46-1338-42
antibody from Invitrogen Antibodies
Targeting: PROM1
AC133, CD133, CORD12, MCDR2, PROML1, RP41, STGD4
Antibody data
- Antibody Data
- Antigen structure
- References [8]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [10]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 46-1338-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD133 (Prominin-1) Monoclonal Antibody (TMP4), PerCP-eFluor™ 710, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The TMP4 monoclonal antibody reacts with human CD133 (Prominin-1), a 120 kDa member of the pentaspan family of proteins, which also includes Prominin-2. Their expression is found within plasma membrane protrusions such as epithelial microvilli. CD133 can exist in a number of alternatively spliced isoforms, and the protein has several N-linked glycosylation sites: the occurrence of both may be tissue-dependent. Human CD133 was first identified as an epitope expressed on CD34+ hematopoietic progenitors. Although the ligand and function of CD133 remain unknown, it has since proven to be very useful as a marker for both stem cells and cancer stem cells. In addition to its expression on hematopoietic precursors, CD133 has been used to identify tumorigenic colon cancer stem cells, brain cancer stem cells, prostate cancer stem cells, in addition to others. The binding of the TMP4 antibody does not block the binding of another anti-human CD133 antibody, EMK08 (Product # 12-1339) indicating that they recognize distinct epitopes. Applications Reported: This TMP4 antibody has been reported for use in flow cytometric analysis. Applications Tested: This TMP4 antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.25 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. PerCP-eFluor® 710 emits at 710 nm and is excited with the blue laser (488 nm); it can be used in place of PerCP-Cyanine5.5. We recommend using a 710/50 bandpass filter, however, the 695/40 bandpass filter is an acceptable alternative. Please make sure that your instrument is capable of detecting this fluorochrome. Light sensitivity: This tandem dye is sensitive to photo-induced oxidation. Please protect this vial and stained samples from light. Fixation: Samples can be stored in IC Fixation Buffer (Product # 00-8222) (100 µL of cell sample + 100 µL of IC Fixation Buffer) or 1-step Fix/Lyse Solution (Product # 00-5333) for up to 3 days in the dark at 4°C with minimal impact on brightness and FRET efficiency/compensation. Some generalizations regarding fluorophore performance after fixation can be made, but clone specific performance should be determined empirically. Excitation: 488 nm; Emission: 710 nm; Laser: Blue Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- TMP4
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Astrocytes influence medulloblastoma phenotypes and CD133 surface expression.
Chemerin enhances the adhesion and migration of human endothelial progenitor cells and increases lipid accumulation in mice with atherosclerosis.
The Deubiquitinase USP4 Stabilizes Twist1 Protein to Promote Lung Cancer Cell Stemness.
Profiling and Targeting of Energy and Redox Metabolism in Grade 2 Bladder Cancer Cells with Different Invasiveness Properties.
Inhibition of Fas associated phosphatase 1 (Fap1) facilitates apoptosis of colon cancer stem cells and enhances the effects of oxaliplatin.
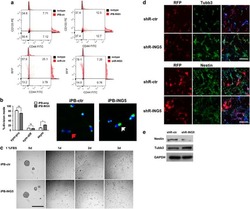
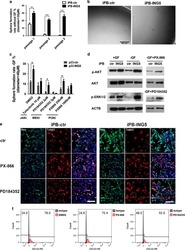
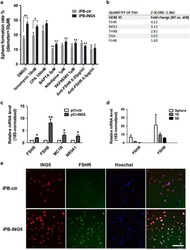
ING5 activity in self-renewal of glioblastoma stem cells via calcium and follicle stimulating hormone pathways.
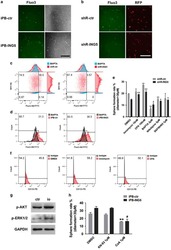
Evaluation of cancer stem cell markers CD133, CD44, CD24: association with AKT isoforms and radiation resistance in colon cancer cells.
Comparative evaluation of differentiation potential of menstrual blood- versus bone marrow-derived stem cells into hepatocyte-like cells.
Gronseth E, Gupta A, Koceja C, Kumar S, Kutty RG, Rarick K, Wang L, Ramchandran R
PloS one 2020;15(7):e0235852
PloS one 2020;15(7):e0235852
Chemerin enhances the adhesion and migration of human endothelial progenitor cells and increases lipid accumulation in mice with atherosclerosis.
Jia J, Yu F, Xiong Y, Wei W, Ma H, Nisi F, Song X, Yang L, Wang D, Yuan G, Zhou H
Lipids in health and disease 2020 Sep 20;19(1):207
Lipids in health and disease 2020 Sep 20;19(1):207
The Deubiquitinase USP4 Stabilizes Twist1 Protein to Promote Lung Cancer Cell Stemness.
Li F, Hu Q, He T, Xu J, Yi Y, Xie S, Ding L, Fu M, Guo R, Xiao ZJ, Niu M
Cancers 2020 Jun 15;12(6)
Cancers 2020 Jun 15;12(6)
Profiling and Targeting of Energy and Redox Metabolism in Grade 2 Bladder Cancer Cells with Different Invasiveness Properties.
Pasquale V, Ducci G, Campioni G, Ventrici A, Assalini C, Busti S, Vanoni M, Vago R, Sacco E
Cells 2020 Dec 11;9(12)
Cells 2020 Dec 11;9(12)
Inhibition of Fas associated phosphatase 1 (Fap1) facilitates apoptosis of colon cancer stem cells and enhances the effects of oxaliplatin.
Huang W, Bei L, Eklund EA
Oncotarget 2018 May 25;9(40):25891-25902
Oncotarget 2018 May 25;9(40):25891-25902
ING5 activity in self-renewal of glioblastoma stem cells via calcium and follicle stimulating hormone pathways.
Wang F, Wang AY, Chesnelong C, Yang Y, Nabbi A, Thalappilly S, Alekseev V, Riabowol K
Oncogene 2018 Jan 18;37(3):286-301
Oncogene 2018 Jan 18;37(3):286-301
Evaluation of cancer stem cell markers CD133, CD44, CD24: association with AKT isoforms and radiation resistance in colon cancer cells.
Sahlberg SH, Spiegelberg D, Glimelius B, Stenerlöw B, Nestor M
PloS one 2014;9(4):e94621
PloS one 2014;9(4):e94621
Comparative evaluation of differentiation potential of menstrual blood- versus bone marrow-derived stem cells into hepatocyte-like cells.
Khanjani S, Khanmohammadi M, Zarnani AH, Akhondi MM, Ahani A, Ghaempanah Z, Naderi MM, Eghtesad S, Kazemnejad S
PloS one 2014;9(2):e86075
PloS one 2014;9(2):e86075
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of normal human peripheral blood cells with Anti-Human CD45 FITC (Product # 11-9459-42) and Mouse IgG1 K Isotype Control PerCP-eFluor® 710 (Product # 46-4714-82) (left) or Anti-Human CD133 PerCP-eFluor® 710 (right). Total viable cells were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
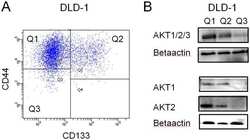
- Experimental details
- Figure 3 AKT expression in CD133/CD44 sorted cells. A) DLD-1 cells were sorted by flow cytometry and different populations with CD44 positive /CD133 negative (Q1), CD44 positive /CD133 positive (Q2), CD44 negative CD133 negative (Q3), were collected. B) The sorted cells were further analyzed with western blot for total AKT, AKT1 or AKT2 and betaactin expression.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
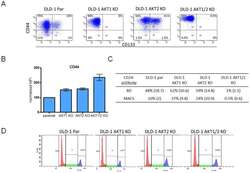
- Experimental details
- Figure 4 Flow cytometry analysis of the expression of CD133, CD24 and CD44 in the colon cancer cell-line DLD-1 with its isogenic knock-out cell-lines of AKT 1, AKT 2 and AKT 1/2. A) In the parental cells, approximately 10% of the cells were CD133 positive cells. However, in the AKT 1 and AKT 1/2 knock-outs, the CD133 positive cells were reduced to 0.3 and 0.1% respectively. This was not seen in the AKT 2 knock-out cell-line, where 33% of the cells were positive for CD133. B) The mean fluorescent intensity of CD44 normalized to the DLD-1 parental cell-line increased to 150% in AKT 1 KO, 160% in AKT 2 KO and 300% in AKT 1/2 KO cell-line. The error bars represent the standard deviation (SD) from at least two experiments. C) The percent of CD24 positive cells analyzed with two different CD24 antibodies from BD Biosciences and Miltenyi/MACS in flow cytometry. The standard deviations are from repeated experiments. D) Cell-cycle distribution in DLD-1 parental, AKT 1 KO, AKT 2 KO and AKT 1/2 KO cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 Identification of EPCs. a Adherent cells grew in a blood island manner. Fluorescent staining of EPCs. b Adherent cells took up UEA-1-lectin. c Adherent cells took up Dil-Ac-LDL. d Adherent cells took up UEA-1-lectin and Dil-Ac-LDL. E. Surface molecular markers of EPCs. Adherent cells expressed CD34, CD133, CD14 and VEGFR-2. All experiments involving cell culture studies were repeated three times with three replicates per experiment
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
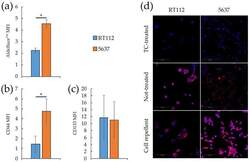
- Experimental details
- Figure 2 Stemness markers of monolayers and spheroids from RT112 and 5637 cells. ( a - c ) Median fluorescence intensity of Aldefluor TM ( a ), CD44 ( b ) and CD133 ( c ) by flow cytometry analysis on RT112 and 5637 cells grown as monolayers. Results are the mean of two ( a ) and three ( b , c ) experimental replicates. Statistical test: t -test, * for p < 0.05. ( d ) Representative images from confocal immunofluorescence (IF) microscopy of RT112 and 5637 cells using SOX2 Antibody (red) and Hoechst 33342 (blue) for nuclei. Cells were grown as monolayer or spheroids on different (Tissue culture-treated, Not-treated or Cell repellent) supports, before being seeded in adherent condition on chamber slides for IF.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
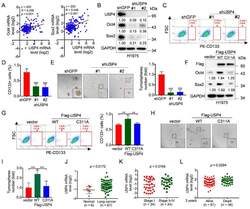
- Experimental details
- Figure 1 USP4 promotes lung cancer cell stemness and its high expression is correlated with human lung cancer patients. ( A ) The Oncomine dataset ""Bhattacharjee Lung"" was used to analyze Pearson correlation of USP4 and Oct4/Sox2 expression. ( B - E ) H1975 cells stably expressing shRNA against USP4 (shUSP4-#1 or shUSP4-#2) were subjected to ( B ) Western blot analyses, ( C - D ) FACS analyses for CD133-stained cells or ( E ) tumorsphere formation assay. Respective images and quantitation were shown. Data from three independent experiments in triplicates were presented as means +- SD. *** p < 0.001. Scale bar = 100 mum. ( F - I ) H1975 cells stably expressing Flag-USP4 or Flag-USP4 C311A were subjected to ( F ) Western blot analyses, ( G ) FACS analyses for CD133-stained cells or ( H - I ) tumorsphere formation assay. Respective images and quantitation were shown. Data from three independent experiments in duplicates were presented as means +- SD. ** p < 0.01, *** p < 0.001. Scale bar = 100 mum. ( J ) The Oncomine dataset ""Gaber lung"" was used to analyze USP4 mRNA levels in normal human lung tissues and lung cancers. ( K ) The Oncomine dataset ""Bild lung"" was used to analyze USP4 mRNA levels in stage I or stage II-IV human lung cancers. ( L ) The Oncomine dataset ""Raponi lung"" was used to analyze USP4 mRNA levels in 3 year-alive or 3 year-dead human lung cancer patients.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
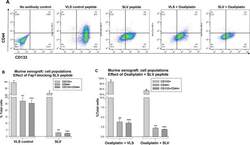
- Figure 5 Fap1-inhibition decreases abundance of CD133 + CD44 + cells in in a murine xenograft model of colon cancer with or without oxaliplatin Tumors from the mice described above were analyzed for cell population distribution after various treatments. (A) Histograms from flow cytometry demonstrate decreased abundance of CD133 + CD44 + cells after treatment with SLV peptide with or without oxaliplatin. A representative histograms for each cohort is shown. (B) Treatment with SLV peptide decreases relative abundance of CD133 + CD44 + cells in xenograft tumors. Tumors were simultaneously harvested from mice treated with SLV peptide versus VLS control (when control group tumors were >2,000 mm 3 ) and analyzed for CD133 and CD44 expression by flow cytometry. Significant differences indicated by * , ** , or *** . (p2,000 mm 3 ) and analyzed for CD133 and CD44 expression by flow cytometry. Significant differences indicated by * , ** , or *** . (p
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry