50-7229-42
antibody from Invitrogen Antibodies
Targeting: IL22
IL-21, IL-22, IL-D110, IL-TIF, ILTIF, MGC79382, MGC79384, TIFa, TIFIL-23, zcyto18
Antibody data
- Antibody Data
- Antigen structure
- References [13]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [9]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 50-7229-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IL-22 Monoclonal Antibody (22URTI), eFluor™ 660, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The 22URTI monoclonal antibody reacts with human interleukin(IL)-22. IL-22 is a 20 kDa member of the IL-10 cytokine family that is secreted primarily by Th17 and NK cells. Nevertheless, other T cells have also been shown to produce IL-22. In in vitro Th17 cultures, induction of IL-22 expression is greater in response to IL-23 than IL-6 or TGF beta, suggesting that this cytokine may be secreted by more fully differentiated Th17 cells in vivo. A heterodimer consisting of IL-10R2 and IL-22R1 serves as the receptor for IL-22.
- Antibody clone number
- 22URTI
- Concentration
- 5 µL/Test
Submitted references Decreased Frequencies of Gamma/Delta T Cells Expressing Th1/Th17 Cytokine, Cytotoxic, and Immune Markers in Latent Tuberculosis-Diabetes/Pre-Diabetes Comorbidity.
CD4(+) T cells persist for years in the human small intestine and display a T(H)1 cytokine profile.
TIRC7 inhibits Th1 cells by upregulating the expression of CTLA‑4 and STAT3 in mice with acute graft‑versus‑host disease.
A Quantitative Multivariate Model of Human Dendritic Cell-T Helper Cell Communication.
IL-22 production of effector CD4(+) T-cells is altered in SLE patients.
The Genetic Polymorphisms of NLRP3 Inflammasome Associated with T Helper Cells in Patients with Multiple Myeloma.
Accumulation of T-helper 22 cells, interleukin-22 and myeloid-derived suppressor cells promotes gastric cancer progression in elderly patients.
Downregulation of RUNX3 moderates the frequency of Th17 and Th22 cells in patients with psoriasis.
Interleukin-25 Mediates Transcriptional Control of PD-L1 via STAT3 in Multipotent Human Mesenchymal Stromal Cells (hMSCs) to Suppress Th17 Responses.
T helper cell subsets specific for Pseudomonas aeruginosa in healthy individuals and patients with cystic fibrosis.
Elevated frequencies of circulating Th22 cell in addition to Th17 cell and Th17/Th1 cell in patients with acute coronary syndrome.
Increased frequencies of Th22 cells as well as Th17 cells in the peripheral blood of patients with ankylosing spondylitis and rheumatoid arthritis.
Induction and effector functions of T(H)17 cells.
Kathamuthu GR, Kumar NP, Moideen K, Menon PA, Babu S
Frontiers in cellular and infection microbiology 2021;11:756854
Frontiers in cellular and infection microbiology 2021;11:756854
CD4(+) T cells persist for years in the human small intestine and display a T(H)1 cytokine profile.
Bartolomé-Casado R, Landsverk OJB, Chauhan SK, Sætre F, Hagen KT, Yaqub S, Øyen O, Horneland R, Aandahl EM, Aabakken L, Bækkevold ES, Jahnsen FL
Mucosal immunology 2021 Mar;14(2):402-410
Mucosal immunology 2021 Mar;14(2):402-410
TIRC7 inhibits Th1 cells by upregulating the expression of CTLA‑4 and STAT3 in mice with acute graft‑versus‑host disease.
Zhu F, Qiu T, Zhu S, Zhao K, Chen C, Qiao J, Pan B, Yan Z, Chen W, Liu Q, Wu Q, Cao J, Sang W, Zeng L, Sun H, Li Z, Xu K
Oncology reports 2020 Jul;44(1):43-54
Oncology reports 2020 Jul;44(1):43-54
A Quantitative Multivariate Model of Human Dendritic Cell-T Helper Cell Communication.
Grandclaudon M, Perrot-Dockès M, Trichot C, Karpf L, Abouzid O, Chauvin C, Sirven P, Abou-Jaoudé W, Berger F, Hupé P, Thieffry D, Sansonnet L, Chiquet J, Lévy-Leduc C, Soumelis V
Cell 2019 Oct 3;179(2):432-447.e21
Cell 2019 Oct 3;179(2):432-447.e21
IL-22 production of effector CD4(+) T-cells is altered in SLE patients.
Dolff S, Scharpenberg C, Specker C, Kribben A, Witzke O, Wilde B
European journal of medical research 2019 Jul 22;24(1):24
European journal of medical research 2019 Jul 22;24(1):24
The Genetic Polymorphisms of NLRP3 Inflammasome Associated with T Helper Cells in Patients with Multiple Myeloma.
Zhao X, Hua M, Yan S, Yu J, Han F, Zhong C, Wang R, Zhang C, Hou M, Ma D
Journal of immunology research 2018;2018:7569809
Journal of immunology research 2018;2018:7569809
Accumulation of T-helper 22 cells, interleukin-22 and myeloid-derived suppressor cells promotes gastric cancer progression in elderly patients.
Chen X, Wang Y, Wang J, Wen J, Jia X, Wang X, Zhang H
Oncology letters 2018 Jul;16(1):253-261
Oncology letters 2018 Jul;16(1):253-261
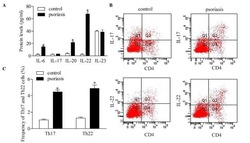
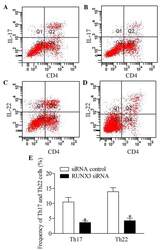
Downregulation of RUNX3 moderates the frequency of Th17 and Th22 cells in patients with psoriasis.
Fu D, Song X, Hu H, Sun M, Li Z, Tian Z
Molecular medicine reports 2016 Jun;13(6):4606-12
Molecular medicine reports 2016 Jun;13(6):4606-12
Interleukin-25 Mediates Transcriptional Control of PD-L1 via STAT3 in Multipotent Human Mesenchymal Stromal Cells (hMSCs) to Suppress Th17 Responses.
Wang WB, Yen ML, Liu KJ, Hsu PJ, Lin MH, Chen PM, Sudhir PR, Chen CH, Chen CH, Sytwu HK, Yen BL
Stem cell reports 2015 Sep 8;5(3):392-404
Stem cell reports 2015 Sep 8;5(3):392-404
T helper cell subsets specific for Pseudomonas aeruginosa in healthy individuals and patients with cystic fibrosis.
Bayes HK, Bicknell S, MacGregor G, Evans TJ
PloS one 2014;9(2):e90263
PloS one 2014;9(2):e90263
Elevated frequencies of circulating Th22 cell in addition to Th17 cell and Th17/Th1 cell in patients with acute coronary syndrome.
Zhang L, Wang T, Wang XQ, Du RZ, Zhang KN, Liu XG, Ma DX, Yu S, Su GH, Li ZH, Guan YQ, Du NL
PloS one 2013;8(12):e71466
PloS one 2013;8(12):e71466
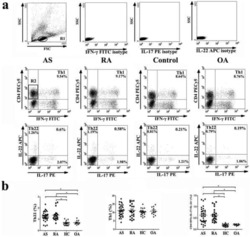
Increased frequencies of Th22 cells as well as Th17 cells in the peripheral blood of patients with ankylosing spondylitis and rheumatoid arthritis.
Zhang L, Li YG, Li YH, Qi L, Liu XG, Yuan CZ, Hu NW, Ma DX, Li ZF, Yang Q, Li W, Li JM
PloS one 2012;7(4):e31000
PloS one 2012;7(4):e31000
Induction and effector functions of T(H)17 cells.
Bettelli E, Korn T, Oukka M, Kuchroo VK
Nature 2008 Jun 19;453(7198):1051-7
Nature 2008 Jun 19;453(7198):1051-7
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- CD4-enriched human peripheral blood cells were polarized under Th17 conditions (with Human IL-23 Recombinant Protein (Product # 14-8239-63) for 10 days. Cells were restimulated with Protein Transport Inhibitor Cocktail (Product # 00-4980-03) (left) or Cell Stimulation Cocktail plus Protein Transport Inhibitors (Product # 00-4975-03) (right) for 6 hours. Cells were intracellularly stained with Anti-Human CD4 PE (Product # 12-0047-42) and Anti-Human IL-22 eFluor® 660 using the Intracellular Fixation & Permeabilization Buffer Set (Product # 88-8824-00). Viable cells, as dtermined by Fixable Viability Dye eFluor® 450 (Product # 65-0863-14), were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 5 Inhibition of RUNX3 regulates the frequencies of Th17 and Th22 in CD4 + T cells from patients with psoriasis. The frequencies of Th17 and Th22 cells in CD4 + T cells from patients with psoriasis transfected with RUNX3 siRNA or an siRNA control was detected using flow cytometry. (A and B) Flow cytometric analysis of Th17 in CD4 + T cells from patients with psoriasis. The percentages of cells in the Q2 region represent the percentage of Th17 cells. (C and D) Flow cytometric analysis of Th22 in CD4 + T cells from patients with psoriasis. The percentages of cells in the Q2 region represent the percentage of Th22 cells. (E) Percentages of Th17 and Th22 cells in CD4 + T cells from patients with psoriasis transfected with RUNX3 siRNA or an siRNA control. Data are presented as the mean +- standard deviation of three experiments. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
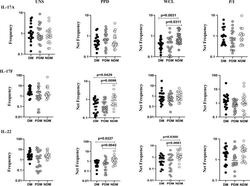
- Figure 2 Decreased frequencies of gammadelta T cells expressing Th17 cytokines in LTB comorbidities. PBMCs were either untreated or treated with Mtb or positive control antigens for 18 h. The absolute (unstimulated, UNS) and antigen-stimulated (PPD, WCL, P/I) net frequencies of Th17 (IL-17A, IL-17F, IL-22) cytokines were shown in LTB DM (n = 20), LTB PDM (n = 20), and LTB NDM (n = 20) groups. Geometric mean values were represented using bars, and every circle denotes a single individual. Kruskal-Wallis test with multiple Dunn's comparison was used to determine the p values.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 8. Changes in cytokines in recipient mice on day 21 post-allo-BMT. (A) Changes in IFN-gamma-positive Th1 in recipient mice on day 21 post-allo-BMT as determined via flow cytometry. (B) Changes in IL-4-positive Th2 cells in recipient mice on day 21 post-allo-BMT as determined via flow cytometry. (C) Changes in IL-17-positive Tg17 cells in recipient mice on day 21 post-allo-BMT as determined via flow cytometry. (D) Changes in IL-22-positive Th22 cells in recipient mice on day 21 post-allo-BMT as determined via flow cytometry. Allo-BMT, allogeneic bone marrow transplant; Th, T helper; IFN, interferon; IL, interleukin.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
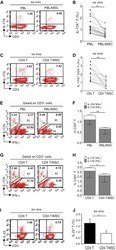
- Figure 1 Multipotent Human Mesenchymal Stromal Cells (hMSCs) Suppress Th17 Responses (A-D) Human peripheral blood CD3 + leukocytes (PBLs) (A, representative data; B, pooled data of 17 PBL donors co-cultured with all three hMSC donors) or CD3 + CD4 T cells (C, representative data; D, pooled data of 11 PBL donors co-cultured with all three hMSC donors) were co-cultured without (left) or with (right) hMSCs ex vivo, followed by PMA/ionomycin stimulation for 6 hr. (E-H) IL-17A production in ex-vivo-cultured CD3 + T cells was assessed by intracellular staining. IL-17A and IFN-gamma production in CD3 + PBLs (E, representative data; F, pooled data) or CD3 + CD4 T cells (G, representative data; H, pooled data) without and with co-culture of hMSCs was analyzed by flow cytometry. Representative intracellular staining is shown for IL-17A + IFN-gamma - - CD3 + T cells (R3 region) and IL-17A + IFN-gamma + (R5 region) CD3 + T cells, and pooled data from PBLs (n = 4) or CD4 T cells (n = 4) co-cultured with two hMSC donors (donors A and B) are provided in (F) and (H), respectively. Gray bars represent the percentages of IL-17A + IFN-gamma - - CD3 + T cells, whereas white bars represent the percentages of IL-17A + IFN-gamma + T cells. (I and J) IL-22 production in four donors of CD3 + CD4 T cells (I, representative data; J, pooled data) without and with co-culture of two donors of hMSCs (donors A and B) was assessed by intracellular staining. Cell percentages are denoted in the dotplot quadran
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
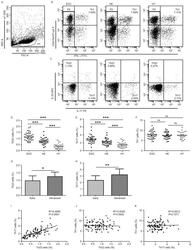
- Figure 1. Flow cytometric analysis was used to determine the distribution of Th22, Th17 and Th1 cells in EGC, HE and HY ( Fig. 1 ). Flow cytometric analysis of Th22, Th17 and Th1 cells in peripheral whole blood from EGC (n=39), HE (n=32) and HY (n=31). (A) Lymphocytes were gated in P1 using flow cytometry. CD4 + IFN-gamma - lymphocytes were gated in P2 using flow cytometry, and representative results of flow cytometric analyses for (B) Th1 (CD4 + IFN-gamma + ), (C) Th22 (CD4 + IFN-gamma - IL-17 - IL-22 + ) and Th17 (CD4 + IFN-gamma - IL-17 + IL-22 - ) cells in the three groups of subjects are presented. The number of cells stained in EGC, HE and HY in P2 were 2,654, 4,696 and 5,185, respectively. The proportion of (D) Th22, (E) Th17 and (F) Th1 cells in the three groups of subjects. The proportion of (G) Th22 and (H) Th17 cells in peripheral whole blood derived from patients with early (n=13) or advanced (n=26) gastric cancer. The association between the proportion of (I) Th22 and Th17 cells, (J) Th22 and Th1 cells, and (K) Th17 and Th1 cells, in peripheral whole blood of all subjects. *P
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry