Antibody data
- Antibody Data
- Antigen structure
- References [32]
- Comments [0]
- Validations
- Western blot [2]
- Other assay [28]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 32-8100 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- alpha Synuclein Monoclonal Antibody (Syn 211)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Description
- This antibody reacts with the alpha-synuclein protein. This antibody does not cross-react with beta- or gamma-synuclein or synoretin. During development reactivity was confirmed with human. Reactivity with other species has not been tested.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- Syn 211
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Mercury is present in neurons and oligodendrocytes in regions of the brain affected by Parkinson's disease and co-localises with Lewy bodies.
Rapid macropinocytic transfer of α-synuclein to lysosomes.
Viral alpha-synuclein knockdown prevents spreading synucleinopathy.
Small molecule inhibitors of α-synuclein oligomers identified by targeting early dopamine-mediated motor impairment in C. elegans.
Detection of pathological alpha-synuclein aggregates in human iPSC-derived neurons and tissue.
Anti-α-synuclein ASO delivered to monoamine neurons prevents α-synuclein accumulation in a Parkinson's disease-like mouse model and in monkeys.
Spinal Cord and Motor Neuron TDP-43 Pathology in a Sporadic Inclusion Body Myositis Patient.
Alpha-Synuclein Continues to Enhance SNARE-Dependent Vesicle Docking at Exorbitant Concentrations.
Increased vulnerability of nigral dopamine neurons after expansion of their axonal arborization size through D2 dopamine receptor conditional knockout.
Recapitulating Parkinson's disease pathology in a three-dimensional human neural cell culture model.
Autophagy inhibition promotes SNCA/alpha-synuclein release and transfer via extracellular vesicles with a hybrid autophagosome-exosome-like phenotype.
Pathophysiological Consequences of Neuronal α-Synuclein Overexpression: Impacts on Ion Homeostasis, Stress Signaling, Mitochondrial Integrity, and Electrical Activity.
Reversible Conformational Conversion of α-Synuclein into Toxic Assemblies by Glucosylceramide.
Deferiprone Rescues Behavioral Deficits Induced by Mild Iron Exposure in a Mouse Model of Alpha-Synuclein Aggregation.
Towards a Non-Human Primate Model of Alpha-Synucleinopathy for Development of Therapeutics for Parkinson's Disease: Optimization of AAV1/2 Delivery Parameters to Drive Sustained Expression of Alpha Synuclein and Dopaminergic Degeneration in Macaque.
α-Synuclein interferes with the ESCRT-III complex contributing to the pathogenesis of Lewy body disease.
Cortical phosphorylated α-Synuclein levels correlate with brain wave spectra in Parkinson's disease.
Post-mortem brain analyses of the Lothian Birth Cohort 1936: extending lifetime cognitive and brain phenotyping to the level of the synapse.
A brain-targeted, modified neurosin (kallikrein-6) reduces α-synuclein accumulation in a mouse model of multiple system atrophy.
The loss of glucose-regulated protein 78 (GRP78) during normal aging or from siRNA knockdown augments human alpha-synuclein (α-syn) toxicity to rat nigral neurons.
Alpha-Synuclein affects neurite morphology, autophagy, vesicle transport and axonal degeneration in CNS neurons.
Lack of additive role of ageing in nigrostriatal neurodegeneration triggered by α-synuclein overexpression.
Hypoestoxide reduces neuroinflammation and α-synuclein accumulation in a mouse model of Parkinson's disease.
Aggregation of αSynuclein promotes progressive in vivo neurotoxicity in adult rat dopaminergic neurons.
Glucose regulated protein 78 diminishes α-synuclein neurotoxicity in a rat model of Parkinson disease.
Neuropathology in mice expressing mouse alpha-synuclein.
Sumoylation inhibits alpha-synuclein aggregation and toxicity.
Neuroprotection by minocycline caused by direct and specific scavenging of peroxynitrite.
Hypocretin (orexin) loss in Parkinson's disease.
Fine epitope mapping of monoclonal antibodies specific to human alpha-synuclein.
Effect of mutant alpha-synuclein on dopamine homeostasis in a new human mesencephalic cell line.
An in vitro model of Parkinson's disease: linking mitochondrial impairment to altered alpha-synuclein metabolism and oxidative damage.
Pamphlett R, Bishop DP
PloS one 2022;17(1):e0262464
PloS one 2022;17(1):e0262464
Rapid macropinocytic transfer of α-synuclein to lysosomes.
Bayati A, Banks E, Han C, Luo W, Reintsch WE, Zorca CE, Shlaifer I, Del Cid Pellitero E, Vanderperre B, McBride HM, Fon EA, Durcan TM, McPherson PS
Cell reports 2022 Jul 19;40(3):111102
Cell reports 2022 Jul 19;40(3):111102
Viral alpha-synuclein knockdown prevents spreading synucleinopathy.
Menon S, Kofoed RH, Nabbouh F, Xhima K, Al-Fahoum Y, Langman T, Mount HTJ, Shihabuddin LS, Sardi SP, Fraser PE, Watts JC, Aubert I, Tandon A
Brain communications 2021;3(4):fcab247
Brain communications 2021;3(4):fcab247
Small molecule inhibitors of α-synuclein oligomers identified by targeting early dopamine-mediated motor impairment in C. elegans.
Chen KS, Menezes K, Rodgers JB, O'Hara DM, Tran N, Fujisawa K, Ishikura S, Khodaei S, Chau H, Cranston A, Kapadia M, Pawar G, Ping S, Krizus A, Lacoste A, Spangler S, Visanji NP, Marras C, Majbour NK, El-Agnaf OMA, Lozano AM, Culotti J, Suo S, Ryu WS, Kalia SK, Kalia LV
Molecular neurodegeneration 2021 Nov 12;16(1):77
Molecular neurodegeneration 2021 Nov 12;16(1):77
Detection of pathological alpha-synuclein aggregates in human iPSC-derived neurons and tissue.
Stojkovska I, Mazzulli JR
STAR protocols 2021 Mar 19;2(1):100372
STAR protocols 2021 Mar 19;2(1):100372
Anti-α-synuclein ASO delivered to monoamine neurons prevents α-synuclein accumulation in a Parkinson's disease-like mouse model and in monkeys.
Alarcón-Arís D, Pavia-Collado R, Miquel-Rio L, Coppola-Segovia V, Ferrés-Coy A, Ruiz-Bronchal E, Galofré M, Paz V, Campa L, Revilla R, Montefeltro A, Kordower JH, Vila M, Artigas F, Bortolozzi A
EBioMedicine 2020 Sep;59:102944
EBioMedicine 2020 Sep;59:102944
Spinal Cord and Motor Neuron TDP-43 Pathology in a Sporadic Inclusion Body Myositis Patient.
Cathcart SJ, Greene EP, Powell SZ, Arumanayagam AS, Rivera AL, Tawil R, Appel SH, Cykowski MD
Journal of neuropathology and experimental neurology 2020 Oct 1;79(10):1130-1133
Journal of neuropathology and experimental neurology 2020 Oct 1;79(10):1130-1133
Alpha-Synuclein Continues to Enhance SNARE-Dependent Vesicle Docking at Exorbitant Concentrations.
Hawk BJD, Khounlo R, Shin YK
Frontiers in neuroscience 2019;13:216
Frontiers in neuroscience 2019;13:216
Increased vulnerability of nigral dopamine neurons after expansion of their axonal arborization size through D2 dopamine receptor conditional knockout.
Giguère N, Delignat-Lavaud B, Herborg F, Voisin A, Li Y, Jacquemet V, Anand-Srivastava M, Gether U, Giros B, Trudeau LÉ
PLoS genetics 2019 Aug;15(8):e1008352
PLoS genetics 2019 Aug;15(8):e1008352
Recapitulating Parkinson's disease pathology in a three-dimensional human neural cell culture model.
Taylor-Whiteley TR, Le Maitre CL, Duce JA, Dalton CF, Smith DP
Disease models & mechanisms 2019 Apr 9;12(4)
Disease models & mechanisms 2019 Apr 9;12(4)
Autophagy inhibition promotes SNCA/alpha-synuclein release and transfer via extracellular vesicles with a hybrid autophagosome-exosome-like phenotype.
Minakaki G, Menges S, Kittel A, Emmanouilidou E, Schaeffner I, Barkovits K, Bergmann A, Rockenstein E, Adame A, Marxreiter F, Mollenhauer B, Galasko D, Buzás EI, Schlötzer-Schrehardt U, Marcus K, Xiang W, Lie DC, Vekrellis K, Masliah E, Winkler J, Klucken J
Autophagy 2018;14(1):98-119
Autophagy 2018;14(1):98-119
Pathophysiological Consequences of Neuronal α-Synuclein Overexpression: Impacts on Ion Homeostasis, Stress Signaling, Mitochondrial Integrity, and Electrical Activity.
Tolö J, Taschenberger G, Leite K, Stahlberg MA, Spehlbrink G, Kues J, Munari F, Capaldi S, Becker S, Zweckstetter M, Dean C, Bähr M, Kügler S
Frontiers in molecular neuroscience 2018;11:49
Frontiers in molecular neuroscience 2018;11:49
Reversible Conformational Conversion of α-Synuclein into Toxic Assemblies by Glucosylceramide.
Zunke F, Moise AC, Belur NR, Gelyana E, Stojkovska I, Dzaferbegovic H, Toker NJ, Jeon S, Fredriksen K, Mazzulli JR
Neuron 2018 Jan 3;97(1):92-107.e10
Neuron 2018 Jan 3;97(1):92-107.e10
Deferiprone Rescues Behavioral Deficits Induced by Mild Iron Exposure in a Mouse Model of Alpha-Synuclein Aggregation.
Carboni E, Tatenhorst L, Tönges L, Barski E, Dambeck V, Bähr M, Lingor P
Neuromolecular medicine 2017 Sep;19(2-3):309-321
Neuromolecular medicine 2017 Sep;19(2-3):309-321
Towards a Non-Human Primate Model of Alpha-Synucleinopathy for Development of Therapeutics for Parkinson's Disease: Optimization of AAV1/2 Delivery Parameters to Drive Sustained Expression of Alpha Synuclein and Dopaminergic Degeneration in Macaque.
Koprich JB, Johnston TH, Reyes G, Omana V, Brotchie JM
PloS one 2016;11(11):e0167235
PloS one 2016;11(11):e0167235
α-Synuclein interferes with the ESCRT-III complex contributing to the pathogenesis of Lewy body disease.
Spencer B, Kim C, Gonzalez T, Bisquertt A, Patrick C, Rockenstein E, Adame A, Lee SJ, Desplats P, Masliah E
Human molecular genetics 2016 Mar 15;25(6):1100-15
Human molecular genetics 2016 Mar 15;25(6):1100-15
Cortical phosphorylated α-Synuclein levels correlate with brain wave spectra in Parkinson's disease.
Caviness JN, Lue LF, Hentz JG, Schmitz CT, Adler CH, Shill HA, Sabbagh MN, Beach TG, Walker DG
Movement disorders : official journal of the Movement Disorder Society 2016 Jul;31(7):1012-9
Movement disorders : official journal of the Movement Disorder Society 2016 Jul;31(7):1012-9
Post-mortem brain analyses of the Lothian Birth Cohort 1936: extending lifetime cognitive and brain phenotyping to the level of the synapse.
Henstridge CM, Jackson RJ, Kim JM, Herrmann AG, Wright AK, Harris SE, Bastin ME, Starr JM, Wardlaw J, Gillingwater TH, Smith C, McKenzie CA, Cox SR, Deary IJ, Spires-Jones TL
Acta neuropathologica communications 2015 Sep 4;3:53
Acta neuropathologica communications 2015 Sep 4;3:53
A brain-targeted, modified neurosin (kallikrein-6) reduces α-synuclein accumulation in a mouse model of multiple system atrophy.
Spencer B, Valera E, Rockenstein E, Trejo-Morales M, Adame A, Masliah E
Molecular neurodegeneration 2015 Sep 23;10:48
Molecular neurodegeneration 2015 Sep 23;10:48
The loss of glucose-regulated protein 78 (GRP78) during normal aging or from siRNA knockdown augments human alpha-synuclein (α-syn) toxicity to rat nigral neurons.
Salganik M, Sergeyev VG, Shinde V, Meyers CA, Gorbatyuk MS, Lin JH, Zolotukhin S, Gorbatyuk OS
Neurobiology of aging 2015 Jun;36(6):2213-23
Neurobiology of aging 2015 Jun;36(6):2213-23
Alpha-Synuclein affects neurite morphology, autophagy, vesicle transport and axonal degeneration in CNS neurons.
Koch JC, Bitow F, Haack J, d'Hedouville Z, Zhang JN, Tönges L, Michel U, Oliveira LM, Jovin TM, Liman J, Tatenhorst L, Bähr M, Lingor P
Cell death & disease 2015 Jul 9;6(7):e1811
Cell death & disease 2015 Jul 9;6(7):e1811
Lack of additive role of ageing in nigrostriatal neurodegeneration triggered by α-synuclein overexpression.
Bourdenx M, Dovero S, Engeln M, Bido S, Bastide MF, Dutheil N, Vollenweider I, Baud L, Piron C, Grouthier V, Boraud T, Porras G, Li Q, Baekelandt V, Scheller D, Michel A, Fernagut PO, Georges F, Courtine G, Bezard E, Dehay B
Acta neuropathologica communications 2015 Jul 25;3:46
Acta neuropathologica communications 2015 Jul 25;3:46
Hypoestoxide reduces neuroinflammation and α-synuclein accumulation in a mouse model of Parkinson's disease.
Kim C, Ojo-Amaize E, Spencer B, Rockenstein E, Mante M, Desplats P, Wrasidlo W, Adame A, Nchekwube E, Oyemade O, Okogun J, Chan M, Cottam H, Masliah E
Journal of neuroinflammation 2015 Dec 18;12:236
Journal of neuroinflammation 2015 Dec 18;12:236
Aggregation of αSynuclein promotes progressive in vivo neurotoxicity in adult rat dopaminergic neurons.
Taschenberger G, Garrido M, Tereshchenko Y, Bähr M, Zweckstetter M, Kügler S
Acta neuropathologica 2012 May;123(5):671-83
Acta neuropathologica 2012 May;123(5):671-83
Glucose regulated protein 78 diminishes α-synuclein neurotoxicity in a rat model of Parkinson disease.
Gorbatyuk MS, Shabashvili A, Chen W, Meyers C, Sullivan LF, Salganik M, Lin JH, Lewin AS, Muzyczka N, Gorbatyuk OS
Molecular therapy : the journal of the American Society of Gene Therapy 2012 Jul;20(7):1327-37
Molecular therapy : the journal of the American Society of Gene Therapy 2012 Jul;20(7):1327-37
Neuropathology in mice expressing mouse alpha-synuclein.
Rieker C, Dev KK, Lehnhoff K, Barbieri S, Ksiazek I, Kauffmann S, Danner S, Schell H, Boden C, Ruegg MA, Kahle PJ, van der Putten H, Shimshek DR
PloS one 2011;6(9):e24834
PloS one 2011;6(9):e24834
Sumoylation inhibits alpha-synuclein aggregation and toxicity.
Krumova P, Meulmeester E, Garrido M, Tirard M, Hsiao HH, Bossis G, Urlaub H, Zweckstetter M, Kügler S, Melchior F, Bähr M, Weishaupt JH
The Journal of cell biology 2011 Jul 11;194(1):49-60
The Journal of cell biology 2011 Jul 11;194(1):49-60
Neuroprotection by minocycline caused by direct and specific scavenging of peroxynitrite.
Schildknecht S, Pape R, Müller N, Robotta M, Marquardt A, Bürkle A, Drescher M, Leist M
The Journal of biological chemistry 2011 Feb 18;286(7):4991-5002
The Journal of biological chemistry 2011 Feb 18;286(7):4991-5002
Hypocretin (orexin) loss in Parkinson's disease.
Fronczek R, Overeem S, Lee SY, Hegeman IM, van Pelt J, van Duinen SG, Lammers GJ, Swaab DF
Brain : a journal of neurology 2007 Jun;130(Pt 6):1577-85
Brain : a journal of neurology 2007 Jun;130(Pt 6):1577-85
Fine epitope mapping of monoclonal antibodies specific to human alpha-synuclein.
Choi JY, Park HJ, Seong YM, Choi EY, Min BR, Rhim H
Neuroscience letters 2006 Apr 10-17;397(1-2):53-8
Neuroscience letters 2006 Apr 10-17;397(1-2):53-8
Effect of mutant alpha-synuclein on dopamine homeostasis in a new human mesencephalic cell line.
Lotharius J, Barg S, Wiekop P, Lundberg C, Raymon HK, Brundin P
The Journal of biological chemistry 2002 Oct 11;277(41):38884-94
The Journal of biological chemistry 2002 Oct 11;277(41):38884-94
An in vitro model of Parkinson's disease: linking mitochondrial impairment to altered alpha-synuclein metabolism and oxidative damage.
Sherer TB, Betarbet R, Stout AK, Lund S, Baptista M, Panov AV, Cookson MR, Greenamyre JT
The Journal of neuroscience : the official journal of the Society for Neuroscience 2002 Aug 15;22(16):7006-15
The Journal of neuroscience : the official journal of the Society for Neuroscience 2002 Aug 15;22(16):7006-15
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
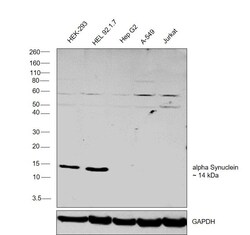
- Western blot was performed using Anti-alpha Synuclein Monoclonal Antibody (Syn 211) (Product # 32-8100) and a 14 kDa band corresponding to alpha Synuclein was observed across the cell lines tested. Membrane enriched extracts (30 µg lysate) of HEK-293 (Lane 1), HEL 92.1.7 (Lane 2), Hep G2 (Lane 3), A-549 (Lane 4), Jurkat (Lane 5) were electrophoresed using NuPAGE™ 4-12% Bis-Tris Protein Gel (Product # NP0321BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was fixed with 0.4% paraformaldehyde for 30 min. The blot was probed with the primary antibody (1:1000 dilution) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177,1:10000 dilution) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of SNCA was performed by loading 30 µg of HeLa wildtype (Lane 1), HeLa SNCA knockout (Lane 2) whole cell extracts. The blot was probed with Anti-SNCA Monoclonal Antibody (Product # 32-8100) (1 µg/ml) and Goat anti-Mouse IgG (H+L), Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177) (1:4000 dilution). Loss of signal upon CRISPR mediated knockout (KO) confirms that antibody is specific to SNCA.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. alpha-Synuclein sumoylation in vitro in cells and in mouse brain. (A) alpha-Synuclein is SUMO2 modified in vitro. 500 ng alpha-synuclein, 500 ng SUMO2, 150 ng Aos1/Uba2, 200 ng Ubc9, and 5-10 ng E3 ligase fragments were incubated for 30 min at 30degC with and without ATP in a volume of 20 ul. Reactions were stopped with SDS sample buffer before analysis by SDS-PAGE and immunoblotting with mouse monoclonal anti-alpha-synuclein antibody (Syn211; Invitrogen). (B) alpha-Synuclein modification by SUMO2 in HEK293T cells. Plasmids encoding His 6 -SUMO2 (His 6 -S2) or/and alpha-synuclein were transfected in HEK293T cells. SUMO substrates were purified by Ni 2+ affinity chromatography (Ni 2+ -NTA) under denaturing conditions, and a sumoylated alpha-synuclein band at ~35 kD (stars in A and B) was detected with an alpha-synuclein antibody (Syn211). 1% of total input (TP) and 25% of elution fractions were loaded. (C, top) A schematic representation of the Thy1.2/His 6 -SUMO transgene. (bottom) alpha-Synuclein sumoylation in mouse brain tissue. Total His 6 -SUMO2-conjugated proteins were isolated using Ni 2+ -NTA affinity chromatography. TBLs and eluates were probed with anti-alpha-synuclein antibody recognizing mouse alpha-synuclein (clone 42). alpha-Synuclein modified by a single SUMO2 molecule, indicated with a star, could be detected in the eluate obtained from His 6 -SUMO2 transgenics.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
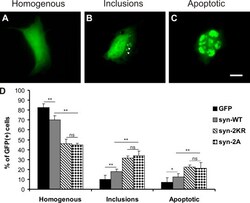
- Figure 4. Sumoylation-impaired alpha-synuclein mutants (2KR and 2A) induce increased toxicity and inclusion formation compared with WT alpha-synuclein in HEK cells. (A-D) HEK293T cells were transfected with bicistronic plasmids expressing alpha-synuclein variants (syn-WT; 2KR: K96 and 102R; and 2A: D98A and E104A) with a PDZ binding tag (HSTTRV) and PDZ domain-EGFP fusion protein (GFP). (A-C) Cells were classified in three groups based on GFP fluorescence distribution: homogenous (A), containing intracellular GFP-positive inclusions (B, arrowheads), and preapoptotic or having fragmented nuclei by DAPI staining (C). Images display exemplary cells to illustrate the different categories into which cells were grouped. Because individual cells displayed variations in expression levels, exposure times were adjusted differently to avoid overexposure. At the level of total cell lysates, expression levels of the different constructs were the same between the different experimental conditions. Bar, 20 um. (D) Bar graphs represent percentages of all GFP-positive cells in the three categories summarized as mean +- SEM. Comparisons were made with KyPlot 5.0 using Student's t test (*, P < 0.05; **, P < 0.01). n = 6. Note that cell counts were performed by direct observation at the microscope using identical optical parameters for all experimental conditions. ns, not significant.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
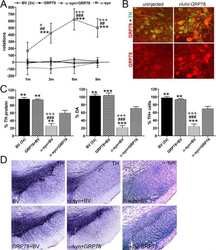
- Figure 1 Overexpression of alpha Syn variants in PMN. ( a ) Vector maps of the plasmids used to overexpress EGFP, alpha Syn-WT, -A30P and -A53T. The respective transcripts are expressed under the control of a human synapsin-1 promoter. ITR: AAV-2 inverted terminal repeat. Int: intron. SV40-pA: SV40 polyadenylation site. WPRE: Woodchuck hepatitis virus posttranscriptional regulatory element. bGH-pA: bovine growth hormone polyadenylation site. ( b and c ) Immunoblots of whole cell protein lysates from PMN transfected with the given plasmids (cells were lysed on DIV 5). In b , an antibody specific for human alpha Syn (LB509, Invitrogen) was used to detect only the alpha Syn expressed by the plasmids. In c , total cellular alpha Syn levels were assessed using an antibody recognizing both human and rat alpha Syn (BD). At the bottom, quantifications of the band intensities normalized to beta -tubulin are shown ( n =3; error bars represent means+-S.E.M.; ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
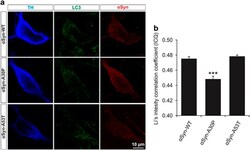
- Figure 5 Colocalization of LC3 and alpha Syn in PMN transfected with different alpha Syn variants. ( a ) Representative confocal micrographs of PMN transfected with the respective alpha Syn variant given on the left side and immunostained against TH, LC3 and alpha Syn as specified on top. ( b ) Quantification of colocalization between alpha Syn and LC3 in the soma of TH-positive neurons after transfection with different alpha Syn variants represented by means of Li's intensity correlation coefficient. Cells transfected with alpha Syn-A30P show significantly less colocalization than those transfected with alpha Syn-WT or -A53T. Statistics: one-way ANOVA followed by Tukey-Kramer post hoc test, *** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Axonal transport of synaptophysin-positive vesicles in PMN transfected with different alpha Syn variants. ( a ) Representative micrograph showing PMN transduced with AAV.synaptophysin-EGFP (expressing EGFP-tagged synaptophysin) and co-transfected with p. alpha Syn-WT and p.dsRed (to identify transfected neurons) (DIV 5). In the higher magnification pictures, the EGFP-positive vesicles (arrows) can be seen along the transfected axon. ( b ) Representative kymographs of the movements of EGFP-tagged synaptophysin along neurites transduced with the plasmids given on the left side within 10 s ( y axis). Arrows point at representative moving vesicles, asterisks mark stationary vesicles. ( c-f ) Quantifications of synaptophysin-EGFP transport in neurites transfected with the given plasmids. Statistics: one-way ANOVA followed by Dunnett's post hoc test, * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
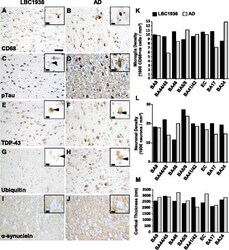
- Fig. 5 Stereological assessments of brain pathology and integrity. Representative images of microglial (CD68; a + b ), phosphorylated-tau ( c + d ), TDP-43 ( e + f ), ubiquitin ( g + h ) and alpha-synuclein ( i + j ) staining in the hippocampus (CA1) from LBC1936 ( a , c , e , g , i ) and AD ( b , d , f , h , j ) brains. Large scale bar = 50 mum, insert scale bars = 10 mum. k Histogram showing neuronal densities in eight cortical regions from the LBC1936 (black bars) and AD (grey bars) brains. l Histogram showing microglial densities (CD68-positive cells) in eight cortical regions from the LBC1936 and AD brains. m Histogram of cortical thickness measurements from eight cortical regions in the LBC1936 and AD brains
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 7 Using synaptoneurosomes to enrich and analyze synaptic proteins by western blotting. a Representative enrichment blot showing the exclusion of nuclear histone (17 kDa) from the synaptoneurosome preparation and retention of synapsin (40-80 kDa). b GluN2B western blot from the LBC1936 (black bars) and AD (grey bars) preparations. To assess protein integrity and control for post-mortem degradation [ 52 ], band2 (black arrow; 150 kDa) was divided by band1 (grey arrow; 170 kDa) to generate a ratio, and a value >=1 (red dashed line) is achieved by all samples except the LBC1936 EC (asterix). c Representative synaptophysin (40 kDa) blot of LBC1936 synaptoneurosomes and AD samples. d Representative total tau (Tau13; 45-60 kDa) blot of LBC1936 samples and AD samples. e Phosphorylated-tau (PHF1; 50-65 kDa) blot shows almost exclusive expression in the AD samples compared to the LBC1936 synapses. f Representative VDAC (29-32 kDa) blot of LBC1936 and AD synaptoneurosomes. GAPDH (36 kDa) or beta-actin (42 kDa) was run as a loading control. Histogram represents the mean of three experimental repeats and the bars represent the quantification of the bands directly above
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 Lentivirus vectors over-expressing mutant neurosin reduce the accumulation and propagation of alpha-synuclein. Wild-type neurosin (LV-wt-NR), neurosin-apoB (LV-wt-NR-apoB), the point mutant neurosin (LV-NR-R80Q) and R80Q-apoB (LV-NR-R80Q-apoB) were cloned into the 3 rd generation lentivirus vector. a B103 neuronal cells were transduced with the vectors and examined by immunoblot for neurosin and the epitope tag V5. b B103 transduced cells were also immunostained for neurosin expression. c Compared to LV-Control, neuronal cells transduced with the neurosin constructs expressed higher levels of neurosin, levels were comparable between the wt and mutant neurosin constructs. d An in vitro neuronal co-culture system was devised to mimic the propagation of alpha-syn. B103 neuronal ""Donor cells"" infected with LV-alpha-syn or LV-Control (red) were plated in cell culture inserts containing a 0.4 mum membrane. B103 neuronal ""Acceptor cells"" infected with LV-NR-R80Q, LV-NR-R80Q-apoB or LV-Control were plated on coverslips. e Confocal microscopy and immunocytochemical analysis showing alpha-syn (red) from the Donor Cells was secreted and taken up by Acceptor cells that were double labeled with an antibody against neurosin (green). f Image analysis of double labeled neuronal cells, results are expressed as percent of acceptor cells containing alpha-syn immunoreactivity. * - indicates one way ANOVA with post hoc Dunnett's, p < 0.05 compared to LV-Control. # - indicates one
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Delivery of LV-NR-R80Q-apoB reduces alpha-syn accumulation in MBP-alpha-syn tg mice. a Bright field microscopy analysis of serial longitudinal vibratome sections from the non-tg and MBP-alpha-syn tg mice immunostained with an antibody against total alpha-syn (syn-1) and analyzed for alpha-syn positive cells following treatment with LV-Control, or LV-NR-R80Q-apoB vector. b Computer aided image analysis for numbers of alpha-syn positive cells in the striatum and the corpus. c Samples that included the cortex, corpus callosum and striatum were fractioned by ultracentrifugation and analyzed by immunoblot analysis with an antibody against total alpha-syn (syn-1). d Densitometry analysis of the levels of alpha-syn immunoreactivity plotted against actin. e Laser scanning confocal microscopy of vibratome sections from the non-tg and MBP-alpha-syn tg immunostained with an antibody against human alpha-syn (syn211) and analyzed for alpha-syn positive cells following treatment with LV-Control, or LV-NR-R80Q-apoB vector. f Computer aided image analysis of the number of alpha-syn positive cells in the striatum and the corpus callosum. g Bright field microscopy analysis of serial longitudinal vibratome sections form the non-tg and MBP-alpha-syn tg immunostained with an antibody against phosphorylated alpha-syn (Ser129) and analyzed for alpha-syn positive cells following treatment with LV-Control, or LV-NR-R80Q-apoB vector. h Computer aided image analysis for numbers of phosphorylated
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5 Delivery of LV-NR-R80Q-apoB reduces the propagation of alpha-syn to astrocytes and clearance via microglia in MBP-alpha-syn tg mice. Vibratome brain sections from the non-tg and MBP-alpha-syn tg that received i.p. injections of LV-Control or LV-NR-R80Q-apoB were double immunofluorescence labeled with antibodies against cellular markers and human alpha-syn and analyzed with the laser scanning confocal microscope with an optical image of 1 mum with fluorescent signals in co-registry. Dotted box to the left depicts the image field zoomed represented under detail. a Double immunolabeling for the astrocyte marker S100 (red) and human alpha-syn (green) with nuclei (DAPI, blue). Co-immunolabeling is represented by signal in yellow. b Computer aided image analysis of the % of S100 cells displaying alpha-syn immunofluorescence in the corpus callosum and striatum. c Double immunolabeling for the microglial marker Iba-1 (red) and human alpha-syn (green) with nuclei (DAPI, blue). Co-immunolabeling is represented by signal in yellow. d Computer aided image analysis of the % of Iba-1 cells displaying alpha-syn immunofluorescence in the corpus callosum and striatum. n = 10 mice per group 9-10 m/o at the end of the treatment. Scale bar = 10 mum, detail = 20 mum. # indicates statistical significance ( p < 0.05, one way ANOVA, post hoc Tukey-Kramer) compared to LV-Control treated MBP-alpha-syn tg mice
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Hypoestoxide reduces deposition of alpha-synuclein in a mouse model of PD. a Representative immunohistochemical staining of alpha-synuclein in the frontal cortex, hippocampus, and striatum. b Optical density analysis for alpha-synuclein-positive neuropil in the frontal cortex ( n = 5 per each group; two-way ANOVA, Bonferroni's multiple comparison post-test; *** p < 0.001). Error bars represent +-SEM. c The numbers of alpha-synuclein-positive cells in the frontal cortex. d , e Optical density analysis of immunoreactivity of alpha-synuclein in the hippocampus ( d ) and in the striatum ( e ) ( n = 5 per each group; two-way ANOVA, Bonferroni's multiple comparison post-test; ** p < 0.01, *** p < 0.001). Error bars represent +-SEM. f , g Biochemical analysis of SDS-soluble ( f ) and SDS-insoluble ( g ) fractions from the frontal cortex for alpha-synuclein ( n = 5 per each group; two-way ANOVA, Bonferroni's multiple comparison post-test; ** p < 0.01, *** p < 0.001). Error bars represent +-SEM. Scale bars = 250 mum (low magnification) and 25 mum (high magnification)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
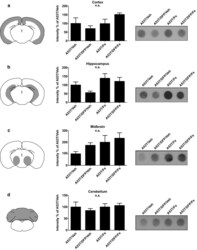
- Fig. 5 Dot blot assay for aggregated human aSyn in A53T mice. The tissue samples were homogenized and incubated for 10 min with proteinase K to digest the soluble forms of aSyn. Samples were blotted and revealed through an antihuman aSyn antibody to visualize proteinase K-resistant fraction. Each row shows a scheme of the analyzed area, the quantification of n =5 blots and an exemplary blot for a . cortex; b . hippocampus; c . midbrain; and d . cerebellum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 6 Histological evaluation of aSyn aggregation in A53T mice at disease onset using a p-S129-aSyn antibody for all the regions tested. This posttranslational modification was shown to be found in the aggregated form of aSyn only. Each row shows a scheme of the analyzed region, a quantification of p-S129-aSyn signal through fluorescence intensity ( n =5 animals for each group), and representative photomicrographs. a . CA3 region; b . cortex (CX); c . dentate gyrus (DG); d . deep mesencephalic reticular nucleus (DpMe); e . red nucleus (RN); f . substantia nigra (SN). Scale bar = 20 um
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
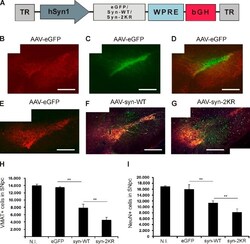
- Figure 5. Reduced sumoylation exacerbates alpha-synuclein neurotoxicity in vivo. (A) A schematic representation of vector genomes used in the in vivo rat model of PD. Recombinant adeno-associated viral vectors (AAV2) express either EGFP, WT alpha-synuclein (syn-WT), or alpha-synuclein K96R and K102R (syn-2KR) under the control of neuron-specific human synapsin 1 gene promoter (hSyn1). Vectors also encode inverted terminal repeats (TR) and small control elements woodchuck hepatitis virus posttranscriptional control element (WPRE) and bovine growth hormone (bGH)-derived polyadenylation site. (B) VMAT2-positive (VMAT2 + ) neurons (red) in the left SN. (C) rAAV2-mediated GFP transduction of SNpc. (D) Overlay of VMAT2 + and GFP-transduced neurons in the left SN. (E-G) Coimmunostaining of VMAT2 + (red) and human alpha-synuclein (green). Survival of VMAT2 + neurons in SNpc of animals injected with rAAV2 vectors expressing EGFP (E), human WT alpha-synuclein (F), or alpha-synuclein-2KR (G). Panels B-G are each a composite of two separate fluorescent images (Axioplan2 with a 5x objective and AxioVision 4.7). (H and I) Respective unbiased stereological quantification of surviving VMAT2 + (H) or NeuN + (I) cells in the transduced SNpc. The graphs display VMAT2 + or NeuN + cell numbers in animals injected with rAAV2 expressing EGFP, WT alpha-synuclein (syn-WT), and alpha-synuclein-2KR (syn-2KR). N.I., contralateral noninjected side. n = 6-8 per experimental group. Results are mean +- SEM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Representative example of alpha-synuclein and thioflavin S staining in cultured cells (A) Transfected SH-SY5Y cells expressing A53T alpha-synuclein (alpha-syn) were cultured as described (), fixed, and stained using alpha-syn syn211 (red) and Thio S (green). The microscopy setup included an Olympus IX-FLA using BP360-370 (DAPI), BP420-480 (Thio S), and BP510-550 (alpha-syn) exciter filters. (B) iPSC-derived midbrain neurons from controls or patients harboring the A53T alpha-synuclein mutation were cultured for 90 days, fixed, and stained using LB509 for alpha-syn (red) and Thio S (green), and nuclei were detected with DAPI (blue), using a similar setup as described in (A). (C) Representative images of Thio S stained alpha-synuclein positive neurites from A53T iPSC-derived neurons as described in (B). The scale bars are 10 mum in all images. Arrowheads indicate large juxtanuclear inclusions, while arrows indicate cells with smaller punctated aggregates and diffuse Thio S staining. Asterisks show examples of background/non-specific staining of dead cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 10.1371/journal.pone.0262464.g001 Fig 1 Mercury in the locus ceruleus, substantia nigra, cerebral cortex and striatum in Parkinson's disease. ( A ) Black mercury grains (eg, open arrowheads) are present in the cytoplasm of most locus ceruleus neurons in PD1 (main image) and PD2 (inset). Lewy bodies with haloes (filled arrowheads) are seen in some mercury-containing neurons. Numerous oligodendrocytes have small mercury deposits (eg, arrows) adjacent to their nuclei. Autometallography/Luxol fast blue. ( B ) Magenta immunostaining of a-synuclein shows co-localisation of Lewy bodies (open arrowheads) with black-staining mercury in locus ceruleus neurons of PD1. An extra-neuronal a-synuclein aggregate (open arrow) has no associated mercury. No Lewy bodies are present in a locus ceruleus neuron not containing mercury (filled arrowhead). The right upper inset shows a nearby locus ceruleus neuron at higher magnification with Lewy body/mercury co-localisation. The left lower inset shows mercury within a Lewy body. Autometallography/a-synuclein Magenta/hematoxylin. ( C ) Brown DAB immunostaining of a-synuclein shows co-localisation of a Lewy body with mercury (filled arrow) in a remaining substantia nigra neuron of PD1. A nearby Lewy neurite (right upper inset) shows associated mercury grains (open arrowhead). Some intraneuronal (filled arrowhead) and extraneuronal (*) Lewy bodies do not appear to contain mercury, possibly because of masking by the dense brown DAB staining. Scattered o
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
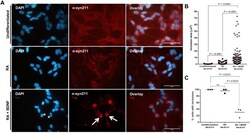
- Fig. 2. Exogenous addition of alpha-syn oligomers induces the intracellular aggregation of alpha-syn in 2D SH-SY5Y cultures. (A) Double immunostaining of undifferentiated and differentiated SH-SY5Y cells reveals intracellular aggregation of alpha-syn following 24 h incubation with seeding oligomers. alpha-Syn inclusions in undifferentiated cells display several distinct, punctate accumulations dispersed throughout the cytoplasm, consistent with aggregates. In contrast, the RA+BDNF cell lines show a single prominent accumulation (red; arrows) and obscure the nucleus (asterisks). Scale bars: 50 um. (B) Quantification of inclusion area (reflected as um 2 ) shows inclusions that are present in differentiated cells to be much larger than those in undifferentiated cells (measurements obtained from three independent experiments; n= 50). All data points are present, with mean shown as a straight line. (C) The percentage of cells containing inclusions was determined by counting 100 cells per repeat. Cells were deemed to contain inclusions if aggregation matched that in reference images when compared to controls. Data points are presented from three independent biological repeats. (B,C) Differences between treatments were tested for significance using Kruskal-Wallis test with post-hoc Dwass-Steel-Chritchlow-Fligner for >6 data points and Conover-Inman for
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
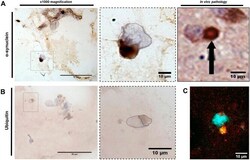
- Fig. 5. alpha-Syn-seeded inclusions in 3D are indicative of in vivo Lewy body inclusions. alpha-Syn oligomer treatments to 3D-differentiated cultures develop alpha-syn-positive inclusions that demonstrate the same morphology as in vivo Lewy bodies (LBs; A) and are positive for the marker ubiquitin (B). Dotted line in purple highlights cell nuclei, whereas brown dotted line highlights stained area. (C) Immunofluorescence shows colocalisation of alpha-syn (red) and ubiquitin (green) staining. Nuclei were visualised using DAPI staining (blue). Scale bars: 50 um (left) and 10 um (right). In vivo pathology image displayed with permission from Springer Nature. This image is not published under the terms of the CC-BY license of this article. For permission to reuse, please see Chesselet et al. (2012) .
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA