Antibody data
- Antibody Data
- Antigen structure
- References [12]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [3]
- Immunohistochemistry [1]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA5-12231 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Cytokeratin Pan Monoclonal Antibody (C-11)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- MA5-12231 targets Cytokeratin Multi in IF, WB and IHC (P) applications and shows reactivity with Amphibian, Bovine, Human, mouse, and Rat samples.
- Antibody clone number
- C-11
- Concentration
- 0.2 mg/mL
Submitted references Myocyte Enhancer Factor 2C as a New Player in Human Breast Cancer Brain Metastases.
Type III Interferon Restriction by Porcine Epidemic Diarrhea Virus and the Role of Viral Protein nsp1 in IRF1 Signaling.
New frontiers in circulating tumor cell analysis: A reference guide for biomolecular profiling toward translational clinical use.
A pilot study to explore circulating tumour cells in pancreatic cancer as a novel biomarker.
Localization of progenitor/stem cells in the human tympanic membrane.
Primary cell cultures from human renal cortex and renal-cell carcinoma evidence a differential expression of two spliced isoforms of Annexin A3.
Characterization of goldfish fin cells in culture: some evidence of an epithelial cell profile.
Induction of persistent colitis by a human commensal, enterotoxigenic Bacteroides fragilis, in wild-type C57BL/6 mice.
Mind bomb 1 in the lymphopoietic niches is essential for T and marginal zone B cell development.
Inhibition of VEGFR2 prevents DMBA-induced mammary tumor formation.
Keratins modulate c-Flip/extracellular signal-regulated kinase 1 and 2 antiapoptotic signaling in simple epithelial cells.
TNP-470 inhibits 7,12-dimethylbenz[a]anthracene-induced mammary tumor formation when administered before the formation of carcinoma in situ but is not additive with tamoxifen.
Galego S, Kauppila LA, Malhó R, Pimentel J, Brito MA
Cells 2021 Feb 12;10(2)
Cells 2021 Feb 12;10(2)
Type III Interferon Restriction by Porcine Epidemic Diarrhea Virus and the Role of Viral Protein nsp1 in IRF1 Signaling.
Zhang Q, Ke H, Blikslager A, Fujita T, Yoo D
Journal of virology 2018 Feb 15;92(4)
Journal of virology 2018 Feb 15;92(4)
New frontiers in circulating tumor cell analysis: A reference guide for biomolecular profiling toward translational clinical use.
Becker TM, Caixeiro NJ, Lim SH, Tognela A, Kienzle N, Scott KF, Spring KJ, de Souza P
International journal of cancer 2014 Jun 1;134(11):2523-33
International journal of cancer 2014 Jun 1;134(11):2523-33
A pilot study to explore circulating tumour cells in pancreatic cancer as a novel biomarker.
Khoja L, Backen A, Sloane R, Menasce L, Ryder D, Krebs M, Board R, Clack G, Hughes A, Blackhall F, Valle JW, Dive C
British journal of cancer 2012 Jan 31;106(3):508-16
British journal of cancer 2012 Jan 31;106(3):508-16
Localization of progenitor/stem cells in the human tympanic membrane.
Knutsson J, von Unge M, Rask-Andersen H
Audiology & neuro-otology 2011;16(4):263-9
Audiology & neuro-otology 2011;16(4):263-9
Primary cell cultures from human renal cortex and renal-cell carcinoma evidence a differential expression of two spliced isoforms of Annexin A3.
Bianchi C, Bombelli S, Raimondo F, Torsello B, Angeloni V, Ferrero S, Di Stefano V, Chinello C, Cifola I, Invernizzi L, Brambilla P, Magni F, Pitto M, Zanetti G, Mocarelli P, Perego RA
The American journal of pathology 2010 Apr;176(4):1660-70
The American journal of pathology 2010 Apr;176(4):1660-70
Characterization of goldfish fin cells in culture: some evidence of an epithelial cell profile.
Mauger PE, Labbé C, Bobe J, Cauty C, Leguen I, Baffet G, Le Bail PY
Comparative biochemistry and physiology. Part B, Biochemistry & molecular biology 2009 Mar;152(3):205-15
Comparative biochemistry and physiology. Part B, Biochemistry & molecular biology 2009 Mar;152(3):205-15
Induction of persistent colitis by a human commensal, enterotoxigenic Bacteroides fragilis, in wild-type C57BL/6 mice.
Rhee KJ, Wu S, Wu X, Huso DL, Karim B, Franco AA, Rabizadeh S, Golub JE, Mathews LE, Shin J, Sartor RB, Golenbock D, Hamad AR, Gan CM, Housseau F, Sears CL
Infection and immunity 2009 Apr;77(4):1708-18
Infection and immunity 2009 Apr;77(4):1708-18
Mind bomb 1 in the lymphopoietic niches is essential for T and marginal zone B cell development.
Song R, Kim YW, Koo BK, Jeong HW, Yoon MJ, Yoon KJ, Jun DJ, Im SK, Shin J, Kong MP, Kim KT, Yoon K, Kong YY
The Journal of experimental medicine 2008 Oct 27;205(11):2525-36
The Journal of experimental medicine 2008 Oct 27;205(11):2525-36
Inhibition of VEGFR2 prevents DMBA-induced mammary tumor formation.
Heffelfinger SC, Yan M, Gear RB, Schneider J, LaDow K, Warshawsky D
Laboratory investigation; a journal of technical methods and pathology 2004 Aug;84(8):989-98
Laboratory investigation; a journal of technical methods and pathology 2004 Aug;84(8):989-98
Keratins modulate c-Flip/extracellular signal-regulated kinase 1 and 2 antiapoptotic signaling in simple epithelial cells.
Gilbert S, Loranger A, Marceau N
Molecular and cellular biology 2004 Aug;24(16):7072-81
Molecular and cellular biology 2004 Aug;24(16):7072-81
TNP-470 inhibits 7,12-dimethylbenz[a]anthracene-induced mammary tumor formation when administered before the formation of carcinoma in situ but is not additive with tamoxifen.
Heffelfinger SC, Gear RB, Schneider J, LaDow K, Yan M, Lu F, Pyle AL, Warshawsky D
Laboratory investigation; a journal of technical methods and pathology 2003 Jul;83(7):1001-11
Laboratory investigation; a journal of technical methods and pathology 2003 Jul;83(7):1001-11
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-Cytokeratin Pan Monoclonal Antibody (C-11) (Product # MA5-12231) and a 40-60 kDa band corresponding to Cytokeratin Pan (Cytokeratin 10, Cytokeratin 13, Cytokeratin 18, Cytokeratin 4, Cytokeratin 5, Cytokeratin 6 and Cytokeratin 8) was observed across in all tissue lysates, except Mouse brain. Tissue extracts (30 µg lysate) of Mouse Esophagus (Lane 1), Mouse Colon (Lane 2), Mouse Lung (Lane 3), Mouse Brain (Lane 4) were electrophoresed using NuPAGE™ 10% Bis-Tris Protein Gel (Product # NP0301BOX). Resolved proteins were then transferred onto a nitrocellulose membrane (Product # IB23002) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1 µg/mL) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177,1:10000 dilution) using the iBright™ FL1500 Imaging System (Product # A44115). Chemiluminescent detection was performed using SuperSignal™ West Dura Extended Duration Substrate (Product # 34076).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of pan Cytokeratin was performed by loading 25 µg of Hela (lane 1), A431 (lane 2) and PC12 (lane 3) onto an SDS polyacrylamide gel. Proteins were transferred to a PVDF membrane and blocked at 4ºC overnight. The membrane was probed with a pan Cytokeratin monoclonal antibody (Product # MA5-12231) at a dilution of 1:50 overnight at 4°C, washed in TBST, and probed with an HRP-conjugated secondary antibody for 1 hr at room temperature in the dark. Chemiluminescent detection was performed using Pierce ECL Plus Western Blotting Substrate (Product # 32132). Results show a band at ~46 and 55 kDa.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
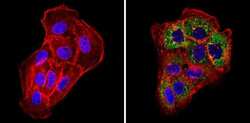
- Experimental details
- Immunofluorescent analysis of pan Cytokeratin (green) showing staining in the cytoplasm of A431 cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a pan Cytokeratin monoclonal antibody (Product # MA5-12231) in 3% BSA-PBS at a dilution of 1:50 and incubated overnight at 4ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. Actin was stained using Alexa Fluor 554 (red) and nuclei were stained with Hoechst or DAPI (blue). Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
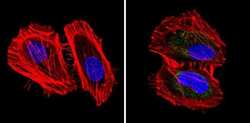
- Experimental details
- Immunofluorescent analysis of pan Cytokeratin (green) showing staining in the cytoplasm of Hela cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a pan Cytokeratin monoclonal antibody (Product # MA5-12231) in 3% BSA-PBS at a dilution of 1:50 and incubated overnight at 4ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. Actin was stained using Alexa Fluor 554 (red) and nuclei were stained with Hoechst or DAPI (blue). Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence analysis of Cytokeratin Pan (Cytokeratin 10, Cytokeratin 13, Cytokeratin 18, Cytokeratin 4, Cytokeratin 5, Cytokeratin 6 and Cytokeratin 8) was performed using 70% confluent log phase A-431 cells. The cells were fixed and permeabilized with ice-cold acetone at 4°C for 5 minutes, and blocked with 2% BSA for 1 hour at room temperature. The cells were labeled with Cytokeratin Pan Monoclonal Antibody (C-11) (Product # MA5-12231, 5 µg/mL) in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32766, 1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b: Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300 dilution). Panel d represents the merged image showing cytoskeletal localization. Panel e represents Raji cells showing no expression of cytokeratin. Panel f represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Formalin-fixed, paraffin-embedded human colon carcinoma stained with Keratin, Multi antibody using peroxidase-conjugate and AEC chromogen. Note cytoplasmic staining of tumor cells.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
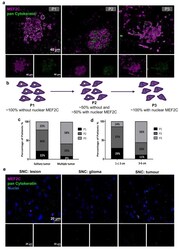
- Experimental details
- Figure 1 Myocyte Enhancer Factor 2C expression in resected human brain metastases derived from breast cancer patients and from glioma patients. Immunofluorescence analysis of myocyte enhancer factor 2C (MEF2C) (purple) and of the epithelial and tumoral marker, pan Cytokeratin (green), in human brain metastases from breast cancer patients, revealed distinct MEF2C labeling patterns that were considered as three different phenotypes: ~100% cells presenting an extranuclear location (P1); ~50% cells presented extranuclear location and ~50% presented overall cell staining (P2); or ~100% cells presenting overall cell staining (P3) ( a ). Schematic representation of the subcellular MEF2C distribution, according to the considered phenotypes (P1, P2, P3) ( b ). Semi-quantitative analysis of the percentage of patients presenting each phenotype, regarding the number ( c ) and the size of the metastases ( d ), shows the progression from P1 to P3 as the number and size of metastases increase. Double labeling of MEF2C and pan Cytokeratin in brain tissue samples derived from glioma patients as non-breast cancer brain metastases control ( e ) showing no relevant expression of the protein. Twenty-four cases of BCBM (P1, n = 3; P2, n = 10; P3, n = 11) and ten control cases were studied; ten fields per section and one section per case were analyzed.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
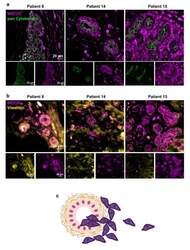
- Experimental details
- Figure 2 Myocyte Enhancer Factor 2C expression in human breast cancer primary tumors. Double immunofluorescence analysis of myocyte enhancer factor 2C (MEF2C; purple) with the epithelial and tumoral marker, pan Cytokeratin (green), showed that MEF2C expressing cells did not significantly express pan Cytokeratin and were found in disorganized mammary ducts as well as in the surrounding tissue ( a ). Double immunofluorescence analysis of MEF2C (purple) with the mesenchymal marker, vimentin (yellow), showed that MEF2C expressing cells in mammary ducts also expressed vimentin ( b ). Schematic representation of the first stages of the metastatic cascade, showing MEF2C expressing cells (purple) in the mammary duct and invading the surrounding tissue ( c ). Three resected human primary BC cases were studied; ten fields per section and one section per case were analyzed.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot