- Immunofluorescent analysis of Mannose 6-Phosphate Receptor using Anti-Mannose 6-Phosphate Receptor Monoclonal Antibody (2G11) (Product # MA1-066) shows staining in Hela Cells. Mannose 6-Phosphate Receptor staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing Mannose 6-Phosphate Receptor (Product # MA1-066) at a dilution of 1:20 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35503, Goat Anti-Mouse). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
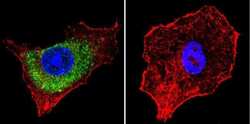
- Immunofluorescent analysis of Mannose 6-Phosphate Receptor using Anti-Mannose 6-Phosphate Receptor Monoclonal Antibody (2G11) (Product # MA1-066) shows staining in HepG2 Cells. Mannose 6-Phosphate Receptor staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing Mannose 6-Phosphate Receptor (Product # MA1-066) at a dilution of 1:20 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35503, Goat Anti-Mouse). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
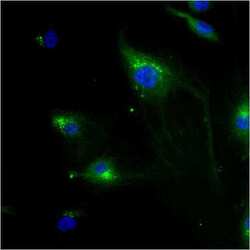
- Immunofluorescent analysis of Mannose 6-Phosphate Receptor using anti-Mannose 6-Phosphate Receptor monoclonal antibody (Product # MA1-066) shows staining in HMVEC Cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
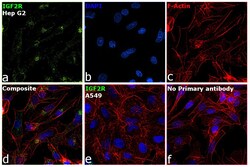
- Experimental details
- Immunofluorescence analysis of IGF2R was performed using 70% confluent log phase Hep G2 cells. The cells were fixed with 4% paraformaldehyde for 5 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with IGF2R Monoclonal Antibody (2G11) (Product # MA1-066) at 5 µg/mL concentration in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766) (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b: Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing Golgi and Cytoplasmic localization. Panel e represents low expressing cell line A549. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
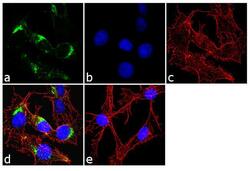
- Experimental details
- Immunofluorescence analysis of Mannose 6 Phosphate Receptor/CD222 was done on 70% confluent log phase HepG2 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Mannose 6 Phosphate Receptor/CD222 (2G11) Mouse Monoclonal Antibody (Product # MA1-066) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A28175) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d is a merged image showing cytoplasmic localization. Panel e is a no primary antibody control. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
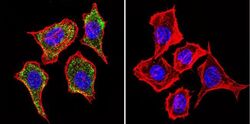
- Experimental details
- Immunofluorescent analysis of Mannose 6-Phosphate Receptor using Anti-Mannose 6-Phosphate Receptor Monoclonal Antibody (2G11) (Product # MA1-066) shows staining in Hela Cells. Mannose 6-Phosphate Receptor staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing Mannose 6-Phosphate Receptor (Product # MA1-066) at a dilution of 1:20 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35503, Goat Anti-Mouse). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
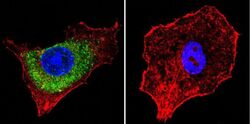
- Experimental details
- Immunofluorescent analysis of Mannose 6-Phosphate Receptor using Anti-Mannose 6-Phosphate Receptor Monoclonal Antibody (2G11) (Product # MA1-066) shows staining in HepG2 Cells. Mannose 6-Phosphate Receptor staining (green), F-Actin staining with Phalloidin (red) and nuclei with DAPI (blue) is shown. Cells were grown on chamber slides and fixed with formaldehyde prior to staining. Cells were probed without (control) or with or an antibody recognizing Mannose 6-Phosphate Receptor (Product # MA1-066) at a dilution of 1:20 over night at 4°C, washed with PBS and incubated with a DyLight-488 conjugated secondary antibody (Product # 35503, Goat Anti-Mouse). Images were taken at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
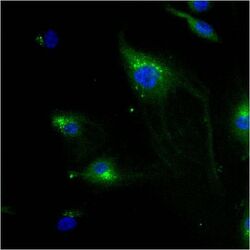
- Experimental details
- Immunofluorescent analysis of Mannose 6-Phosphate Receptor using anti-Mannose 6-Phosphate Receptor monoclonal antibody (Product # MA1-066) shows staining in HMVEC Cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
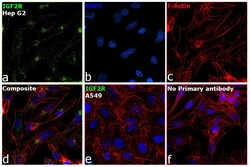
- Experimental details
- Immunofluorescence analysis of IGF2R was performed using 70% confluent log phase Hep G2 cells. The cells were fixed with 4% paraformaldehyde for 5 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with IGF2R Monoclonal Antibody (2G11) (Product # MA1-066) at 5 µg/mL concentration in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766) (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b: Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing Golgi and Cytoplasmic localization. Panel e represents low expressing cell line A549. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
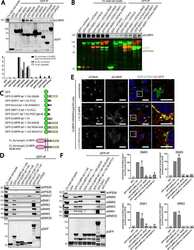
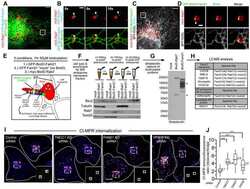
- Figure 3. CI-MPR segregates in SNX1/2-SNX5/6 tubular profiles which are not decorated with retromer. (A) Endogenous CI-MPR localizes to a highly packed vesicular cluster that colocalized with the TGN and partially colocalized with retromer-positive endosomes. HeLa cells were fixed and immunostained for endogenous TGN46 (TGN marker), VPS35 (retromer), SNX1, and SNX2 (both retromer-linked SNX-BARs). Bars: (main images) 20 um; (zooms) 5 um. (B, top) CI-MPR colocalizes with SNX2-positive subdomains on enlarged endosomes. HeLa cells were transfected with BFP-Rab5Q79L and immunostained for endogenous CI-MPR, SNX2, and VPS35 after 48 h. The dashed line in the top left image indicates the contour of the nucleus, and the dashed line on the top right refers to the enlarged endosome from which the intensity line scan was measured. Bars: (main images) 10 um; (zooms) 5 um. (B, bottom) Line scan of signal intensity across the circumference of enlarged endosome. (B, right) Distribution of CI-MPR in retromer subdomains; n = 3 independent experiments. CI-MPR signal was quantified in 36 enlarged endosomes (means +- SEM). (C) Overexpression of a WT GFP-CI-MPR chimera leads to the formation of extended CI-MPR tubules, some of which are positive for endogenous SNX1. HeLa cells were transfected with GFP-CI-MPR chimera WT and immunostained for SNX1 after 48 h. Bars: (main images) 20 um; (zooms) 10 um. (D) A subpopulation of GFP-CI-MPR chimera tubules is decorated with endogenous SNX1 but not
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
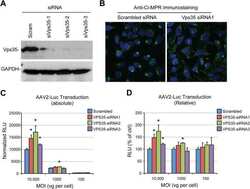
- Figure 7. Gene editing confirms the essential role of SNX1/2-SNX5/6 in endosome-to-TGN recycling of CI-MPR. (A, left) CI-MPR levels and CI-MPR steady-state localization in retromer and SNX1/2-SNX5/6 KO cells. HeLa cells were transfected with CRISPR-Cas9 plasmids against SNX1 and SNX2, SNX5 and SNX6, or VPS35. 96 h after transfection, endogenous protein levels were analyzed by Western blotting, and cells were fixed and stained for endogenous CI-MPR. n = 3 independent experiments (one-way ANOVA compared with parental HeLa). Molecular masses are given in kilodaltons. (A, middle) Representative images. Bars, 20 um. (A, right) quantification of the percentage of cells displaying each phenotype. n = 3 blindly scored independent experiments; parental, 116 cells; VPS35, 106 cells; SNX5 + SNX6, 111 cells; VPS35, 106 cells. (B) Immunofluorescence of endogenous CI-MPR and endogenous SNX1 and VPS35 in retromer KO cells and SNX1 and SNX2 KO cells. Bars: (top) 20 um; (bottom) 5 um. (C) Immunofluorescence and colocalization analysis of endogenous CI-MPR and TGN marker TGN46 and early endosomal marker EEA1 in SNX1 and SNX2, SNX5 and SNX6, and VPS35 CRISPR-Cas9 KO HeLa cells. Bars: (main images) 20 um; (insets) 10 um. (C, top right) n = 3 independent experiments; parental HeLa, 80 cells; SNX1+2 KO, 99 cells; SNX5+6, 81 cells; VPS35 KO, 76 cells. (C, bottom right) n = 4 independent experiments; parental HeLa, 102 cells, SNX1+2 KO, 114 cells; SNX5+6, 104 cells; VPS35 KO, 102 cells (means +- SEM
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
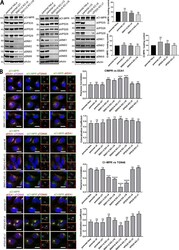
- Figure 8. Clonal HeLa KO cell lines recapitulate the SNX1/2-SNX5/6 mediated retromer-independent retrograde transport of CI-MPR. (A) Clonal cell lines were isolated from a heterogeneous population of CRISPR-Cas9 KO. Two independent lines were biochemically characterized as SNX1+2 KOs, two as SNX5+6 KOs, two as VPS35 KOs, and three as parental clonal lines. Molecular masses are given in kilodaltons. (A, right) CI-MPR levels were analyzed by Western blotting. n = 3 independent experiments. (B, left) Immunofluorescence and colocalization analysis of endogenous CI-MPR, TGN marker TGN46, and early endosomal marker EEA1 in parental HeLa and clonally selected KO lines. Bars, 20 um. (B, top right two graphs) n = 3 independent experiments; parental HeLa, 58 cells; parental c8, 79 cells; parental c21, 81 cells; SNX1+2 KO c4, 75 cells; SNX1+2 KO c16, 70 cells; SNX5+6 KO c13, 82 cells; SNX5+6 KO c18, 89 cells; VPS35 KO c5, 83 cells; VPS35 KO c7, 72 cells. (B, bottom right two graphs) n = 3 independent experiments; parental HeLa, 67 cells; parental c8, 72 cells; parental c21, 87 cells; SNX1+2 KO c4, 83 cells; SNX1+2 KO c16, 80 cells; SNX5+6 KO c13, 84 cells; SNX5+6 KO c18, 98 cells; VPS35 KO c5, 73 cells; VPS35 KO c7, 68 cells (means +- SEM; one-way ANOVA compared with parental HeLa. *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, P < 0.001).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
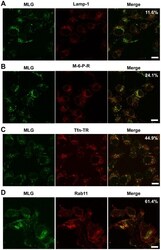
- Figure 6 Colocalization of MLG with markers for recycling endosomes. A synchronized fusion assay was performed as described for Fig. 4B , the cells were fixed at t-60, stained for (A) LAMP-1, (B) mannose-6-phosphate receptor, (C) Tfn-TR, or (D) Rab11 and then examined by LSCM (Bars = 10 um). Percent colocalization of MLG with the indicated marker from approximately 20 individual cells from two independent experiments is shown in the upper right-hand corner of the merged images.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
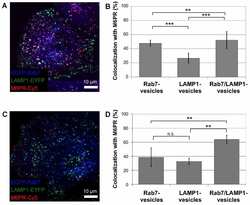
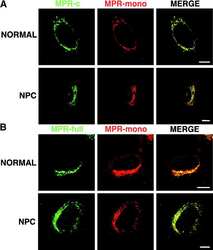
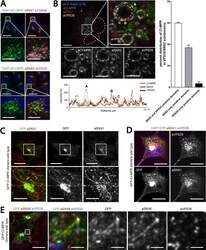
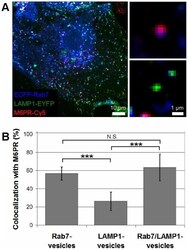
- Figure 3 Colocalization of M6PR with Rab7-, LAMP1-, and Rab7/LAMP1-vesicles. ( A ) Confocal microscopy image of ECFP-Rab7 (blue), LAMP1-EYFP (green), and an antibody against M6PR labeled with a Cy5-labeled secondary antibody (red). Smaller images show a M6PR-positive (red) Rab7-vesicle (blue, top) and a M6PR-negative LAMP1-vesicle (green, bottom). ( B ) A large fraction of Rab7- and Rab7/LAMP1-vesicles are positive for M6PR; 57+-7% and 63+-15%, respectively. A smaller fraction of LAMP1-vesicles are positive for M6PR, 23+-10%. Error bars show standard deviations. P-values0.05. The graph shows the analysis of 10-20 of each type of vesicle per cell in 10 cells from 4 distinct experiments. Similar results were obtained for BS-C-1 cells stably expressing ECFP-Rab7 and for HeLa cells ( Figure S4 ). Unmerged images are shown in Figure S5 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
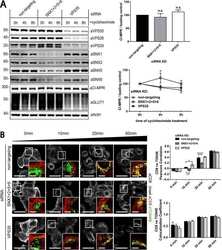
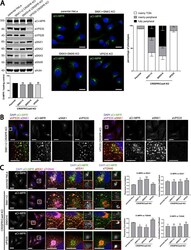
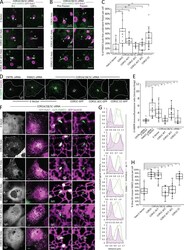
- Figure 5. The COR1C CC limits bud actin to facilitate ER contact, endosome fission, and CI-M6PR sorting. (A) Representative images of LE buds stable for duration of acquisition in conditions that did not rescued fission rate (C). COS-7 cells were cotransfected with COR1A/1B/1C siRNAs to deplete all type I coronins and with GFP-Rab7 (LE, magenta), mCh-FAM21 (WASH complex, green), and with either Halo E-vec, siRES COR1C DeltaCC-Halo, or siRES COR1C ACT- DeltaCC-Halo. Arrows indicate bud of interest. (B) Representative images of LE fission events in conditions that rescued fission rate (C). COS-7 cells were cotransfected with COR1A/1B/1C siRNAs to deplete all type I coronins and with GFP-Rab7 (LE, magenta), mCh-FAM21 (WASH complex, green), and with either siRES COR1C-Halo, siRES COR1C ACT--Halo, or siRES COR1C CC-Halo. Arrows indicate bud of interest. (C) Quantification of data in A and B. Graph shows percentage of FAM21-labeled LE buds that underwent fission per cell during a 2-min time lapse. Note that only constructs containing the CC were able to restore fission. Data for graph from Halo E-vec: 139 endosomes in n = 15 cells; siRES COR1C: 122 endosomes in n = 15 cells; siRES COR1C ACT-: 213 endosomes in n = 17 cells; siRES COR1C DeltaCC: 281 endosomes in n = 18 cells; siRES COR1C ACT- DeltaCC: 296 endosomes n = 17 cells; and siRES COR1C CC: 331 endosomes in n = 22 cells, performed in triplicate. (D) Representative images of the M6PR trafficking assay. The relative fluorescenc
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
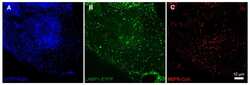
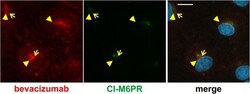
- Fig. 4 Bevacizumab is somewhat co-localized with CI-M6PR. After treatment of iBREC with the humanized IgG bevacizumab for 4 h, cells were immunostained to visualize the IgG (red) or CI-M6PR (green). The prominent CI-M6PR-specific signals close to the nucleus only to a small extent overlapped with those of bevacizumab (yellow arrowheads). Scale bar, 10 mum
 Explore
Explore Validate
Validate Learn
Learn