25-7229-42
antibody from Invitrogen Antibodies
Targeting: IL22
IL-21, IL-22, IL-D110, IL-TIF, ILTIF, MGC79382, MGC79384, TIFa, TIFIL-23, zcyto18
Antibody data
- Antibody Data
- Antigen structure
- References [20]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [10]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 25-7229-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IL-22 Monoclonal Antibody (22URTI), PE-Cyanine7, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 22URTI monoclonal antibody reacts with human interleukin(IL)-22. IL-22 is a 20 kDa member of the IL-10 cytokine family that is secreted primarily by Th17 and NK cells. Nevertheless, other T cells have also been shown to produce IL-22. In in vitro Th17 cultures, induction of IL-22 expression is greater in response to IL-23 than IL-6 or TGF beta, suggesting that this cytokine may be secreted by more fully differentiated Th17 cells in vivo. A heterodimer consisting of IL-10R2 and IL-22R1 serves as the receptor for IL-22.
- Antibody clone number
- 22URTI
- Concentration
- 5 µL/Test
Submitted references Decreased Frequencies of Gamma/Delta T Cells Expressing Th1/Th17 Cytokine, Cytotoxic, and Immune Markers in Latent Tuberculosis-Diabetes/Pre-Diabetes Comorbidity.
Functional SARS-CoV-2-Specific Immune Memory Persists after Mild COVID-19.
TIRC7 inhibits Th1 cells by upregulating the expression of CTLA‑4 and STAT3 in mice with acute graft‑versus‑host disease.
HIV Skews a Balanced Mtb-Specific Th17 Response in Latent Tuberculosis Subjects to a Pro-inflammatory Profile Independent of Viral Load.
Thymic stromal lymphopoietin epigenetically upregulates Fc receptor γ subunit-related receptors on antigen-presenting cells and induces T(H)2/T(H)17 polarization through dectin-2.
Accumulation of T-helper 22 cells, interleukin-22 and myeloid-derived suppressor cells promotes gastric cancer progression in elderly patients.
CD4 T-Cell Dysregulation in Psoriatic Arthritis Reveals a Regulatory Role for IL-22.
Human CD5(+) Innate Lymphoid Cells Are Functionally Immature and Their Development from CD34(+) Progenitor Cells Is Regulated by Id2.
Antigen-Presenting Human γδ T Cells Promote Intestinal CD4(+) T Cell Expression of IL-22 and Mucosal Release of Calprotectin.
Systemic Human ILC Precursors Provide a Substrate for Tissue ILC Differentiation.
Immune cell-derived cytokines contribute to obesity-related inflammation, fibrogenesis and metabolic deregulation in human adipose tissue.
Downregulation of RUNX3 moderates the frequency of Th17 and Th22 cells in patients with psoriasis.
Compartment-specific immunity in the human gut: properties and functions of dendritic cells in the colon versus the ileum.
Interleukin-25 Mediates Transcriptional Control of PD-L1 via STAT3 in Multipotent Human Mesenchymal Stromal Cells (hMSCs) to Suppress Th17 Responses.
Jejunal T Cell Inflammation in Human Obesity Correlates with Decreased Enterocyte Insulin Signaling.
Early initiation of combined antiretroviral therapy preserves immune function in the gut of HIV-infected patients.
IL32 is progressively expressed in mycosis fungoides independent of helper T-cell 2 and helper T-cell 9 polarization.
T cell-derived IL-22 amplifies IL-1β-driven inflammation in human adipose tissue: relevance to obesity and type 2 diabetes.
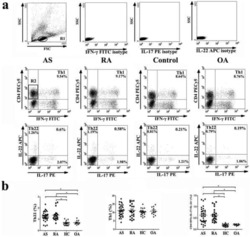
Increased frequencies of Th22 cells as well as Th17 cells in the peripheral blood of patients with ankylosing spondylitis and rheumatoid arthritis.
Human Th17 cells comprise heterogeneous subsets including IFN-gamma-producing cells with distinct properties from the Th1 lineage.
Kathamuthu GR, Kumar NP, Moideen K, Menon PA, Babu S
Frontiers in cellular and infection microbiology 2021;11:756854
Frontiers in cellular and infection microbiology 2021;11:756854
Functional SARS-CoV-2-Specific Immune Memory Persists after Mild COVID-19.
Rodda LB, Netland J, Shehata L, Pruner KB, Morawski PA, Thouvenel CD, Takehara KK, Eggenberger J, Hemann EA, Waterman HR, Fahning ML, Chen Y, Hale M, Rathe J, Stokes C, Wrenn S, Fiala B, Carter L, Hamerman JA, King NP, Gale M Jr, Campbell DJ, Rawlings DJ, Pepper M
Cell 2021 Jan 7;184(1):169-183.e17
Cell 2021 Jan 7;184(1):169-183.e17
TIRC7 inhibits Th1 cells by upregulating the expression of CTLA‑4 and STAT3 in mice with acute graft‑versus‑host disease.
Zhu F, Qiu T, Zhu S, Zhao K, Chen C, Qiao J, Pan B, Yan Z, Chen W, Liu Q, Wu Q, Cao J, Sang W, Zeng L, Sun H, Li Z, Xu K
Oncology reports 2020 Jul;44(1):43-54
Oncology reports 2020 Jul;44(1):43-54
HIV Skews a Balanced Mtb-Specific Th17 Response in Latent Tuberculosis Subjects to a Pro-inflammatory Profile Independent of Viral Load.
Rakshit S, Hingankar N, Alampalli SV, Adiga V, Sundararaj BK, Sahoo PN, Finak G, Uday Kumar J AJ, Dhar C, D'Souza G, Virkar RG, Ghate M, Thakar MR, Paranjape RS, De Rosa SC, Ottenhoff THM, Vyakarnam A
Cell reports 2020 Dec 1;33(9):108451
Cell reports 2020 Dec 1;33(9):108451
Thymic stromal lymphopoietin epigenetically upregulates Fc receptor γ subunit-related receptors on antigen-presenting cells and induces T(H)2/T(H)17 polarization through dectin-2.
Liang Y, Yu B, Chen J, Wu H, Xu Y, Yang B, Lu Q
The Journal of allergy and clinical immunology 2019 Oct;144(4):1025-1035.e7
The Journal of allergy and clinical immunology 2019 Oct;144(4):1025-1035.e7
Accumulation of T-helper 22 cells, interleukin-22 and myeloid-derived suppressor cells promotes gastric cancer progression in elderly patients.
Chen X, Wang Y, Wang J, Wen J, Jia X, Wang X, Zhang H
Oncology letters 2018 Jul;16(1):253-261
Oncology letters 2018 Jul;16(1):253-261
CD4 T-Cell Dysregulation in Psoriatic Arthritis Reveals a Regulatory Role for IL-22.
Ezeonyeji A, Baldwin H, Vukmanovic-Stejic M, Ehrenstein MR
Frontiers in immunology 2017;8:1403
Frontiers in immunology 2017;8:1403
Human CD5(+) Innate Lymphoid Cells Are Functionally Immature and Their Development from CD34(+) Progenitor Cells Is Regulated by Id2.
Nagasawa M, Germar K, Blom B, Spits H
Frontiers in immunology 2017;8:1047
Frontiers in immunology 2017;8:1047
Antigen-Presenting Human γδ T Cells Promote Intestinal CD4(+) T Cell Expression of IL-22 and Mucosal Release of Calprotectin.
Tyler CJ, McCarthy NE, Lindsay JO, Stagg AJ, Moser B, Eberl M
Journal of immunology (Baltimore, Md. : 1950) 2017 May 1;198(9):3417-3425
Journal of immunology (Baltimore, Md. : 1950) 2017 May 1;198(9):3417-3425
Systemic Human ILC Precursors Provide a Substrate for Tissue ILC Differentiation.
Lim AI, Li Y, Lopez-Lastra S, Stadhouders R, Paul F, Casrouge A, Serafini N, Puel A, Bustamante J, Surace L, Masse-Ranson G, David E, Strick-Marchand H, Le Bourhis L, Cocchi R, Topazio D, Graziano P, Muscarella LA, Rogge L, Norel X, Sallenave JM, Allez M, Graf T, Hendriks RW, Casanova JL, Amit I, Yssel H, Di Santo JP
Cell 2017 Mar 9;168(6):1086-1100.e10
Cell 2017 Mar 9;168(6):1086-1100.e10
Immune cell-derived cytokines contribute to obesity-related inflammation, fibrogenesis and metabolic deregulation in human adipose tissue.
Caër C, Rouault C, Le Roy T, Poitou C, Aron-Wisnewsky J, Torcivia A, Bichet JC, Clément K, Guerre-Millo M, André S
Scientific reports 2017 Jun 7;7(1):3000
Scientific reports 2017 Jun 7;7(1):3000
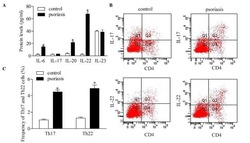
Downregulation of RUNX3 moderates the frequency of Th17 and Th22 cells in patients with psoriasis.
Fu D, Song X, Hu H, Sun M, Li Z, Tian Z
Molecular medicine reports 2016 Jun;13(6):4606-12
Molecular medicine reports 2016 Jun;13(6):4606-12
Compartment-specific immunity in the human gut: properties and functions of dendritic cells in the colon versus the ileum.
Mann ER, Bernardo D, English NR, Landy J, Al-Hassi HO, Peake ST, Man R, Elliott TR, Spranger H, Lee GH, Parian A, Brant SR, Lazarev M, Hart AL, Li X, Knight SC
Gut 2016 Feb;65(2):256-70
Gut 2016 Feb;65(2):256-70
Interleukin-25 Mediates Transcriptional Control of PD-L1 via STAT3 in Multipotent Human Mesenchymal Stromal Cells (hMSCs) to Suppress Th17 Responses.
Wang WB, Yen ML, Liu KJ, Hsu PJ, Lin MH, Chen PM, Sudhir PR, Chen CH, Chen CH, Sytwu HK, Yen BL
Stem cell reports 2015 Sep 8;5(3):392-404
Stem cell reports 2015 Sep 8;5(3):392-404
Jejunal T Cell Inflammation in Human Obesity Correlates with Decreased Enterocyte Insulin Signaling.
Monteiro-Sepulveda M, Touch S, Mendes-Sá C, André S, Poitou C, Allatif O, Cotillard A, Fohrer-Ting H, Hubert EL, Remark R, Genser L, Tordjman J, Garbin K, Osinski C, Sautès-Fridman C, Leturque A, Clément K, Brot-Laroche E
Cell metabolism 2015 Jul 7;22(1):113-24
Cell metabolism 2015 Jul 7;22(1):113-24
Early initiation of combined antiretroviral therapy preserves immune function in the gut of HIV-infected patients.
Kök A, Hocqueloux L, Hocini H, Carrière M, Lefrou L, Guguin A, Tisserand P, Bonnabau H, Avettand-Fenoel V, Prazuck T, Katsahian S, Gaulard P, Thiébaut R, Lévy Y, Hüe S
Mucosal immunology 2015 Jan;8(1):127-40
Mucosal immunology 2015 Jan;8(1):127-40
IL32 is progressively expressed in mycosis fungoides independent of helper T-cell 2 and helper T-cell 9 polarization.
Ohmatsu H, Humme D, Gulati N, Gonzalez J, Möbs M, Suárez-Fariñas M, Cardinale I, Mitsui H, Guttman-Yassky E, Sterry W, Krueger JG
Cancer immunology research 2014 Sep;2(9):890-900
Cancer immunology research 2014 Sep;2(9):890-900
T cell-derived IL-22 amplifies IL-1β-driven inflammation in human adipose tissue: relevance to obesity and type 2 diabetes.
Dalmas E, Venteclef N, Caer C, Poitou C, Cremer I, Aron-Wisnewsky J, Lacroix-Desmazes S, Bayry J, Kaveri SV, Clément K, André S, Guerre-Millo M
Diabetes 2014 Jun;63(6):1966-77
Diabetes 2014 Jun;63(6):1966-77
Increased frequencies of Th22 cells as well as Th17 cells in the peripheral blood of patients with ankylosing spondylitis and rheumatoid arthritis.
Zhang L, Li YG, Li YH, Qi L, Liu XG, Yuan CZ, Hu NW, Ma DX, Li ZF, Yang Q, Li W, Li JM
PloS one 2012;7(4):e31000
PloS one 2012;7(4):e31000
Human Th17 cells comprise heterogeneous subsets including IFN-gamma-producing cells with distinct properties from the Th1 lineage.
Boniface K, Blumenschein WM, Brovont-Porth K, McGeachy MJ, Basham B, Desai B, Pierce R, McClanahan TK, Sadekova S, de Waal Malefyt R
Journal of immunology (Baltimore, Md. : 1950) 2010 Jul 1;185(1):679-87
Journal of immunology (Baltimore, Md. : 1950) 2010 Jul 1;185(1):679-87
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Intracellular staining of CD4-enriched Th17-polarized (using Human IL-23 Recombinant Protein (Product # 14-8239-63), normal human peripheral blood cells with Anti-Human CD4 FITC (Product # 11-0049-42) and Anti-Human IL-22 PE-Cyanine7. Cultures were treated with Protein Transport Inhibitor Cocktail alone (Product # 00-4980-03) (left) or Cell Stimulation Cocktail (plus protein transport inhibitors) (Product # 00-4975-03) (right) for 5 hours prior to intracellular staining using the Intracellular Fixation & Permeabilization Buffer Set (Product # 88-8824-00). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
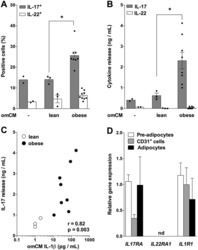
- Figure 1 Obese omental adipose tissue microenvironment promotes IL-17 release. ( A ) IL-17 + and IL-22 + cells were quantified by FACS analysis of blood-derived memory CD4 + T cells cultured for 7 days without or with conditioned medium of omental adipose tissue (omCM) from lean (n = 3) or obese (n = 8) subjects. *P < 0.05. ( B ) IL-17 and IL-22 concentrations were determined by Luminex in blood-derived memory CD4 + T cells culture medium. *P < 0.05. ( C ) Correlation analysis between IL-17 concentration in blood CD4 + T cells culture medium and IL-1beta concentration in omCM. The correlation coefficient (r) and p value were obtained by Spearman's test. (D) IL17RA, IL22RA1 and IL1R1 mRNA were determined in pre-adipocytes (n = 7), CD31 + endothelial cells (n = 6) and adipocytes (n = 4) obtained from lipoaspirate adipose tissue samples. Data are shown as mean +- SEM. nd: not detected.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
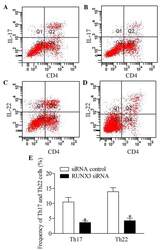
- Figure 5 Inhibition of RUNX3 regulates the frequencies of Th17 and Th22 in CD4 + T cells from patients with psoriasis. The frequencies of Th17 and Th22 cells in CD4 + T cells from patients with psoriasis transfected with RUNX3 siRNA or an siRNA control was detected using flow cytometry. (A and B) Flow cytometric analysis of Th17 in CD4 + T cells from patients with psoriasis. The percentages of cells in the Q2 region represent the percentage of Th17 cells. (C and D) Flow cytometric analysis of Th22 in CD4 + T cells from patients with psoriasis. The percentages of cells in the Q2 region represent the percentage of Th22 cells. (E) Percentages of Th17 and Th22 cells in CD4 + T cells from patients with psoriasis transfected with RUNX3 siRNA or an siRNA control. Data are presented as the mean +- standard deviation of three experiments. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
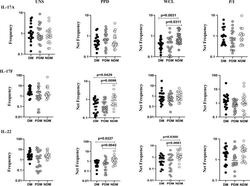
- Figure 2 Decreased frequencies of gammadelta T cells expressing Th17 cytokines in LTB comorbidities. PBMCs were either untreated or treated with Mtb or positive control antigens for 18 h. The absolute (unstimulated, UNS) and antigen-stimulated (PPD, WCL, P/I) net frequencies of Th17 (IL-17A, IL-17F, IL-22) cytokines were shown in LTB DM (n = 20), LTB PDM (n = 20), and LTB NDM (n = 20) groups. Geometric mean values were represented using bars, and every circle denotes a single individual. Kruskal-Wallis test with multiple Dunn's comparison was used to determine the p values.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 8. Changes in cytokines in recipient mice on day 21 post-allo-BMT. (A) Changes in IFN-gamma-positive Th1 in recipient mice on day 21 post-allo-BMT as determined via flow cytometry. (B) Changes in IL-4-positive Th2 cells in recipient mice on day 21 post-allo-BMT as determined via flow cytometry. (C) Changes in IL-17-positive Tg17 cells in recipient mice on day 21 post-allo-BMT as determined via flow cytometry. (D) Changes in IL-22-positive Th22 cells in recipient mice on day 21 post-allo-BMT as determined via flow cytometry. Allo-BMT, allogeneic bone marrow transplant; Th, T helper; IFN, interferon; IL, interleukin.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
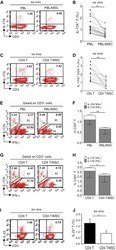
- Figure 1 Multipotent Human Mesenchymal Stromal Cells (hMSCs) Suppress Th17 Responses (A-D) Human peripheral blood CD3 + leukocytes (PBLs) (A, representative data; B, pooled data of 17 PBL donors co-cultured with all three hMSC donors) or CD3 + CD4 T cells (C, representative data; D, pooled data of 11 PBL donors co-cultured with all three hMSC donors) were co-cultured without (left) or with (right) hMSCs ex vivo, followed by PMA/ionomycin stimulation for 6 hr. (E-H) IL-17A production in ex-vivo-cultured CD3 + T cells was assessed by intracellular staining. IL-17A and IFN-gamma production in CD3 + PBLs (E, representative data; F, pooled data) or CD3 + CD4 T cells (G, representative data; H, pooled data) without and with co-culture of hMSCs was analyzed by flow cytometry. Representative intracellular staining is shown for IL-17A + IFN-gamma - - CD3 + T cells (R3 region) and IL-17A + IFN-gamma + (R5 region) CD3 + T cells, and pooled data from PBLs (n = 4) or CD4 T cells (n = 4) co-cultured with two hMSC donors (donors A and B) are provided in (F) and (H), respectively. Gray bars represent the percentages of IL-17A + IFN-gamma - - CD3 + T cells, whereas white bars represent the percentages of IL-17A + IFN-gamma + T cells. (I and J) IL-22 production in four donors of CD3 + CD4 T cells (I, representative data; J, pooled data) without and with co-culture of two donors of hMSCs (donors A and B) was assessed by intracellular staining. Cell percentages are denoted in the dotplot quadran
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
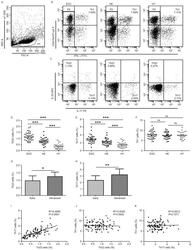
- Figure 1. Flow cytometric analysis was used to determine the distribution of Th22, Th17 and Th1 cells in EGC, HE and HY ( Fig. 1 ). Flow cytometric analysis of Th22, Th17 and Th1 cells in peripheral whole blood from EGC (n=39), HE (n=32) and HY (n=31). (A) Lymphocytes were gated in P1 using flow cytometry. CD4 + IFN-gamma - lymphocytes were gated in P2 using flow cytometry, and representative results of flow cytometric analyses for (B) Th1 (CD4 + IFN-gamma + ), (C) Th22 (CD4 + IFN-gamma - IL-17 - IL-22 + ) and Th17 (CD4 + IFN-gamma - IL-17 + IL-22 - ) cells in the three groups of subjects are presented. The number of cells stained in EGC, HE and HY in P2 were 2,654, 4,696 and 5,185, respectively. The proportion of (D) Th22, (E) Th17 and (F) Th1 cells in the three groups of subjects. The proportion of (G) Th22 and (H) Th17 cells in peripheral whole blood derived from patients with early (n=13) or advanced (n=26) gastric cancer. The association between the proportion of (I) Th22 and Th17 cells, (J) Th22 and Th1 cells, and (K) Th17 and Th1 cells, in peripheral whole blood of all subjects. *P
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry