Antibody data
- Antibody Data
- Antigen structure
- References [14]
- Comments [0]
- Validations
- Other assay [9]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 17-7049-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IL-4 Monoclonal Antibody (8D4-8), APC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 8D4-8 antibody reacts with human interleukin-4 (IL-4), a 15-19 kDa cytokine secreted by Th2 cells. Applications Reported:The 8D4-8 antibody has been reported for use as a capture antibody for human IL-4 ELISA and for intracellular staining for flow cytometric analysis. Applications Tested: Has been pre-titrated and tested in intraceµLlµLar staining and flow cytometric analysis of stimulated human peripheral blood cells. This can be used at 5 µL (0.06 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 633-647 nm; Emission: 660 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 8D4-8
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Cord Blood T Cells Expressing High and Low PKCζ Levels Develop into Cells with a Propensity to Display Th1 and Th9 Cytokine Profiles, Respectively.
Th2 Biased Immunity With Altered B Cell Profiles in Circulation of Patients With Sporotrichosis Caused by Sporothrix globosa.
Platelet-Derived GARP Induces Peripheral Regulatory T Cells-Potential Impact on T Cell Suppression in Patients with Melanoma-Associated Thrombocytosis.
The Phenotype of Monocytes in Anterior Uveitis Depends on the HLA-B27 Status.
Effects of Anti-Integrin Treatment With Vedolizumab on Immune Pathways and Cytokines in Inflammatory Bowel Diseases.
The Genetic Polymorphisms of NLRP3 Inflammasome Associated with T Helper Cells in Patients with Multiple Myeloma.
CD4 T cell loss and Th2 and Th17 bias are associated with the severity of severe fever with thrombocytopenia syndrome (SFTS).
Concomitant suppression of T(H)2 and T(H)17 cell responses in allergic asthma by targeting retinoic acid receptor-related orphan receptor γt.
TIGIT expressing CD4+T cells represent a tumor-supportive T cell subset in chronic lymphocytic leukemia.
Proteinase 3 on apoptotic cells disrupts immune silencing in autoimmune vasculitis.
DCLRE1C (ARTEMIS) mutations causing phenotypes ranging from atypical severe combined immunodeficiency to mere antibody deficiency.
A SnoRNA-derived piRNA interacts with human interleukin-4 pre-mRNA and induces its decay in nuclear exosomes.
CD4+ T cells contain early extrapulmonary tuberculosis (TB) dissemination and rapid TB progression and sustain multieffector functions of CD8+ T and CD3- lymphocytes: mechanisms of CD4+ T cell immunity.
A polymorphism in TIM1 is associated with susceptibility to severe hepatitis A virus infection in humans.
Perveen K, Quach A, McPhee A, Prescott SL, Barry SC, Hii CS, Ferrante A
International journal of molecular sciences 2021 May 5;22(9)
International journal of molecular sciences 2021 May 5;22(9)
Th2 Biased Immunity With Altered B Cell Profiles in Circulation of Patients With Sporotrichosis Caused by Sporothrix globosa.
Zu J, Yao L, Song Y, Cui Y, Guan M, Chen R, Zhen Y, Li S
Frontiers in immunology 2020;11:570888
Frontiers in immunology 2020;11:570888
Platelet-Derived GARP Induces Peripheral Regulatory T Cells-Potential Impact on T Cell Suppression in Patients with Melanoma-Associated Thrombocytosis.
Zimmer N, Krebs FK, Zimmer S, Mitzel-Rink H, Kumm EJ, Jurk K, Grabbe S, Loquai C, Tuettenberg A
Cancers 2020 Dec 5;12(12)
Cancers 2020 Dec 5;12(12)
The Phenotype of Monocytes in Anterior Uveitis Depends on the HLA-B27 Status.
Kasper M, Walscheid K, Laffer B, Bauer D, Busch M, Wildschütz L, Wang B, Loser K, Vogl T, Grajewski RS, Langmann T, Heiligenhaus A
Frontiers in immunology 2018;9:1773
Frontiers in immunology 2018;9:1773
Effects of Anti-Integrin Treatment With Vedolizumab on Immune Pathways and Cytokines in Inflammatory Bowel Diseases.
Rath T, Billmeier U, Ferrazzi F, Vieth M, Ekici A, Neurath MF, Atreya R
Frontiers in immunology 2018;9:1700
Frontiers in immunology 2018;9:1700
The Genetic Polymorphisms of NLRP3 Inflammasome Associated with T Helper Cells in Patients with Multiple Myeloma.
Zhao X, Hua M, Yan S, Yu J, Han F, Zhong C, Wang R, Zhang C, Hou M, Ma D
Journal of immunology research 2018;2018:7569809
Journal of immunology research 2018;2018:7569809
CD4 T cell loss and Th2 and Th17 bias are associated with the severity of severe fever with thrombocytopenia syndrome (SFTS).
Li MM, Zhang WJ, Weng XF, Li MY, Liu J, Xiong Y, Xiong SE, Zou CC, Wang H, Lu MJ, Yang DL, Peng C, Zheng X
Clinical immunology (Orlando, Fla.) 2018 Oct;195:8-17
Clinical immunology (Orlando, Fla.) 2018 Oct;195:8-17
Concomitant suppression of T(H)2 and T(H)17 cell responses in allergic asthma by targeting retinoic acid receptor-related orphan receptor γt.
Na H, Lim H, Choi G, Kim BK, Kim SH, Chang YS, Nurieva R, Dong C, Chang SH, Chung Y
The Journal of allergy and clinical immunology 2018 Jun;141(6):2061-2073.e5
The Journal of allergy and clinical immunology 2018 Jun;141(6):2061-2073.e5
TIGIT expressing CD4+T cells represent a tumor-supportive T cell subset in chronic lymphocytic leukemia.
Catakovic K, Gassner FJ, Ratswohl C, Zaborsky N, Rebhandl S, Schubert M, Steiner M, Gutjahr JC, Pleyer L, Egle A, Hartmann TN, Greil R, Geisberger R
Oncoimmunology 2017;7(1):e1371399
Oncoimmunology 2017;7(1):e1371399
Proteinase 3 on apoptotic cells disrupts immune silencing in autoimmune vasculitis.
Millet A, Martin KR, Bonnefoy F, Saas P, Mocek J, Alkan M, Terrier B, Kerstein A, Tamassia N, Satyanarayanan SK, Ariel A, Ribeil JA, Guillevin L, Cassatella MA, Mueller A, Thieblemont N, Lamprecht P, Mouthon L, Perruche S, Witko-Sarsat V
The Journal of clinical investigation 2015 Nov 2;125(11):4107-21
The Journal of clinical investigation 2015 Nov 2;125(11):4107-21
DCLRE1C (ARTEMIS) mutations causing phenotypes ranging from atypical severe combined immunodeficiency to mere antibody deficiency.
Volk T, Pannicke U, Reisli I, Bulashevska A, Ritter J, Björkman A, Schäffer AA, Fliegauf M, Sayar EH, Salzer U, Fisch P, Pfeifer D, Di Virgilio M, Cao H, Yang F, Zimmermann K, Keles S, Caliskaner Z, Güner SÜ, Schindler D, Hammarström L, Rizzi M, Hummel M, Pan-Hammarström Q, Schwarz K, Grimbacher B
Human molecular genetics 2015 Dec 20;24(25):7361-72
Human molecular genetics 2015 Dec 20;24(25):7361-72
A SnoRNA-derived piRNA interacts with human interleukin-4 pre-mRNA and induces its decay in nuclear exosomes.
Zhong F, Zhou N, Wu K, Guo Y, Tan W, Zhang H, Zhang X, Geng G, Pan T, Luo H, Zhang Y, Xu Z, Liu J, Liu B, Gao W, Liu C, Ren L, Li J, Zhou J, Zhang H
Nucleic acids research 2015 Dec 2;43(21):10474-91
Nucleic acids research 2015 Dec 2;43(21):10474-91
CD4+ T cells contain early extrapulmonary tuberculosis (TB) dissemination and rapid TB progression and sustain multieffector functions of CD8+ T and CD3- lymphocytes: mechanisms of CD4+ T cell immunity.
Yao S, Huang D, Chen CY, Halliday L, Wang RC, Chen ZW
Journal of immunology (Baltimore, Md. : 1950) 2014 Mar 1;192(5):2120-32
Journal of immunology (Baltimore, Md. : 1950) 2014 Mar 1;192(5):2120-32
A polymorphism in TIM1 is associated with susceptibility to severe hepatitis A virus infection in humans.
Kim HY, Eyheramonho MB, Pichavant M, Gonzalez Cambaceres C, Matangkasombut P, Cervio G, Kuperman S, Moreiro R, Konduru K, Manangeeswaran M, Freeman GJ, Kaplan GG, DeKruyff RH, Umetsu DT, Rosenzweig SD
The Journal of clinical investigation 2011 Mar;121(3):1111-8
The Journal of clinical investigation 2011 Mar;121(3):1111-8
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
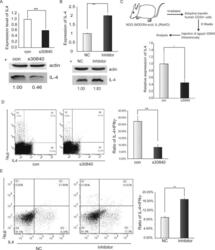
- Figure 6. piR30840 regulates the development of Th2 lymphocytes. ( A and B ) Human CD4 naive cells were cultured in a conditioned medium for Th2 development and then transfected with an s30840 (A) or piR30840 inhibitor (B). IL-4 expression was detected by qRT-PCR and western blotting analyses. P < 0.01. The data represents three independent experiments. ( C ) Detection of IL-4 expression in human PBMCs isolated from humanized NOG mice. IL-4 expression was detected by qRT-PCR analysis. The data was expressed as the mean of three independent experiments. Wilcoxon test was used to calculate the P -value. P < 0.05. ( D ) CD4 naive cells were cultured in the conditioned medium for Th2 development and transfected with s30840 or control. After 7 days, the Th2 T-lymphocytes (intracellular stained as IL-4 + IFN-gamma - ) were detected with FACS analysis. The data was shown as a mean of six independent experiments. P < 0.01. ( E ) The CD4 naive cells were cultured in a conditioned medium for Th2 development with or without the transfection of inhibitor. We adoptively transferred the naive cells into the irradiated NOG mice which were first humanized. After 7 days, Th2 T-lymphocytes (intracellularly stained as IL-4 + IFNgamma - ) were detected by using FACS analysis. The data were expressed as the mean of six independent experiments. Statistical significance between two samples was determined by using the student's t -test. P < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Blockade of transforming growth factor (TGF)-beta I-III did in part prevent regulatory T cells (Treg) induction. ( A ) CFSE-labeled CD4 + CD25 - T cells were cocultured with platelets in the ratio of 1:50 and were stimulated with anti-CD3 mAb (0.5 ug/mL) and anti-CD28 mAb (1.0 ug/mL) in the presence of either anti-TGF-beta I-III (10 ug/mL) or anti-TGF-beta receptor II (10 ug/mL) antibodies. Antibodies were added at day 0. The expression of Foxp3 and GARP and cell proliferation were determined on day 3 via flow cytometry. ( B ) Production of IL-2 and IFN-gamma was assessed by intracellular flow cytometry on day 6. The graphs show cells cultured in the presence of platelets normalized to CD4 + CD25 - T cells without platelets. Dot plots show one representative result of 10 independent experiments ( n = 10, box and whiskers, medians +- min/max, * p < 0.05, ** p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Combining blockade of TGF-beta signaling and GARP led to a complete inhibition of platelet effects. ( A ) CFSE-labeled CD4 + CD25 - T cells were cocultured with platelets in the ratio of 1:50 and were stimulated with anti-CD3 mAb (0.5 ug/mL) and anti-CD28 mAb (1.0 ug/mL). CD4 + CD25 - T cells were incubated for 15 min with TGF-beta receptor II (10 ug/mL) antibody prior to coculture, as indicated. Excess antibody was removed. Pre-treated CD4 + CD25 - T cells were cultured in the presence of either anti-TGF-beta I-III (10 ug/mL) and/or anti-GARP Ab (10 ug/mL) antibodies. Antibodies were added at day 0. The expression of Foxp3, GARP and cell proliferation were determined on day 3 via flow cytometry. ( B ) Production of IL-2 and IFN-gamma was assessed by intracellular flow cytometry on day 6. The graphs show cells cultured in the presence of platelets normalized to CD4 + CD25 - T cells without platelets. Dot plots show 1 representative result of 10 independent experiments ( n = 3, means +- SD, * p < 0.05, ** p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
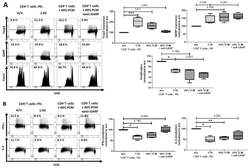
- Figure 7 Platelet-conditioned medium (PCM) inhibited IFN-gamma production, but failed to induce a Treg phenotype. ( A ) CD4 + CD25 - T cells were cultured in X-Vivo 15 (Lonza, Basel, Switzerland) with 60% PCM content, with or without 10 ug/mL anti-GARP Ab and stimulated with 0.5 ug/mL anti-CD3 mAb and 1.0 ug/mL anti-CD28 mAb. Antibodies were added at day 0. The expression of Foxp3, GARP and cell proliferation were determined at day 3 with flow cytometry. ( B ) Cytokine production of IL-2 and IFN-gamma was measured by intracellular flow cytometry on day 6. Dot plots show one representative result of five independent experiments ( n = 5, box and whiskers, medians +- min/max, * p < 0.05, ** p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure A4 Thrombin-activated platelets induced a regulatory phenotype in CD4 + CD25 - T cells. 1 x 10 6 CD4 + CD25 - T cells were stimulated with 0.5 ug/mL anti-CD3 mAb and 1.0 ug/mL anti-CD28 mAb with or without 50 x 10 6 platelets for 6 days and treated with or without 10 U/mL thrombin. ( A ) Foxp3 and GARP expression and proliferation were determined at day 3 via flow cytometry. ( B ) Using intracellular flow cytometry, we analyzed cytokine production of IL-2 and IFN-gamma on day 6. Dot plots show one representative result of five independent experiments ( n = 5, box and whiskers, medians +- min/max, * p < 0.05, ** p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure A5 TRAP-6-activated platelets induced a regulatory phenotype in CD4 + CD25 - T cells. 1 x 10 6 CD4 + CD25 - T cells were stimulated with 0.5 ug/mL anti-CD3 mAb and 1.0 ug/mL anti-CD28 mAb with or without 50 x 10 6 platelets for 6 days and treated with or without 5 uM TRAP-6. ( A ) Foxp3 and GARP expression and proliferation were determined at day 3 via flow cytometry. ( B ) Using intracellular flow cytometry, we analyzed cytokine production of IL-2 and IFN-gamma on day 6. Dot plots show one representative result of five independent experiments ( n = 5, box and whiskers, medians +- min/max, * p < 0.05, ** p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
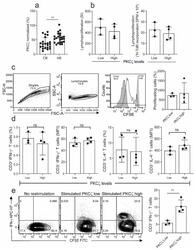
- Figure 1 Comparison of proliferation and cytokine production in naive cord blood T cells (CBTC) in Protein Kinase C zeta (PKCzeta) low or high group. ( a ) Comparison of PKCzeta between cord blood (CB) and adult blood (AB) T cells, n = 24 for each AB or CB. ( b ) Shows lymphoproliferation as a Stimulation index (SI) and disintegrations per minute (DPM) in 3 H-thymidine pulsed cultures stimulated with Phytohaemagglutinin (PHA) and Phorbol myristate acetate (PMA) ( c ) Purified CBCTs were stained with Carboxyfluorescein succinimidyl ester (CFSE) dye and stimulated with immobilized anti-CD3/-CD28 antibodies for 3 days. Gating and representative histogram for CFSE dilution after exclusion of doublets and dead cells. Overlaid histograms for stained unstimulated and unstained stimulated samples were used as control and for gating the non-proliferating cells and for auto-fluorescence, respectively. ( d ) Naive CB CD3 + T cells were stimulated with PHA/PMA (18 h) and percentage of CD3 + T cells producing interleukin-4 (IL-4) and Interferon-gamma (IFN-gamma) and median fluorescent intensity (MFI) were examined by flow cytometry assays. ( e ) On day 5 of CFSE stained culture (anti-CD3/-CD28), cells were re-stimulated with PHA/PMA (18 h) for detection of intracellular cytokine. Representative flow dot plots and data for CFSE dye dilution and IFN-gamma producing cells in high and low PKCzeta group. Data mean +- SD of n = 3 for of low and n = 4 high PKCzeta group. ** p < 0.01. ns: not sig
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
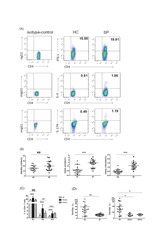
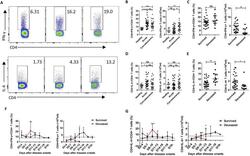
- Fig. 3 Changes in the percentages and absolute numbers of Th1 and Th2 cells in SFTS patients. (A): Th1 cells (CD4 + IFN-gamma+) and Th2 cells (CD4 + IL-4+) by flow cytometry in the healthy controls, surviving SFTS patients, and deceased SFTS patients, as defined by flow cytometry. The cells were gated on the CD3 + CD4+ population within the single-cell lymphocyte gate. (B), (D): The percentages and numbers of Th1 and Th2 cells in the healthy controls (n = 11) and the surviving patients with SFTS in the acute phase (n = 30) and SFTS in the recovery phase (n = 30). (C), (E): The percentages and numbers of Th1 and Th2 cells at admission in the surviving patients (n = 30) and the deceased patients (n = 12). (F), (G): Dynamic changes in the percentages and numbers of Th1 and Th2 cells in the surviving patients (n = 30) and the deceased patients (n = 12). These parameters were monitored at indicated time points for the entire hospital stay of the patients, and the dashed line represents the median of the uninfected controls. The data are shown as the median +- 95% CI. Statistical analysis was performed using the Mann-Whitney U test or the Wilcoxon matched pair test. The level of significance is indicated as follows: ns, not significant; *p < .05; **p < .01; ***p < .001; ****p < .0001; Fig. 3
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry Other assay
Other assay