17-7222-80
antibody from Invitrogen Antibodies
Targeting: IL22
IL-21, IL-22, IL-D110, IL-TIF, ILTIF, MGC79382, MGC79384, TIFa, TIFIL-23, zcyto18
Antibody data
- Antibody Data
- Antigen structure
- References [42]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [17]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 17-7222-80 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IL-22 Monoclonal Antibody (IL22JOP), APC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The monoclonal antibody IL22JOP reacts with and inhibits the bioactivity of human and mouse IL-22. IL-22 is a 20 kDa member of the IL-10 cytokine family that is secreted primarily by Th17 cells, NK cells, and other T cells. Compared to IL-6 or TGF beta, IL-23 can induce greater levels of IL-22 in in vitro-differentiated Th17 cells. This observation suggests that IL-22 may be secreted by more fully differentiated Th17 cells in vivo. Recently, it was demonstrated that IL-22 could protect hosts from bacterial infection of the lungs and gut. Moreover, it has been reported that anti-CD3/CD28-induced production of IL-22 by PBMCs was elevated significantly in asthma patients compared to control patients. Flow cytometric analysis also showed that the frequencies of IL-17+IL-22+ CD4 T cells were increased in PBMCs from patients with ankylosing spondylitis and rheumatoid arthritis. IL22JOP is published to recognize rhesus IL-22. Applications Reported: This IL22JOP antibody has been reported for use in intracellular staining followed by flow cytometric analysis. Applications Tested: This IL22JOP antibody has been tested on Th17-polarized CD4+ normal human peripheral blood cells. This can be used at less than or equal to 0.25 µg per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. Excitation: 633-647 nm; Emission: 660 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human, Mouse
- Host
- Rat
- Isotype
- IgG
- Antibody clone number
- IL22JOP
- Vial size
- 25 µg
- Concentration
- 0.2 mg/mL
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Group 3 innate lymphocytes make a distinct contribution to type 17 immunity in bladder defence.
Interleukin-1β secretion induced by mucosa-associated gut commensal bacteria promotes intestinal barrier repair.
The transcription factor HIF-1α mediates plasticity of NKp46+ innate lymphoid cells in the gut.
ILC3s control airway inflammation by limiting T cell responses to allergens and microbes.
Prolonged residence of an albumin-IL-4 fusion protein in secondary lymphoid organs ameliorates experimental autoimmune encephalomyelitis.
IL-21 and IFNα therapy rescues terminally differentiated NK cells and limits SIV reservoir in ART-treated macaques.
Mitochondrial transcription factor A in RORγt(+) lymphocytes regulate small intestine homeostasis and metabolism.
Baricitinib treatment resolves lower-airway macrophage inflammation and neutrophil recruitment in SARS-CoV-2-infected rhesus macaques.
Redundant cytokine requirement for intestinal microbiota-induced Th17 cell differentiation in draining lymph nodes.
A Central Role for Lipocalin-2 in the Adaptation to Short-Bowel Syndrome Through Down-Regulation of IL22 in Mice.
Baricitinib treatment resolves lower airway inflammation and neutrophil recruitment in SARS-CoV-2-infected rhesus macaques.
GM-CSF Calibrates Macrophage Defense and Wound Healing Programs during Intestinal Infection and Inflammation.
Single-cell multi-omics analysis presents the landscape of peripheral blood T-cell subsets in human chronic prostatitis/chronic pelvic pain syndrome.
Host immunology and rational immunotherapy for carbapenem-resistant Klebsiella pneumoniae infection.
Characterization of Transcriptional Regulatory Networks that Promote and Restrict Identities and Functions of Intestinal Innate Lymphoid Cells.
Cell-Type-Specific Responses to Interleukin-1 Control Microbial Invasion and Tumor-Elicited Inflammation in Colorectal Cancer.
ATF3 Sustains IL-22-Induced STAT3 Phosphorylation to Maintain Mucosal Immunity Through Inhibiting Phosphatases.
CXCL4 is a novel inducer of human Th17 cells and correlates with IL-17 and IL-22 in psoriatic arthritis.
Oxysterol Sensing through the Receptor GPR183 Promotes the Lymphoid-Tissue-Inducing Function of Innate Lymphoid Cells and Colonic Inflammation.
Inhibition of p38 MAPK in combination with ART reduces SIV-induced immune activation and provides additional protection from immune system deterioration.
The Environmental Sensor AHR Protects from Inflammatory Damage by Maintaining Intestinal Stem Cell Homeostasis and Barrier Integrity.
Fecal Microbiota Transplantation, Commensal Escherichia coli and Lactobacillus johnsonii Strains Differentially Restore Intestinal and Systemic Adaptive Immune Cell Populations Following Broad-spectrum Antibiotic Treatment.
Bacillus anthracis lethal toxin negatively modulates ILC3 function through perturbation of IL-23-mediated MAPK signaling.
IL-21 and probiotic therapy improve Th17 frequencies, microbial translocation, and microbiome in ARV-treated, SIV-infected macaques.
Interleukin-22-Induced Antimicrobial Phospholipase A2 Group IIA Mediates Protective Innate Immunity of Nonhematopoietic Cells against Listeria monocytogenes.
Loss of Function of Intestinal IL-17 and IL-22 Producing Cells Contributes to Inflammation and Viral Persistence in SIV-Infected Rhesus Macaques.
Lymphocyte Isolation, Th17 Cell Differentiation, Activation, and Staining.
CD69 controls the uptake of L-tryptophan through LAT1-CD98 and AhR-dependent secretion of IL-22 in psoriasis.
Innate Lymphocyte/Ly6C(hi) Monocyte Crosstalk Promotes Klebsiella Pneumoniae Clearance.
Antibiotics in neonatal life increase murine susceptibility to experimental psoriasis.
The existence of Th22, pure Th17 and Th1 cells in CIN and Cervical Cancer along with their frequency variation in different stages of cervical cancer.
Expression of GM-CSF in T Cells Is Increased in Multiple Sclerosis and Suppressed by IFN-β Therapy.
Interleukin-21 combined with ART reduces inflammation and viral reservoir in SIV-infected macaques.
Interleukin-22 promotes T helper 1 (Th1)/Th17 immunity in chlamydial lung infection.
OMIP-022: Comprehensive assessment of antigen-specific human T-cell functionality and memory.
CX₃CR1⁺ mononuclear phagocytes support colitis-associated innate lymphoid cell production of IL-22.
Loss of mucosal CD103+ DCs and IL-17+ and IL-22+ lymphocytes is associated with mucosal damage in SIV infection.
Interleukin 23 production by intestinal CD103(+)CD11b(+) dendritic cells in response to bacterial flagellin enhances mucosal innate immune defense.
Th17 cytokines stimulate CCL20 expression in keratinocytes in vitro and in vivo: implications for psoriasis pathogenesis.
IL-22: a critical mediator in mucosal host defense.
Induction and effector functions of T(H)17 cells.
Inpatient capillary glucose monitoring: a useful adjunct to the management of diabetes in community hospitals.
Riding AM, Loudon KW, Guo A, Ferdinand JR, Lok LSC, Richoz N, Stewart A, Castro-Dopico T, Tuong ZK, Fiancette R, Bowyer GS, Fleming A, Gillman ES, Suchanek O, Mahbubani KT, Saeb-Parsy K, Withers D, Dougan G, Clare S, Clatworthy MR
iScience 2022 Jul 15;25(7):104660
iScience 2022 Jul 15;25(7):104660
Interleukin-1β secretion induced by mucosa-associated gut commensal bacteria promotes intestinal barrier repair.
Wu WH, Kim M, Chang LC, Assie A, Saldana-Morales FB, Zegarra-Ruiz DF, Norwood K, Samuel BS, Diehl GE
Gut microbes 2022 Jan-Dec;14(1):2014772
Gut microbes 2022 Jan-Dec;14(1):2014772
The transcription factor HIF-1α mediates plasticity of NKp46+ innate lymphoid cells in the gut.
Krzywinska E, Sobecki M, Nagarajan S, Zacharjasz J, Tambuwala MM, Pelletier A, Cummins E, Gotthardt D, Fandrey J, Kerdiles YM, Peyssonnaux C, Taylor CT, Sexl V, Stockmann C
The Journal of experimental medicine 2022 Feb 7;219(2)
The Journal of experimental medicine 2022 Feb 7;219(2)
ILC3s control airway inflammation by limiting T cell responses to allergens and microbes.
Teng F, Tachó-Piñot R, Sung B, Farber DL, Worgall S, Hammad H, Lambrecht BN, Hepworth MR, Sonnenberg GF
Cell reports 2021 Nov 23;37(8):110051
Cell reports 2021 Nov 23;37(8):110051
Prolonged residence of an albumin-IL-4 fusion protein in secondary lymphoid organs ameliorates experimental autoimmune encephalomyelitis.
Ishihara A, Ishihara J, Watkins EA, Tremain AC, Nguyen M, Solanki A, Katsumata K, Mansurov A, Budina E, Alpar AT, Hosseinchi P, Maillat L, Reda JW, Kageyama T, Swartz MA, Yuba E, Hubbell JA
Nature biomedical engineering 2021 May;5(5):387-398
Nature biomedical engineering 2021 May;5(5):387-398
IL-21 and IFNα therapy rescues terminally differentiated NK cells and limits SIV reservoir in ART-treated macaques.
Harper J, Huot N, Micci L, Tharp G, King C, Rascle P, Shenvi N, Wang H, Galardi C, Upadhyay AA, Villinger F, Lifson J, Silvestri G, Easley K, Jacquelin B, Bosinger S, Müller-Trutwin M, Paiardini M
Nature communications 2021 May 17;12(1):2866
Nature communications 2021 May 17;12(1):2866
Mitochondrial transcription factor A in RORγt(+) lymphocytes regulate small intestine homeostasis and metabolism.
Fu Z, Dean JW, Xiong L, Dougherty MW, Oliff KN, Chen ZE, Jobin C, Garrett TJ, Zhou L
Nature communications 2021 Jul 22;12(1):4462
Nature communications 2021 Jul 22;12(1):4462
Baricitinib treatment resolves lower-airway macrophage inflammation and neutrophil recruitment in SARS-CoV-2-infected rhesus macaques.
Hoang TN, Pino M, Boddapati AK, Viox EG, Starke CE, Upadhyay AA, Gumber S, Nekorchuk M, Busman-Sahay K, Strongin Z, Harper JL, Tharp GK, Pellegrini KL, Kirejczyk S, Zandi K, Tao S, Horton TR, Beagle EN, Mahar EA, Lee MYH, Cohen J, Jean SM, Wood JS, Connor-Stroud F, Stammen RL, Delmas OM, Wang S, Cooney KA, Sayegh MN, Wang L, Filev PD, Weiskopf D, Silvestri G, Waggoner J, Piantadosi A, Kasturi SP, Al-Shakhshir H, Ribeiro SP, Sekaly RP, Levit RD, Estes JD, Vanderford TH, Schinazi RF, Bosinger SE, Paiardini M
Cell 2021 Jan 21;184(2):460-475.e21
Cell 2021 Jan 21;184(2):460-475.e21
Redundant cytokine requirement for intestinal microbiota-induced Th17 cell differentiation in draining lymph nodes.
Sano T, Kageyama T, Fang V, Kedmi R, Martinez CS, Talbot J, Chen A, Cabrera I, Gorshko O, Kurakake R, Yang Y, Ng C, Schwab SR, Littman DR
Cell reports 2021 Aug 24;36(8):109608
Cell reports 2021 Aug 24;36(8):109608
A Central Role for Lipocalin-2 in the Adaptation to Short-Bowel Syndrome Through Down-Regulation of IL22 in Mice.
Zhang A, Sodhi CP, Wang M, Shores DR, Fulton W, Prindle T, Brosten S, O'Hare E, Lau A, Ding H, Jia H, Lu P, White JR, Hui J, Sears CL, Hackam DJ, Alaish SM
Cellular and molecular gastroenterology and hepatology 2020;10(2):309-326
Cellular and molecular gastroenterology and hepatology 2020;10(2):309-326
Baricitinib treatment resolves lower airway inflammation and neutrophil recruitment in SARS-CoV-2-infected rhesus macaques.
Hoang TN, Pino M, Boddapati AK, Viox EG, Starke CE, Upadhyay AA, Gumber S, Busman-Sahay K, Strongin Z, Harper JL, Tharp GK, Pellegrini KL, Kirejczyk S, Zandi K, Tao S, Horton TR, Beagle EN, Mahar EA, Lee MY, Cohen J, Jean SM, Wood JS, Connor-Stroud F, Stammen RL, Delmas OM, Wang S, Cooney KA, Sayegh MN, Wang L, Weiskopf D, Filev PD, Waggoner J, Piantadosi A, Kasturi SP, Al-Shakhshir H, Ribeiro SP, Sekaly RP, Levit RD, Estes JD, Vanderford TH, Schinazi RF, Bosinger SE, Paiardini M
bioRxiv : the preprint server for biology 2020 Sep 16;
bioRxiv : the preprint server for biology 2020 Sep 16;
GM-CSF Calibrates Macrophage Defense and Wound Healing Programs during Intestinal Infection and Inflammation.
Castro-Dopico T, Fleming A, Dennison TW, Ferdinand JR, Harcourt K, Stewart BJ, Cader Z, Tuong ZK, Jing C, Lok LSC, Mathews RJ, Portet A, Kaser A, Clare S, Clatworthy MR
Cell reports 2020 Jul 7;32(1):107857
Cell reports 2020 Jul 7;32(1):107857
Single-cell multi-omics analysis presents the landscape of peripheral blood T-cell subsets in human chronic prostatitis/chronic pelvic pain syndrome.
Zhang M, Liu Y, Chen J, Chen L, Meng J, Yang C, Yin S, Zhang X, Zhang L, Hao Z, Chen X, Liang C
Journal of cellular and molecular medicine 2020 Dec;24(23):14099-14109
Journal of cellular and molecular medicine 2020 Dec;24(23):14099-14109
Host immunology and rational immunotherapy for carbapenem-resistant Klebsiella pneumoniae infection.
Iwanaga N, Sandquist I, Wanek A, McCombs J, Song K, Kolls JK
JCI insight 2020 Apr 23;5(8)
JCI insight 2020 Apr 23;5(8)
Characterization of Transcriptional Regulatory Networks that Promote and Restrict Identities and Functions of Intestinal Innate Lymphoid Cells.
Pokrovskii M, Hall JA, Ochayon DE, Yi R, Chaimowitz NS, Seelamneni H, Carriero N, Watters A, Waggoner SN, Littman DR, Bonneau R, Miraldi ER
Immunity 2019 Jul 16;51(1):185-197.e6
Immunity 2019 Jul 16;51(1):185-197.e6
Cell-Type-Specific Responses to Interleukin-1 Control Microbial Invasion and Tumor-Elicited Inflammation in Colorectal Cancer.
Dmitrieva-Posocco O, Dzutsev A, Posocco DF, Hou V, Yuan W, Thovarai V, Mufazalov IA, Gunzer M, Shilovskiy IP, Khaitov MR, Trinchieri G, Waisman A, Grivennikov SI
Immunity 2019 Jan 15;50(1):166-180.e7
Immunity 2019 Jan 15;50(1):166-180.e7
ATF3 Sustains IL-22-Induced STAT3 Phosphorylation to Maintain Mucosal Immunity Through Inhibiting Phosphatases.
Glal D, Sudhakar JN, Lu HH, Liu MC, Chiang HY, Liu YC, Cheng CF, Shui JW
Frontiers in immunology 2018;9:2522
Frontiers in immunology 2018;9:2522
CXCL4 is a novel inducer of human Th17 cells and correlates with IL-17 and IL-22 in psoriatic arthritis.
Affandi AJ, Silva-Cardoso SC, Garcia S, Leijten EFA, van Kempen TS, Marut W, van Roon JAG, Radstake TRDJ
European journal of immunology 2018 Mar;48(3):522-531
European journal of immunology 2018 Mar;48(3):522-531
Oxysterol Sensing through the Receptor GPR183 Promotes the Lymphoid-Tissue-Inducing Function of Innate Lymphoid Cells and Colonic Inflammation.
Emgård J, Kammoun H, García-Cassani B, Chesné J, Parigi SM, Jacob JM, Cheng HW, Evren E, Das S, Czarnewski P, Sleiers N, Melo-Gonzalez F, Kvedaraite E, Svensson M, Scandella E, Hepworth MR, Huber S, Ludewig B, Peduto L, Villablanca EJ, Veiga-Fernandes H, Pereira JP, Flavell RA, Willinger T
Immunity 2018 Jan 16;48(1):120-132.e8
Immunity 2018 Jan 16;48(1):120-132.e8
Inhibition of p38 MAPK in combination with ART reduces SIV-induced immune activation and provides additional protection from immune system deterioration.
Chaudhary O, Narayan V, Lelis F, Linz B, Watkins M, Veazey R, Aldovini A
PLoS pathogens 2018 Aug;14(8):e1007268
PLoS pathogens 2018 Aug;14(8):e1007268
The Environmental Sensor AHR Protects from Inflammatory Damage by Maintaining Intestinal Stem Cell Homeostasis and Barrier Integrity.
Metidji A, Omenetti S, Crotta S, Li Y, Nye E, Ross E, Li V, Maradana MR, Schiering C, Stockinger B
Immunity 2018 Aug 21;49(2):353-362.e5
Immunity 2018 Aug 21;49(2):353-362.e5
Fecal Microbiota Transplantation, Commensal Escherichia coli and Lactobacillus johnsonii Strains Differentially Restore Intestinal and Systemic Adaptive Immune Cell Populations Following Broad-spectrum Antibiotic Treatment.
Ekmekciu I, von Klitzing E, Neumann C, Bacher P, Scheffold A, Bereswill S, Heimesaat MM
Frontiers in microbiology 2017;8:2430
Frontiers in microbiology 2017;8:2430
Bacillus anthracis lethal toxin negatively modulates ILC3 function through perturbation of IL-23-mediated MAPK signaling.
Seshadri S, Allan DSJ, Carlyle JR, Zenewicz LA
PLoS pathogens 2017 Oct;13(10):e1006690
PLoS pathogens 2017 Oct;13(10):e1006690
IL-21 and probiotic therapy improve Th17 frequencies, microbial translocation, and microbiome in ARV-treated, SIV-infected macaques.
Ortiz AM, Klase ZA, DiNapoli SR, Vujkovic-Cvijin I, Carmack K, Perkins MR, Calantone N, Vinton CL, Riddick NE, Gallagher J, Klatt NR, McCune JM, Estes JD, Paiardini M, Brenchley JM
Mucosal immunology 2016 Mar;9(2):458-67
Mucosal immunology 2016 Mar;9(2):458-67
Interleukin-22-Induced Antimicrobial Phospholipase A2 Group IIA Mediates Protective Innate Immunity of Nonhematopoietic Cells against Listeria monocytogenes.
Okita Y, Shiono T, Yahagi A, Hamada S, Umemura M, Matsuzaki G
Infection and immunity 2016 Feb;84(2):573-9
Infection and immunity 2016 Feb;84(2):573-9
Loss of Function of Intestinal IL-17 and IL-22 Producing Cells Contributes to Inflammation and Viral Persistence in SIV-Infected Rhesus Macaques.
Ryan ES, Micci L, Fromentin R, Paganini S, McGary CS, Easley K, Chomont N, Paiardini M
PLoS pathogens 2016 Feb;12(2):e1005412
PLoS pathogens 2016 Feb;12(2):e1005412
Lymphocyte Isolation, Th17 Cell Differentiation, Activation, and Staining.
Kumar P, Kolls JK
Bio-protocol 2016 Dec 5;6(23)
Bio-protocol 2016 Dec 5;6(23)
CD69 controls the uptake of L-tryptophan through LAT1-CD98 and AhR-dependent secretion of IL-22 in psoriasis.
Cibrian D, Saiz ML, de la Fuente H, Sánchez-Díaz R, Moreno-Gonzalo O, Jorge I, Ferrarini A, Vázquez J, Punzón C, Fresno M, Vicente-Manzanares M, Daudén E, Fernández-Salguero PM, Martín P, Sánchez-Madrid F
Nature immunology 2016 Aug;17(8):985-96
Nature immunology 2016 Aug;17(8):985-96
Innate Lymphocyte/Ly6C(hi) Monocyte Crosstalk Promotes Klebsiella Pneumoniae Clearance.
Xiong H, Keith JW, Samilo DW, Carter RA, Leiner IM, Pamer EG
Cell 2016 Apr 21;165(3):679-89
Cell 2016 Apr 21;165(3):679-89
Antibiotics in neonatal life increase murine susceptibility to experimental psoriasis.
Zanvit P, Konkel JE, Jiao X, Kasagi S, Zhang D, Wu R, Chia C, Ajami NJ, Smith DP, Petrosino JF, Abbatiello B, Nakatsukasa H, Chen Q, Belkaid Y, Chen ZJ, Chen W
Nature communications 2015 Sep 29;6:8424
Nature communications 2015 Sep 29;6:8424
The existence of Th22, pure Th17 and Th1 cells in CIN and Cervical Cancer along with their frequency variation in different stages of cervical cancer.
Zhang W, Tian X, Mumtahana F, Jiao J, Zhang T, Croce KD, Ma D, Kong B, Cui B
BMC cancer 2015 Oct 16;15:717
BMC cancer 2015 Oct 16;15:717
Expression of GM-CSF in T Cells Is Increased in Multiple Sclerosis and Suppressed by IFN-β Therapy.
Rasouli J, Ciric B, Imitola J, Gonnella P, Hwang D, Mahajan K, Mari ER, Safavi F, Leist TP, Zhang GX, Rostami A
Journal of immunology (Baltimore, Md. : 1950) 2015 Jun 1;194(11):5085-93
Journal of immunology (Baltimore, Md. : 1950) 2015 Jun 1;194(11):5085-93
Interleukin-21 combined with ART reduces inflammation and viral reservoir in SIV-infected macaques.
Micci L, Ryan ES, Fromentin R, Bosinger SE, Harper JL, He T, Paganini S, Easley KA, Chahroudi A, Benne C, Gumber S, McGary CS, Rogers KA, Deleage C, Lucero C, Byrareddy SN, Apetrei C, Estes JD, Lifson JD, Piatak M Jr, Chomont N, Villinger F, Silvestri G, Brenchley JM, Paiardini M
The Journal of clinical investigation 2015 Dec;125(12):4497-513
The Journal of clinical investigation 2015 Dec;125(12):4497-513
Interleukin-22 promotes T helper 1 (Th1)/Th17 immunity in chlamydial lung infection.
Peng Y, Gao X, Yang J, Shekhar S, Wang S, Fan Y, Zhao W, Yang X
Molecular medicine (Cambridge, Mass.) 2014 Mar 20;20(1):109-19
Molecular medicine (Cambridge, Mass.) 2014 Mar 20;20(1):109-19
OMIP-022: Comprehensive assessment of antigen-specific human T-cell functionality and memory.
Graves AJ, Padilla MG, Hokey DA
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2014 Jul;85(7):576-9
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2014 Jul;85(7):576-9
CX₃CR1⁺ mononuclear phagocytes support colitis-associated innate lymphoid cell production of IL-22.
Longman RS, Diehl GE, Victorio DA, Huh JR, Galan C, Miraldi ER, Swaminath A, Bonneau R, Scherl EJ, Littman DR
The Journal of experimental medicine 2014 Jul 28;211(8):1571-83
The Journal of experimental medicine 2014 Jul 28;211(8):1571-83
Loss of mucosal CD103+ DCs and IL-17+ and IL-22+ lymphocytes is associated with mucosal damage in SIV infection.
Klatt NR, Estes JD, Sun X, Ortiz AM, Barber JS, Harris LD, Cervasi B, Yokomizo LK, Pan L, Vinton CL, Tabb B, Canary LA, Dang Q, Hirsch VM, Alter G, Belkaid Y, Lifson JD, Silvestri G, Milner JD, Paiardini M, Haddad EK, Brenchley JM
Mucosal immunology 2012 Nov;5(6):646-57
Mucosal immunology 2012 Nov;5(6):646-57
Interleukin 23 production by intestinal CD103(+)CD11b(+) dendritic cells in response to bacterial flagellin enhances mucosal innate immune defense.
Kinnebrew MA, Buffie CG, Diehl GE, Zenewicz LA, Leiner I, Hohl TM, Flavell RA, Littman DR, Pamer EG
Immunity 2012 Feb 24;36(2):276-87
Immunity 2012 Feb 24;36(2):276-87
Th17 cytokines stimulate CCL20 expression in keratinocytes in vitro and in vivo: implications for psoriasis pathogenesis.
Harper EG, Guo C, Rizzo H, Lillis JV, Kurtz SE, Skorcheva I, Purdy D, Fitch E, Iordanov M, Blauvelt A
The Journal of investigative dermatology 2009 Sep;129(9):2175-83
The Journal of investigative dermatology 2009 Sep;129(9):2175-83
IL-22: a critical mediator in mucosal host defense.
Aujla SJ, Kolls JK
Journal of molecular medicine (Berlin, Germany) 2009 May;87(5):451-4
Journal of molecular medicine (Berlin, Germany) 2009 May;87(5):451-4
Induction and effector functions of T(H)17 cells.
Bettelli E, Korn T, Oukka M, Kuchroo VK
Nature 2008 Jun 19;453(7198):1051-7
Nature 2008 Jun 19;453(7198):1051-7
Inpatient capillary glucose monitoring: a useful adjunct to the management of diabetes in community hospitals.
Borst GC, Rice S, Robinson WL
The Journal of the Kentucky Medical Association 1989 Sep;87(9):451-5
The Journal of the Kentucky Medical Association 1989 Sep;87(9):451-5
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Intracellular staining of 7-day Th17-polarized CD4+ normal human peripheral blood cells (using Human IL-23 Recombinant Protein (Product # 14-8239-63) (left) and 12-day Th17-polarized mouse splenocytes (using Mouse IL-23 Recombinant Protein (Product # 14-8231-63) (right) with 0.125 µg of Anti-Human/Mouse IL-22 APC. Both sets of polarized cells were incubated with PMA, Ionomycin, and Brefeldin A for 5 hours prior to treatment with the Intracellular Fixation & Permeabilization Buffer Set (Product # 88-8824-00). Human cells were then costained with Anti-Human CD4 eFluor 450 (Product # 48-0049-42) and mouse cells were costained with Anti-Mouse CD4 FITC (Product # 11-0042-82). Quadrants were set based on Th17 cells treated with Brefeldin A alone and cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Gene profiling reveals tissue-specific transcriptomic signature of gammadeltaT17 cells. a Left panel, principal component analysis (PCA) plot of the RNA-seq of Tfam-sufficient splenic (Sp), small intestinal (SI), and large intestinal (LI) gammadeltaT17 cells from 3-week-old ( n = 3 for each group) mice. gammadeltaT17 cells of each sample were sorted from an individual mouse. Right panel, Euclidean distance comparison of the transcriptome of each splenic gammadeltaT17 cell sample and the mean intestine centroid vs. the mean small intestine centroid to each large intestinal gammadeltaT17 cell sample ( n = 3 for each group) (*** P = 0.0002). b Heatmap of tissue-specific genes (TSGs) identified by the RNA-seq in Sp, SI, and LI gammadeltaT17 cells. Genes were ranked in a descending order based on the fold changes of expression (Sp/[SI and LI average mean]; [SI and LI average mean]/Sp). c RNA-seq FPKM values of Ahr direct target genes of Sp, SI, and LI gammadeltaT17 cells (mean +- SD) ( n = 3 for each group) (Sp vs. SI: Ahr , *** P = 0.0005, Ahrr , ** P = 0.0037, Asb2 , *** P = 0.0004, Cd93 , **** P < 0.0001, Cd160 , *** P = 0.0002, Gem , ** P = 0.0096, Hic1 , *** P = 0.0002, Il17a , **** P < 0.0001, Il22 , * P = 0.0180, Kit , *** P = 0.0004, Nuerl3 , **** P < 0.0001, Osgin1 , *** P = 0.0002, Vps37b , **** P < 0.0001; Sp vs. LI: Ahr , * P = 0.0460, Ahrr , P = 0.0506, Asb2 , ** P = 0.0076, Cd93 , **** P < 0.0001, Cd160 , *** P = 0.0001, Gem , * P = 0.0108, Hic1 , *** P = 0.00
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
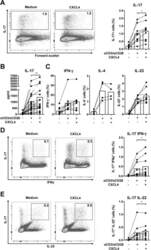
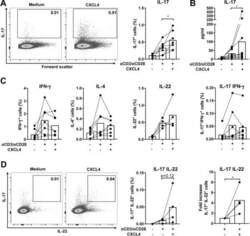
- Figure 1 CXCL4 increases the percentage of IL-17 producing cells in CD3/CD28-stimulated human CD4 + T cells. CD4 + T cells were isolated from healthy donors and cultured with CD3/CD28 coated Dynabeads and CXCL4 for five days. (A, B) The effect of CXCL4 on IL-17 production by CD4 + T cells was assessed by (A) flow cytometric intracellular cytokine staining and (B) enzyme-linked immunosorbent assay. (C) The percentage of of IFN-gamma-, IL-4- and IL-22-producing CD4 + T cells were measured by flow cytometry. (D, E) The amount of IL-17 producing cells co-expressing IFN-gamma (D) or IL-22 (E) were measured by flow cytometry. Cells were gated on live, single cells. Means (bars) and values from each donor are shown. Data are pooled from two to four independent experiments, except for panel B from 14 independent experiments, with one to four donor samples per experiment. Each dot on the bar graphs represent a single donor and paired t -test was used for statistical analysis. * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
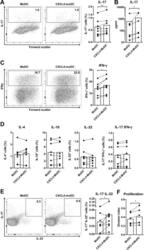
- Figure 2 CXCL4 induces IL-17 producing cells differentiated from naive human CD4 + T cells. Naive CD4 + T cells were purified by fluorescence-activated cell sorting and cultured with CD3/CD28 coated Dynabeads and CXCL4 for seven days. (A, B) The effect of CXCL4 on IL-17 production by CD4 + T cells was assessed by (A) flow cytometric intracellular cytokine staining and (B) enzyme-linked immunosorbent assay. (C) The levels of IFN-gamma-, IL-4-, IL-22-, and IL-17/IFN-gamma-producing CD4 + T cells were measured by flow cytometry. (D) The amount of IL-17 producing cells co-expressing or IL-22 was measured by flow cytometry. Cells were gated on live, single cells. Means (bars) and values from each donor are shown. Data are pooled from four to 10 independent experiments, with one to two donor samples per experiment. Each dot on the bar graphs represents a single donor and paired t -test was used for statistical analysis. * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 4 CXCL4-differentiated monocyte-derived dendritic cells enhance pro-inflammatory cytokine production and proliferation by CD4 + T cells. Monocytes from healthy donors were isolated and differentiated into dendritic cells in the absence or presence of CXCL4 (moDCs or CXCL4-moDCs). moDCs were then co-cultured with autologous CD4 + T cells in the presence of superantigen from Staphylococcal Enterotoxin B (SEB) for three days and restimulated with PMA and ionomycin. (A-C) Comparison of co-culture with moDC or CXCL4-moDC on IL-17 or IFN-gamma production by CD4 + T cells was assessed by (A, C) intracellular cytokine staining and (B) enzyme-linked immunosorbent assay are shown. (D) Intracellular cytokine staining was performed for the measurement of IL-4 + , IL-10 + , IL-22 + , and IL-17 + IFN-gamma + cells gated on live CD4 + T cells. (E) The amount of IL-17 producing cells co-expressing IL-22 as measured by flow cytometry. (F) CD4 + T cells were labeled with CellTrace Violet prior co-culture and proliferation was analyzed as division index. Cells were gated on live, single, CD4 + T cells. Means (bars) and values from each donor are shown. Data are pooled from two to three independent experiments, with two to three donor samples per experiment. Each dot on the bar graphs represent a single donor and paired t -test was used for statistical analysis. * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
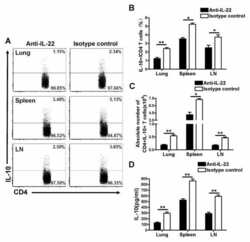
- Figure 9 LCN2 reduces intestinal adaptation by inhibiting IL22 expression after 75% SBR on POD 7 . ( A ) IL22 protein expression in the serum was measured by ELISA (n = 7 mice per group). ( B ) Transcriptional levels of the Il22 gene in the intestine were evaluated by quantitative PCR (n = 6 per group). ( C ) CD4 + IL22 + LPLs were quantified with flow cytometry analysis (n = 5 per group). ( D ) Representative IL22 protein expression in supernatant from Th22 cells in vitro with and without LCN2 stimulation as measured by ELISA (n = 3 per group/each experiment, each experiment was repeated 3 times). ( E-H ) Representative flow cytometry dot plots of the LPL population from small intestinal tissue. Single-cell suspensions prepared from the lamina propria were stained with CD4 and IL22 antibodies. Gate 1 identified lymphocytes based on FSC-A/SSC-A properties. The numbers within the quadrants represent the percentage of CD4 + IL22 - , CD4 + IL22 + , CD4 - IL22 - , and CD4 - IL22 + cells within the lymphocyte gate. Data are representative of analyses of 5 mice per group. Forward scatter area (FSC-A)/side scatter area (SSC-A). * P < .05, ** P < .01, and *** P < .001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
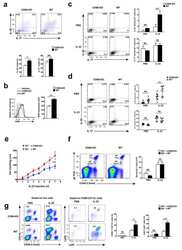
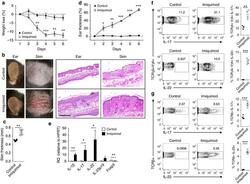
- Figure 1 Imiquimod increases IL-17 and IL-22 in both gammadelta and alphabeta T cells in skin. Skin and ear of wild type C57BL/6 mice were treated by topical application of imiquimod cream (Fougera, n =4) or control cream (Vaseline, n =3) for 6 consecutive days. ( a ) Weight loss of imiquimod and control cream treated mice monitored daily. ( b ) Photographs of imiquimod and control cream treated skin and ear; photos were taken at day 6. H&E stained ear and back skin sections of imiquimod treated and control mice. Scale bar, 200 mum. ( c ) Thickness of skin measured by Digimatic Caliper at day 6 in control and imiquimod treated mice. Data showed represents average of at least two measurements. ( d ) Ear thickness of imiquimod and control cream treated mice monitored daily. Ear thickness was measured using Digimatic Caliper. ( e ) Quantitative PCR analysis of Th17 associated cytokines and Foxp3 in skin after 6 days of imiquimod and control cream treatment. ( f ) Representative flow cytometric analysis of TCRgammadelta + Vgamma4 + IL-17 + (upper raw) and TCRgammadelta + Vgamma4 + IL-22 + cells (lower row) in the skin of control ( n =3) and imiquimod treated mice ( n =4). ( g ) Representative flow cytometric analysis of single-positive TCRbeta + IL-17 + (upper raw) or TCRbeta + IL-22 + (lower raw) cells in the skin of control ( n =3) and imiquimod treated mice ( n =4). Data representative of more than three experiments, results are shown as mean+-s.e.m., significance determined b
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
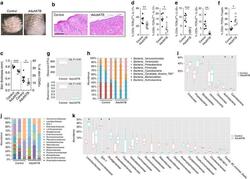
- Figure 2 Antibiotics in adult mice ameliorate psoriasis by reduction of IL-17 and IL-22 in T cells. Young adult mice (4-weeks-old) were exposed to antibiotics in drinking water (adultATB) for four weeks, and last six consecutive days followed by imiquimod treatment. ( a ) Representative photographs of back skin of control and adultATB mice followed by imiquimod treatment. Photos were taken at day 6. ( b ) H&E stained skin sections of control and adultATB mice after imiquimod treatment. Scale bar, 200 mum. ( c ) Thickness of skin measured by digimatic caliper at day 6 in control and imiquimod treated mice. Data showed represents average of at least two measurements. Increased skin infiltration in control compared adultATB mice based on staining of skin CD45 + cells. ( d ) Flow cytometric analysis of single-positive CD3 + TCRgammadelta low+ IL-17 + cells or CD3 + TCRgammadelta - IL-17 + cells in control ( n =6-7) and adultATB ( n =6-7) mice after imiquimod treatment. ( e ) Flow cytometric analysis of single-positive CD3 + TCRgammadelta low+ IL-22 + cells or CD3 + TCRgammadelta - IL-22 + cells in control ( n =6) and adultATB ( n =6) mice after imiquimod treatment. ( f ) Frequency of skin CD3 + TCRbeta + Foxp3 + cells in the skin of control ( n =5) and adultATB ( n =7) mice after imiquimod treatment. ( g ) Diversity of the microbiota (Observed OTUs, Shannon Index) in adultATB and control mice. Primary samples were collected per cage (Pooled samples from 3 independent experiments)
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
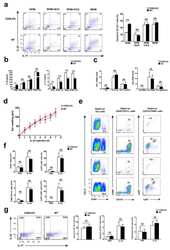
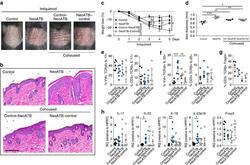
- Figure 3 Neonatal antibiotic treatment of mice increases severity of psoriasis. Breeding pairs were given antibiotics vancomycin+polymyxin B in drinking water immediately after pups delivery for 3 weeks of rearing (neoATB). After weaning, mice were housed or cohoused (mice without antibiotics treatment together with mice treated with antibiotics) in normal specific pathogen-free conditions for next 8 weeks. The mice were then treated with imiquimod cream for 4 consecutive days. ( a ) Representative photographs of back skin in controls (mice without antibiotic treatment); neonatally antibiotics treated mice (neoATB) or cohoused mice (control-neoATB resp. NeoATB-control) after imiquimod treatment. Photos were taken at day 5. ( b ) H&E stained skin sections of control, neoATB and cohoused mice after imiquimod treatment. Scale bar, 200 mum. ( c ) Weight loss of control, neoATB and cohoused mice during imiquimod treatment monitored daily. ( d ) Skin thickness of control, neoATB and cohoused mice measured at day 5. Skin thickness was measured by digimatic caliper by at least two investigators and data showed represents average of at least two measurements. ( e ) Flow cytometric analysis of single-positive TCRgammadelta + Vgamma4 + IL-17 + cells or CD3 + TCRbeta + IL-17 + cells in control ( n =8), neoATB ( n =7) and cohoused mice ( n =5) after imiquimod treatment. ( f ) Flow cytometric analysis of single-positive TCRgammadelta + Vgamma4 + IL-22 + cells or CD3 + TCRbeta + IL-22 + cel
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
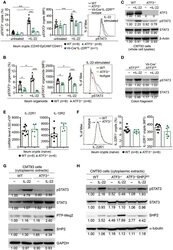
- Figure 7 ATF3 promotes IL-22-induced STAT3 phosphorylation by suppressing phosphatases. (A) Freshly isolated ileum crypts, or (B) ileum organoids at day 6 of culture, were stimulated with IL-22, followed by fixation and intracellular staining of phospho-STAT3, and analyzed by flow cytometry. Western blot analysis of (C) IL-22-stimulated CMT93 cells, or (D) IL-22-stimuated colon fragments isolated from the indicated mice, for the expression of the indicated proteins. (E) Quantitative real-time PCR analysis of IL-22R1 and IL-10R2 mRNA levels in freshly isolated ileum crypts from mice. (F) Flow cytometry analysis of IL-22R1 in freshly isolated ileum crypt cells gated on the CD45 - EpCAM + population. (G,H) Western blot analysis of unstimulated or IL-22-stimulated CMT93 cells for the indicated proteins. ATF3 -/- CMT93 cells with SHP2 knockdown (ATF3 -/- SHP2 KD ) were indicated. Images were representative of four independent experiments (G-H) . Results were from two independent experiments (A-F) . ""n"" refers to the number of mice analyzed (A,B,E,F) . Statistical analysis was done by multiple comparison in Two-way ANOVA test using Prism software. * P < 0.05, ** P < 0.005, *** P < 0.0005.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
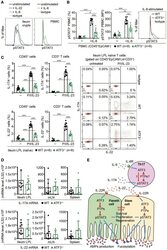
- Figure 8 ATF3 regulates IL-6-pSTAT3 signaling in intestinal Th17 cells. Flow cytometry analysis of phospho-STAT3 in (A) IL-6 or IL-22 stimulated freshly isolated ileum crypts or IL-6-stimulated peripheral blood mononuclear cell (PBMC) from wild-type mice, or in (B) IL-6-stimulated PBMC from wild-type and ATF3-deficient mice. (C) Flow cytometry analysis of intracellular IL-17A and IL-22 expression in naive lamina propria T cells from the indicated mice. Cells were treated with PMA, ionomycin and IL-23 in the presence of BFA for 4 h before analysis and gated on live CD45 + EpCAM - Lin - CD3 + population as shown. (D) Quantitative real-time PCR analysis of IL-17A and IL-22 mRNA levels in freshly isolated lamina propria (LPL) cells, mesenteric lymph nodes (mLN), or splenocytes. (E) Model of ATF3-mediated mucosal immunity via cross-regulation between IL-22-pSTAT3 signaling in epithelium (associated with AMP production and epithelial fucosylation) and IL-6-pSTAT3 signaling in Th17 cells (associated with signature IL-17A and IL-22 production). ""n"" refers to the number of mice analyzed. Statistical analysis was done by multiple comparison in Two-way ANOVA test using Prism software. * P < 0.05, ** P < 0.005, *** P < 0.0005.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
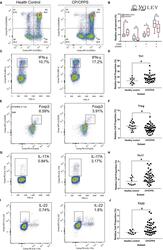
- 5 FIGURE Flow cytometry revealed central memory T cell, Th1, Th17, Th22 and Treg proportions increased in PBMCs derived from CP/CPPS patients than healthy controls. PBMCs were incubated with various fluorescein-labelled antigens for surface staining. PerCP/Cyanine5.5-conjugated CD3 and FITC-conjugated CD4 were used for the central memory T cell, Th1, Th17 and Th22 cell staining, and PE-conjugated CD25 and FITC-conjugated CD4 were incubated for staining Treg cells. After fixing and permeabilizing with cell fixation/permeabilization kit, (A) for samples staining central memory T cells were incubated with PE-conjugated CD45RA and APC-conjugated CD62L; (C) for samples staining, Th1 cells were incubated with PE-conjugated IFN-gamma; (E) for samples staining, Treg cells were incubated with eFluor 660-conjugated FoxP3; (G) for samples staining, Th17 cells were incubated with PE-conjugated IL-17A; and (I) for samples staining, Th22 cells were incubated with PE-conjugated IL-17A and APC-conjugated IL-22. The quantification data were presented in B, D, F, H and J. * P < 0.05; PBMC, peripheral blood mononuclear cell
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry