Antibody data
- Antibody Data
- Antigen structure
- References [12]
- Comments [0]
- Validations
- Other assay [12]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 37-6200 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- DDB1 Monoclonal Antibody (2B12D1)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Reactivity
- Human, Mouse
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 2B12D1
- Vial size
- 100 μg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Global and transcription-coupled repair of 8-oxoG is initiated by nucleotide excision repair proteins.
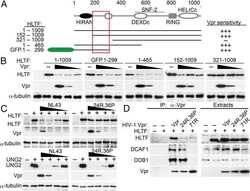
HIV-1 Vpr counteracts HLTF-mediated restriction of HIV-1 infection in T cells.
Restriction of hepatitis B virus replication by c-Abl-induced proteasomal degradation of the viral polymerase.
HIV-1 Vpr Reprograms CLR4(DCAF1) E3 Ubiquitin Ligase to Antagonize Exonuclease 1-Mediated Restriction of HIV-1 Infection.
CRL4 antagonizes SCFFbxo7-mediated turnover of cereblon and BK channel to regulate learning and memory.
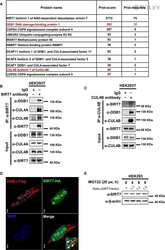
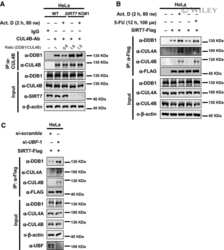
SIRT7 deacetylates DDB1 and suppresses the activity of the CRL4 E3 ligase complexes.
Activation of c-Abl Kinase Potentiates the Anti-myeloma Drug Lenalidomide by Promoting DDA1 Protein Recruitment to the CRL4 Ubiquitin Ligase.
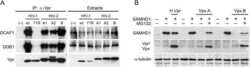
HIV-1 and HIV-2 exhibit divergent interactions with HLTF and UNG2 DNA repair proteins.
CRL4A(CRBN) E3 ubiquitin ligase restricts BK channel activity and prevents epileptogenesis.
Ubiquitin hydrolase UCH-L1 destabilizes mTOR complex 1 by antagonizing DDB1-CUL4-mediated ubiquitination of raptor.
Lentiviral Vpx accessory factor targets VprBP/DCAF1 substrate adaptor for cullin 4 E3 ubiquitin ligase to enable macrophage infection.
Lentiviral Vpr usurps Cul4-DDB1[VprBP] E3 ubiquitin ligase to modulate cell cycle.
Kumar N, Theil AF, Roginskaya V, Ali Y, Calderon M, Watkins SC, Barnes RP, Opresko PL, Pines A, Lans H, Vermeulen W, Van Houten B
Nature communications 2022 Feb 21;13(1):974
Nature communications 2022 Feb 21;13(1):974
HIV-1 Vpr counteracts HLTF-mediated restriction of HIV-1 infection in T cells.
Yan J, Shun MC, Zhang Y, Hao C, Skowronski J
Proceedings of the National Academy of Sciences of the United States of America 2019 May 7;116(19):9568-9577
Proceedings of the National Academy of Sciences of the United States of America 2019 May 7;116(19):9568-9577
Restriction of hepatitis B virus replication by c-Abl-induced proteasomal degradation of the viral polymerase.
Hou L, Zhao J, Gao S, Ji T, Song T, Li Y, Wang J, Geng C, Long M, Chen J, Lin H, Cai X, Cang Y
Science advances 2019 Feb;5(2):eaau7130
Science advances 2019 Feb;5(2):eaau7130
HIV-1 Vpr Reprograms CLR4(DCAF1) E3 Ubiquitin Ligase to Antagonize Exonuclease 1-Mediated Restriction of HIV-1 Infection.
Yan J, Shun MC, Hao C, Zhang Y, Qian J, Hrecka K, DeLucia M, Monnie C, Ahn J, Skowronski J
mBio 2018 Oct 23;9(5)
mBio 2018 Oct 23;9(5)
CRL4 antagonizes SCFFbxo7-mediated turnover of cereblon and BK channel to regulate learning and memory.
Song T, Liang S, Liu J, Zhang T, Yin Y, Geng C, Gao S, Feng Y, Xu H, Guo D, Roberts A, Gu Y, Cang Y
PLoS genetics 2018 Jan;14(1):e1007165
PLoS genetics 2018 Jan;14(1):e1007165
SIRT7 deacetylates DDB1 and suppresses the activity of the CRL4 E3 ligase complexes.
Mo Y, Lin R, Liu P, Tan M, Xiong Y, Guan KL, Yuan HX
The FEBS journal 2017 Nov;284(21):3619-3636
The FEBS journal 2017 Nov;284(21):3619-3636
Activation of c-Abl Kinase Potentiates the Anti-myeloma Drug Lenalidomide by Promoting DDA1 Protein Recruitment to the CRL4 Ubiquitin Ligase.
Gao S, Geng C, Song T, Lin X, Liu J, Cai Z, Cang Y
The Journal of biological chemistry 2017 Mar 3;292(9):3683-3691
The Journal of biological chemistry 2017 Mar 3;292(9):3683-3691
HIV-1 and HIV-2 exhibit divergent interactions with HLTF and UNG2 DNA repair proteins.
Hrecka K, Hao C, Shun MC, Kaur S, Swanson SK, Florens L, Washburn MP, Skowronski J
Proceedings of the National Academy of Sciences of the United States of America 2016 Jul 5;113(27):E3921-30
Proceedings of the National Academy of Sciences of the United States of America 2016 Jul 5;113(27):E3921-30
CRL4A(CRBN) E3 ubiquitin ligase restricts BK channel activity and prevents epileptogenesis.
Liu J, Ye J, Zou X, Xu Z, Feng Y, Zou X, Chen Z, Li Y, Cang Y
Nature communications 2014 May 21;5:3924
Nature communications 2014 May 21;5:3924
Ubiquitin hydrolase UCH-L1 destabilizes mTOR complex 1 by antagonizing DDB1-CUL4-mediated ubiquitination of raptor.
Hussain S, Feldman AL, Das C, Ziesmer SC, Ansell SM, Galardy PJ
Molecular and cellular biology 2013 Mar;33(6):1188-97
Molecular and cellular biology 2013 Mar;33(6):1188-97
Lentiviral Vpx accessory factor targets VprBP/DCAF1 substrate adaptor for cullin 4 E3 ubiquitin ligase to enable macrophage infection.
Srivastava S, Swanson SK, Manel N, Florens L, Washburn MP, Skowronski J
PLoS pathogens 2008 May 9;4(5):e1000059
PLoS pathogens 2008 May 9;4(5):e1000059
Lentiviral Vpr usurps Cul4-DDB1[VprBP] E3 ubiquitin ligase to modulate cell cycle.
Hrecka K, Gierszewska M, Srivastava S, Kozaczkiewicz L, Swanson SK, Florens L, Washburn MP, Skowronski J
Proceedings of the National Academy of Sciences of the United States of America 2007 Jul 10;104(28):11778-83
Proceedings of the National Academy of Sciences of the United States of America 2007 Jul 10;104(28):11778-83
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
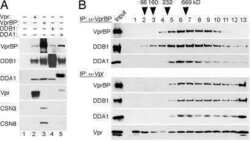
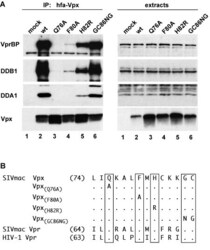
- Figure 1 Conserved amino acid residues in Vpx C-terminal region mediate the association with DDA1-DDB1-VprBP complex. (A) hfa-tagged wild type (lane 2) or mutant (lanes 3-6) Vpx proteins were transiently expressed in HEK 293T cells and precipitated from detergent extracts with FLAG-M2 affinity resin. DDB1, VprBP, DDA1 and hfa-Vpx were detected in immune complexes (left panel) and cell extracts (right panel) by immunoblotting and visualized by enhanced chemiluminescence. (B) Amino acid sequences of the C-terminal regions of SIVmac 239 Vpx, SIVmac 239 Vpr, and HIV-1 NL43 Vpr are aligned and amino acid substitutions for the conserved residues in Vpx are indicated. Numbers indicate the positions of the first amino acid residue shown in each protein sequence. Dots identify amino acid identities and letters specify amino acids in the single-letter code.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
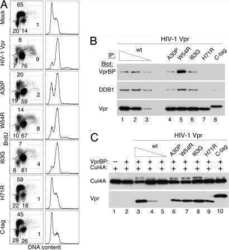
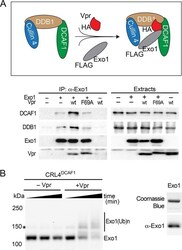
- FIG 4 Vpr recruits Exo1 to the CRL4 DCAF1 E3 for ubiquitination. (A) Vpr connects Exo1 to the endogenously expressed DDB1-DCAF1 module of the CRL4 DCAF1 E3 complex in vivo . (Top) Schematic representation of Vpr-mediated recruitment of Exo1 to the CRL4 DCAF1 E3 complex. The placement of HA and FLAG epitope tags on Vpr and Exo1, respectively, is indicated. (Bottom) FLAG-Exo1 was transiently coexpressed with HA-tagged wild-type HIV-1 Vpr (wt) or Vpr(F69A), which does not bind DCAF1, in HEK293T cells, as indicated. Endogenous DCAF1, DDB1, and ectopic Vpr and Exo1 were revealed in Exo1 immune complexes and in detergent extracts by immunoblotting. (B) Vpr-dependent Exo1 ubiquitination by recombinant CRL4 DCAF1 E3 in vitro . (Left) In vitro ubiquitination assays were performed with recombinant Exo1 incubated with CRL4 DCAF1c E3 reconstituted from recombinant subunits, in the absence or presence of recombinant HIV-1 NL4-3 Vpr. Reactions were sampled over time, and native (Exo1) and ubiquitinated [Exo1(Ub)n] forms of Exo1 were revealed by immunoblotting with alpha-Exo1 antibody. An asterisk indicates a nonspecific band. (Right) Recombinant Exo1 expressed and purified from E. coli was analyzed by SDS-PAGE, gel stained with Coomassie blue (upper), and confirmed by immunoblotting with alpha-Exo1 antibody.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
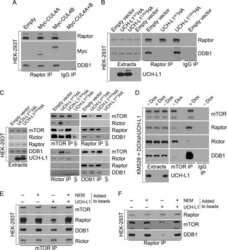
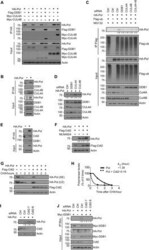
- Fig. 3 CRL4 cdt2 E3 ubiquitin ligase targets HBV polymerase for ubiquitination. ( A ) Co-IP of HA polymerase with CRL4 components in HEK293T cells by overexpression HA polymerase, Myc-CUL4A, Myc-CUL4B, and Flag-DDB1. ( B ) Co-IP of HA polymerase with endogenous CRL4 in HEK293T cells. ( C ) Ubiquitination of polymerase by Flag IP in Huh7 cells expressing HA polymerase, Flag-ub, and siDDB1, siCUL4A, siCUL4B, or a combination of siCUL4A and siCUL4B and treated with MG132 for 8 hours before harvest. Flag immunoprecipitates (top) or total cell lysates (bottom) were then analyzed by Western blot. ( D ) Immunoblot for polymerase protein level in Huh7 cells knocking down CRL4 components. ( E ) Co-IP of HA polymerase with endogenous Cdt2 and DDB1 in Huh7 cells. ( F ) The transfected HEK293T cells were treated with DMSO or MLN4924 (0.2 muM) for 8 hours before harvest. ( G ) CHX chasing analysis for polymerase stability in HEK293T cells expressing Flag-Cdt2. Cells were treated with CHX (100 mug/mL) for the indicated time. ( H ) Time course of polymerase protein decay from Fig. 3G . Each band of the Western blot for polymerase was quantified with NIH ImageJ software. Half-life ( t 1/2 ) was estimated as the time for degradation of 50% of the protein. ( I ) HEK293T cells were transfected with control siRNA or siCdt2 and HA polymerase. Total cell lysates were subjected to Western blotting. ( J ) Co-IP of DDB1 with polymerase in Huh7 cells by overexpressing HA polymerase, Myc-DDB1, and siCd
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA Other assay
Other assay