Antibody data
- Antibody Data
- Antigen structure
- References [5]
- Comments [0]
- Validations
- Western blot [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- ABIN954206 - Provider product page

- Provider
- antibodies-online
- Product name
- anti-Procollagen-Lysine, 2-Oxoglutarate 5-Dioxygenase 3 (PLOD3) (AA 85-113), (N-Term) antibody
- Antibody type
- Polyclonal
- Description
- Protein A column, followed by peptide affinity purification
- Reactivity
- Human
- Host
- Rabbit
- Epitope
- N-Term, AA 85-113
- Vial size
- 0.4 mL
- Storage
- Store undiluted at 2-8°C for one month or (in aliquots) at -20°C for longer.
- Handling
- Avoid repeated freezing and thawing.
Submitted references The glycosyltransferase activities of lysyl hydroxylase 3 (LH3) in the extracellular space are important for cell growth and viability.
A connective tissue disorder caused by mutations of the lysyl hydroxylase 3 gene.
Lysyl hydroxylase 3 (LH3) modifies proteins in the extracellular space, a novel mechanism for matrix remodeling.
The third activity for lysyl hydroxylase 3: galactosylation of hydroxylysyl residues in collagens in vitro.
Characterization of three fragments that constitute the monomers of the human lysyl hydroxylase isoenzymes 1-3. The 30-kDa N-terminal fragment is not required for lysyl hydroxylase activity.
Wang C, Kovanen V, Raudasoja P, Eskelinen S, Pospiech H, Myllylä R
Journal of cellular and molecular medicine 2009 Mar;13(3):508-21
Journal of cellular and molecular medicine 2009 Mar;13(3):508-21
A connective tissue disorder caused by mutations of the lysyl hydroxylase 3 gene.
Salo AM, Cox H, Farndon P, Moss C, Grindulis H, Risteli M, Robins SP, Myllylä R
American journal of human genetics 2008 Oct;83(4):495-503
American journal of human genetics 2008 Oct;83(4):495-503
Lysyl hydroxylase 3 (LH3) modifies proteins in the extracellular space, a novel mechanism for matrix remodeling.
Salo AM, Wang C, Sipilä L, Sormunen R, Vapola M, Kervinen P, Ruotsalainen H, Heikkinen J, Myllylä R
Journal of cellular physiology 2006 Jun;207(3):644-53
Journal of cellular physiology 2006 Jun;207(3):644-53
The third activity for lysyl hydroxylase 3: galactosylation of hydroxylysyl residues in collagens in vitro.
Wang C, Luosujärvi H, Heikkinen J, Risteli M, Uitto L, Myllylä R
Matrix biology : journal of the International Society for Matrix Biology 2002 Nov;21(7):559-66
Matrix biology : journal of the International Society for Matrix Biology 2002 Nov;21(7):559-66
Characterization of three fragments that constitute the monomers of the human lysyl hydroxylase isoenzymes 1-3. The 30-kDa N-terminal fragment is not required for lysyl hydroxylase activity.
Rautavuoma K, Takaluoma K, Passoja K, Pirskanen A, Kvist AP, Kivirikko KI, Myllyharju J
The Journal of biological chemistry 2002 Jun 21;277(25):23084-91
The Journal of biological chemistry 2002 Jun 21;277(25):23084-91
No comments: Submit comment
Supportive validation
- Submitted by
- antibodies-online (provider)
- Main image
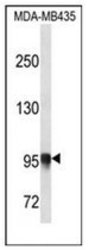
- Experimental details
- WB
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot