Antibody data
- Antibody Data
- Antigen structure
- References [5]
- Comments [0]
- Validations
- Flow cytometry [2]
- Other assay [9]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 17-1389-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD138 (Syndecan-1) Monoclonal Antibody (DL-101), APC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The DL-101 monoclonal antibody reacts with human CD138, also known as syndecan-1. CD138 is a transmembrane protein containing chondroitin sulfate and heparin sulfate moieties responsible for binding to the extracellular matrix components. CD138 is not expressed by mature B cells but is present on pre-B cells and the finally differentiated plasma cells. Applications Reported: This DL-101 antibody has been reported for use in flow cytometric analysis. Applications Tested: This DL-101 antibody has been pre-titrated and tested by flow cytometric analysis of the U266 cell line. This can be used at 5 µL (0.125 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 633-647 nm; Emission: 660 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- DL-101
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Highly efficient CRISPR-Cas9-mediated gene knockout in primary human B cells for functional genetic studies of Epstein-Barr virus infection.
Comprehensive Cell Surface Antigen Analysis Identifies Transferrin Receptor Protein-1 (CD71) as a Negative Selection Marker for Human Neuronal Cells.
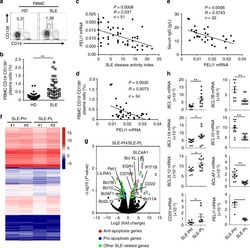
Peli1 negatively regulates noncanonical NF-κB signaling to restrain systemic lupus erythematosus.
Mitochondrial-Targeted Decyl-Triphenylphosphonium Enhances 2-Deoxy-D-Glucose Mediated Oxidative Stress and Clonogenic Killing of Multiple Myeloma Cells.
PPARγ negatively regulates T cell activation to prevent follicular helper T cells and germinal center formation.
Akidil E, Albanese M, Buschle A, Ruhle A, Pich D, Keppler OT, Hammerschmidt W
PLoS pathogens 2021 Apr;17(4):e1009117
PLoS pathogens 2021 Apr;17(4):e1009117
Comprehensive Cell Surface Antigen Analysis Identifies Transferrin Receptor Protein-1 (CD71) as a Negative Selection Marker for Human Neuronal Cells.
Menon V, Thomas R, Elgueta C, Horl M, Osborn T, Hallett PJ, Bartos M, Isacson O, Pruszak J
Stem cells (Dayton, Ohio) 2019 Oct;37(10):1293-1306
Stem cells (Dayton, Ohio) 2019 Oct;37(10):1293-1306
Peli1 negatively regulates noncanonical NF-κB signaling to restrain systemic lupus erythematosus.
Liu J, Huang X, Hao S, Wang Y, Liu M, Xu J, Zhang X, Yu T, Gan S, Dai D, Luo X, Lu Q, Mao C, Zhang Y, Shen N, Li B, Huang M, Zhu X, Jin J, Cheng X, Sun SC, Xiao Y
Nature communications 2018 Mar 19;9(1):1136
Nature communications 2018 Mar 19;9(1):1136
Mitochondrial-Targeted Decyl-Triphenylphosphonium Enhances 2-Deoxy-D-Glucose Mediated Oxidative Stress and Clonogenic Killing of Multiple Myeloma Cells.
Schibler J, Tomanek-Chalkley AM, Reedy JL, Zhan F, Spitz DR, Schultz MK, Goel A
PloS one 2016;11(11):e0167323
PloS one 2016;11(11):e0167323
PPARγ negatively regulates T cell activation to prevent follicular helper T cells and germinal center formation.
Park HJ, Kim DH, Choi JY, Kim WJ, Kim JY, Senejani AG, Hwang SS, Kim LK, Tobiasova Z, Lee GR, Craft J, Bothwell AL, Choi JM
PloS one 2014;9(6):e99127
PloS one 2014;9(6):e99127
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
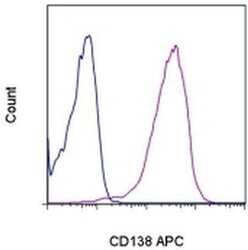
- Experimental details
- Staining of U266 cells with Mouse IgG1 K Isotype Control APC (Product # 17-4714-81) (blue histogram) or Anti-Human CD138 (Syndecan-1) APC (purple histogram). Total viable cells were used for analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of U266 cells with Mouse IgG1 K Isotype Control APC (Product # 17-4714-81) (blue histogram) or Anti-Human CD138 (Syndecan-1) APC (purple histogram). Total viable cells were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
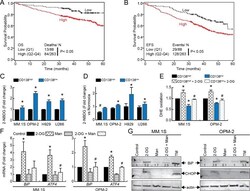
- Fig 4 Glycolytic gene expression correlates with MM patient survival and increased glucose uptake can induce 2-DG-mediated oxidative and ER stress in of CD138 low cells. Kaplan-Meier graphs from TT2 trial clustered on glycolytic gene signatures ( HK2 , ALDOA , TPI1 , GAPDH , PGK1 , PKM2 , and LDHA ) showing cumulative probabilities of A. OS and B. EFS in MM patients. Glucose uptake assays in HMCLs cultured under C. normoxia or D. hypoxia for 3 days. Cells were incubated with 2-NBDG followed by APC-CD138 staining and flow analysis. For panel C, mean fluorescence values (MFI) values was normalized to CD138 high cells and presented fold change. *p < 0.01 vs. control. Bars represent mean of three independent runs +- SEM. For panel D, 2-NBDG uptake under hypoxia was compared with normoxia for CD138 low and CD138 high separately and depicted as hypoxia-induced fold change. *p < 0.01 vs. CD138 high cells under normoxia, # p < 0.01 vs. CD138 low cells under normoxia. E. MM.1S and OPM-2 cells were treated without or with 2-DG for 1.5 h followed by DHE staining and flow analysis. Normalized MFI relative to CD138 high cells are shown. MM.1S and OPM-2 cells were treated with 2-DG and/or mannose or 24 h followed by F. qRT-PCR analysis of BiP or ATF4 G. Western blot analysis for BiP (78 kDa), CHOP (27 kDa) or beta-actin (42 kDa, loading control), tunicamycin (TM, 5 muM is used as a positive control. The quantification of BiP after normalization to untreated control is shown below each band
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 5 CD138 low cells have altered mitochondrial properties that can be utilized to induce oxidative stress by 10-TPP treatment. A. Representative electron micrographs of sorted CD138 low and CD138 high MM.1S and OPM-2 cells. For ultrastructure analysis, all mitochondria were selected (indicated by *), manually scored, and assigned either condensed or orthodox morphology. Low magnification shows the entire cell with inset used for analysis of mitochondria under higher magnification. *p < 0.01 vs. CD138 low cells. HMCLs were co-stained with APC-CD138 antibody and B. MitoTracker Green or C. Rhodamine 123 and analyzed by flow cytometry. Data is presented as the fold change relative to CD138 high cells. *p < 0.01 vs. CD138 high cells. D. Structure of 10-TPP; 10-TPP-induced H 2 DCF-DA oxidation in CD138 high and CD138 low cells in MM1.S and OPM-2 cells. Data of three independent runs is presented as the fold change relative to CD138 high cells. *p < 0.01 vs. CD138 high cells, # p < 0.01 vs. CD138 low cells. E. HMCLs were transduced with Ad-CMV or Ad-MnSOD, treated with 10-TPP, and MitoSOX oxidation was analyzed by flow cytometry. Data is presented as fold change normalized to control cells expressing Ad-CMV. Antimycin A treatment was used as a positive control. *p < 0.01 vs. control cells (Ad-CMV or Ad-MnSOD), # p < 0.05 vs. 10-TPP treated Ad-CMV cells. Representative Western blot of HMCLs transduced with Ad-CMV or Ad-MnSOD (MOI = 50), whole-cell extract was made at 48 h and probe
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
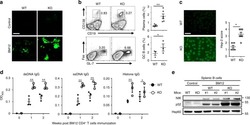
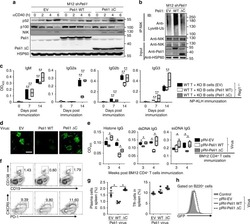
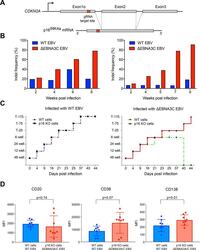
- Fig 4 p16 INK4a is a functional barrier to EBV driven proliferation of lymphoblastoid cells. (A) Blueprint of the primary transcript and the spliced mRNA with the three exons of CDKN2A on chromosome 9 encoding the p16 INK4a protein. The target site of the RNP complex within the 1st exon (exon1alpha) (chr9:21,974,678-21,974,827) is shown. (B) Study of the biological effect of the CDKN2A knockout in a time course experiment. WT and p16 KO cells were mixed such that the fraction of the latter was in the order of 10 to 20%, when the cells were infected with WT or DeltaEBNA3C EBV strains. The knockout status of the CDKN2A gene was studied by next generation sequencing to analyze the CD46 locus of the mixed cell populations over time. The fraction of cells with a disabled CDKN2A gene increased in cells infected with DeltaEBNA3C EBV exceeding 80% after eight weeks, whereas the knockout status of CDKN2A in the population of cells infected with WT EBV did not show a clear trend. Results from two biological replicates are shown, additional replicates can be found in S4A Fig . (C) Cell numbers of four different B cell populations were plotted as a function of days post nucleofection (x-axis) versus the format of the cell culture vessel (y-axis) starting with a single well in a 48-well cluster plate. 2x10 6 B cells with an intact CDKN2A locus (WT cells) or cells with an edited CDKN2A gene (p16 KO cells) were infected with wild-type (WT) EBV (left panel) or DeltaEBNA3C EBV (right panel).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
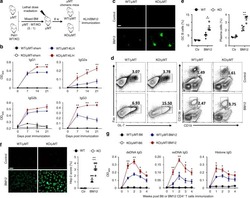
- 10.1371/journal.pone.0167323.g001 Fig 1 Flow cytometric analysis of stem-like cells in HMCLs. A. Representative dot plots for CD138 vs. side scatter and B. quantification of % CD138 low fractions. C. Hoechst 33342 staining for SP with or without verapamil. Gate represents the % SP fractions, MP = main population. D. Quantification of % SP cells in MM.1S and OPM-2 cell lines +- verapamil. Bars represent mean of three independent runs +- SEM, *p < 0.05 vs. control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- 10.1371/journal.pone.0167323.g002 Fig 2 Hypoxia increases CD138 low population and alters transcriptional profile of HMCLs. Cell were cultured at either normoxia (21% O 2 ) or hypoxia (1% O 2 ) for 3 days, labeled with CD138-APC antibody and the percentage of CD138 low and CD138 high cells were analyzed by flow cytometry. A. Representative dot plots of different HMCLs and B. quantification of % CD138 low fractions under normoxia or hypoxia. C. qRT-PCR analysis of SDC1 , stem cell genes ( NANOG , OCT4 ), and VEGF-A . For panels B and C, bars represent mean of three independent runs +- SEM. *p < 0.05 vs. normoxia.
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry