Antibody data
- Antibody Data
- Antigen structure
- References [80]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [17]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 12-7179-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IL-17A Monoclonal Antibody (eBio64DEC17), PE, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The eBio64DEC17 antibody reacts with human IL-17A. The eBio64DEC17 antibody is a neutralizing antibody. Interleukin-17A (IL-17A) is a CD4+ T cell-derived cytokine that promotes inflammatory responses in cell lines and is elevated in rheumatoid arthritis, asthma, multiple sclerosis, psoriasis, and transplant rejection. The cDNA encoding human IL-17A was isolated from a library of CD4+ T cells; the encoded protein exhibits 72 percent amino acid identity with HVS13 , an open reading frame from a T lymphotropic Herpesvirus saimiri, and 63 percent with mouse CTLA-8 (cytotoxic T-lymphocyte associated antigen-8). Human IL-17A exists as glycosylated 20-30 kD homodimers. High levels of IL-17A homodimer are produced by activated peripheral blood CD4+ T-cells. IL-17A enhances expression of the intracellular adhesion molecule-1 (ICAM-1) in human fibroblasts. Human IL-17A also stimulates epithelial, endothelial, or fibroblastic cells to secrete IL-6, IL-8, G-CSF, and PGE2. In the presence of human IL-17A, fibroblasts can sustain the proliferation of CD34+ hematopoietic progenitors and induce maturation into neutrophils. Mouse, rat, and human IL-17A can induce IL-6 secretion in mouse stromal cells, indicating that all homologs can recognize the mouse IL-17A receptor. IL-23-dependent, IL-17A-producing CD4+ T cells (Th-17 cells) have been identified as a unique subset of Th cells that develops along a pathway that is distinct from the Th1- and Th2- cell differentiation pathways. The hallmark effector molecules of Th1 and Th2 cells, e.g., IFN gamma and IL-4, have each been found to negatively regulate the generation of these Th-17 cells. Intracellular staining by eBio64DEC17 antibody identifies the same cell population as the eBio64CAP17 antibody, as can be seen in co-staining experiments using both antibodies. Applications Reported: This eBio64DEC17 antibody has been reported for use in intracellular staining followed by flow cytometric analysis. Applications Tested: This eBio64DEC17 antibody has been pre-titrated and tested by intracellular staining and flow cytometric analysis of stimulated normal human peripheral blood cells. This can be used at 5 µL (0.25 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488-561 nm; Emission: 578 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Yellow dye
- Isotype
- IgG
- Antibody clone number
- eBio64DEC17
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references EC-18 prevents autoimmune arthritis by suppressing inflammatory cytokines and osteoclastogenesis.
Dysregulated Peripheral Invariant Natural Killer T Cells in Plaque Psoriasis Patients.
Immunomodulatory Effects of Canine Adipose Tissue Mesenchymal Stem Cell-Derived Extracellular Vesicles on Stimulated CD4(+) T Cells Isolated from Peripheral Blood Mononuclear Cells.
CD4(+) T cells persist for years in the human small intestine and display a T(H)1 cytokine profile.
Immune-checkpoint blockade of CTLA-4 (CD152) in antigen-specific human T-cell responses differs profoundly between neonates, children, and adults.
The neutrophil antimicrobial peptide cathelicidin promotes Th17 differentiation.
Small-molecule antagonist of VLA-4 (GW559090) attenuated neuro-inflammation by targeting Th17 cell trafficking across the blood-retinal barrier in experimental autoimmune uveitis.
IL-12-Induced Immune Suppressive Deficit During CD8+ T-Cell Differentiation.
Robust T Cell Immunity in Convalescent Individuals with Asymptomatic or Mild COVID-19.
Th17/Treg imbalance modulates rat myocardial fibrosis and heart failure by regulating LOX expression.
Paradoxical psoriasis induced by TNF-α blockade shows immunological features typical of the early phase of psoriasis development.
Serum Amyloid A Proteins Induce Pathogenic Th17 Cells and Promote Inflammatory Disease.
Differential influences of the BPA, BPS and BPF on in vitro IL-17 secretion by mouse and human T cells.
In Vitro Induction of T Helper 17 Cells by Synergistic Activation of Human Monocyte-Derived Langerhans Cell-Like Cells with Bacterial Agonists.
IL-2 Inhibition of Th17 Generation Rather Than Induction of Treg Cells Is Impaired in Primary Sjögren's Syndrome Patients.
Effect of Equipotent Doses of Propofol versus Sevoflurane Anesthesia on Regulatory T Cells after Breast Cancer Surgery.
The NRF2 pathway as potential biomarker for dimethyl fumarate treatment in multiple sclerosis.
The transcription factor CREM drives an inflammatory phenotype of T cells in oligoarticular juvenile idiopathic arthritis.
Accumulation of T-helper 22 cells, interleukin-22 and myeloid-derived suppressor cells promotes gastric cancer progression in elderly patients.
The Translational Machinery of Human CD4(+) T Cells Is Poised for Activation and Controls the Switch from Quiescence to Metabolic Remodeling.
CD39(+) regulatory T cells accumulate in colon adenocarcinomas and display markers of increased suppressive function.
Age-associated alteration in Th17 cell response is related to endothelial cell senescence and atherosclerotic cerebral infarction.
Protective effect of 1α,25-dihydroxyvitamin D3 on effector CD4+ T cell induced injury in human renal proximal tubular epithelial cells.
Transcriptional signature of human pro-inflammatory T(H)17 cells identifies reduced IL10 gene expression in multiple sclerosis.
Oncostatin M drives intestinal inflammation and predicts response to tumor necrosis factor-neutralizing therapy in patients with inflammatory bowel disease.
Cytochrome P450s in human immune cells regulate IL-22 and c-Kit via an AHR feedback loop.
IL-1β induced HIF-1α inhibits the differentiation of human FOXP3(+) T cells.
Berberine exerts an anti-inflammatory role in ocular Behcet's disease.
Single-cell profiling reveals GPCR heterogeneity and functional patterning during neuroinflammation.
Enhancing the toolbox to study IL-17A in cattle and sheep.
TGF-β Affects the Differentiation of Human GM-CSF(+) CD4(+) T Cells in an Activation- and Sodium-Dependent Manner.
CD16(+) Monocyte Subset Was Enriched and Functionally Exacerbated in Driving T-Cell Activation and B-Cell Response in Systemic Lupus Erythematosus.
Booster vaccination against tetanus and diphtheria: insufficient protection against diphtheria in young and elderly adults.
CCR6(-) regulatory T cells blunt the restoration of gut Th17 cells along the CCR6-CCL20 axis in treated HIV-1-infected individuals.
FOXP3(+) associated with the pro-inflammatory regulatory T and T helper 17 effector cells in asthma patients.
IL-21 and probiotic therapy improve Th17 frequencies, microbial translocation, and microbiome in ARV-treated, SIV-infected macaques.
Interaction among activated lymphocytes and mesenchymal cells through podoplanin is critical for a high IL-17 secretion.
Impact of immunosuppressive drugs on the therapeutic efficacy of ex vivo expanded human regulatory T cells.
ILC3 GM-CSF production and mobilisation orchestrate acute intestinal inflammation.
IL-17A- and IFNγ-Producing T Cells in Healthy Skin.
Vaccination against canine leishmaniosis increases the phagocytic activity, nitric oxide production and expression of cell activation/migration molecules in neutrophils and monocytes.
Increase of Th17 Cell Phenotype in Kidney Transplant Recipients with Chronic Allograft Dysfunction.
Interleukin-25 Mediates Transcriptional Control of PD-L1 via STAT3 in Multipotent Human Mesenchymal Stromal Cells (hMSCs) to Suppress Th17 Responses.
Cross-reactivity of commercially available anti-human monoclonal antibodies with canine cytokines: establishment of a reliable panel to detect the functional profile of peripheral blood lymphocytes by intracytoplasmic staining.
Optimization of adipose tissue-derived mesenchymal stem cells by rapamycin in a murine model of acute graft-versus-host disease.
Human Mucosa-Associated Invariant T Cells Accumulate in Colon Adenocarcinomas but Produce Reduced Amounts of IFN-γ.
NY-ESO-1 specific antibody and cellular responses in melanoma patients primed with NY-ESO-1 protein in ISCOMATRIX and boosted with recombinant NY-ESO-1 fowlpox virus.
Effector Vγ9Vδ2 T cells dominate the human fetal γδ T-cell repertoire.
DCLRE1C (ARTEMIS) mutations causing phenotypes ranging from atypical severe combined immunodeficiency to mere antibody deficiency.
Identification of novel HIV-1 dependency factors in primary CCR4(+)CCR6(+)Th17 cells via a genome-wide transcriptional approach.
Whole CMV proteome pattern recognition analysis after HSCT identifies unique epitope targets associated with the CMV status.
Immunomodulation of human intestinal T cells by the synthetic CD80 antagonist RhuDex®.
Agonistic induction of PPARγ reverses cigarette smoke-induced emphysema.
Decreases in colonic and systemic inflammation in chronic HIV infection after IL-7 administration.
Patients with the most advanced rheumatoid arthritis remain with Th1 systemic defects after TNF inhibitors treatment despite clinical improvement.
The disturbance of TH17-Treg cell balance in adenomyosis.
Elevated frequencies of circulating Th22 cell in addition to Th17 cell and Th17/Th1 cell in patients with acute coronary syndrome.
Peripheral Th17/Treg imbalance in patients with atherosclerotic cerebral infarction.
An ACT1 mutation selectively abolishes interleukin-17 responses in humans with chronic mucocutaneous candidiasis.
Induction and stability of human Th17 cells require endogenous NOS2 and cGMP-dependent NO signaling.
Association of a NOD2 gene polymorphism and T-helper 17 cells with presumed ocular toxoplasmosis.
Patients with cystic fibrosis have inducible IL-17+IL-22+ memory cells in lung draining lymph nodes.
Frequency of Th17 CD4+ T cells in early rheumatoid arthritis: a marker of anti-CCP seropositivity.
Dysregulation of Th17 cells during the early post-transplant period in patients under calcineurin inhibitor based immunosuppression.
Increased frequencies of Th22 cells as well as Th17 cells in the peripheral blood of patients with ankylosing spondylitis and rheumatoid arthritis.
The colocalization potential of HIV-specific CD8+ and CD4+ T-cells is mediated by integrin β7 but not CCR6 and regulated by retinoic acid.
Characterization of Th17 responses to Streptococcus pneumoniae in humans: comparisons between adults and children in a developed and a developing country.
Cigarette smoke induction of osteopontin (SPP1) mediates T(H)17 inflammation in human and experimental emphysema.
Complement component C5a promotes expression of IL-22 and IL-17 from human T cells and its implication in age-related macular degeneration.
IL-1β and TGF-β act antagonistically in induction and differentially in propagation of human proinflammatory precursor CD4+ T cells.
Memory CCR6+CD4+ T cells are preferential targets for productive HIV type 1 infection regardless of their expression of integrin β7.
Generation of IL-23 producing dendritic cells (DCs) by airborne fungi regulates fungal pathogenicity via the induction of T(H)-17 responses.
Peripheral blood CCR4+CCR6+ and CXCR3+CCR6+CD4+ T cells are highly permissive to HIV-1 infection.
Human plasmacytoid dendritic cells support Th17 cell effector function in response to TLR7 ligation.
Interleukin-12 is the optimum cytokine to expand human Th17 cells in vitro.
The effects of trastuzumab on the CD4+CD25+FoxP3+ and CD4+IL17A+ T-cell axis in patients with breast cancer.
Prolonged, granulocyte-macrophage colony-stimulating factor-dependent, neutrophil survival following rheumatoid synovial fibroblast activation by IL-17 and TNFalpha.
Phenotypic analysis of prostate-infiltrating lymphocytes reveals TH17 and Treg skewing.
Interleukins 1beta and 6 but not transforming growth factor-beta are essential for the differentiation of interleukin 17-producing human T helper cells.
Distinct regulation of interleukin-17 in human T helper lymphocytes.
Park JS, Yang SC, Jeong HY, Lee SY, Ryu JG, Choi JW, Kang HY, Kim SM, Hwang SH, Cho ML, Park SH
Arthritis research & therapy 2022 Nov 17;24(1):254
Arthritis research & therapy 2022 Nov 17;24(1):254
Dysregulated Peripheral Invariant Natural Killer T Cells in Plaque Psoriasis Patients.
Hu Y, Chen Y, Chen Z, Zhang X, Guo C, Yu Z, Xu P, Sun L, Zhou X, Gong Y, Yu Q, Shi Y
Frontiers in cell and developmental biology 2021;9:799560
Frontiers in cell and developmental biology 2021;9:799560
Immunomodulatory Effects of Canine Adipose Tissue Mesenchymal Stem Cell-Derived Extracellular Vesicles on Stimulated CD4(+) T Cells Isolated from Peripheral Blood Mononuclear Cells.
Teshima T, Yuchi Y, Suzuki R, Matsumoto H, Koyama H
Journal of immunology research 2021;2021:2993043
Journal of immunology research 2021;2021:2993043
CD4(+) T cells persist for years in the human small intestine and display a T(H)1 cytokine profile.
Bartolomé-Casado R, Landsverk OJB, Chauhan SK, Sætre F, Hagen KT, Yaqub S, Øyen O, Horneland R, Aandahl EM, Aabakken L, Bækkevold ES, Jahnsen FL
Mucosal immunology 2021 Mar;14(2):402-410
Mucosal immunology 2021 Mar;14(2):402-410
Immune-checkpoint blockade of CTLA-4 (CD152) in antigen-specific human T-cell responses differs profoundly between neonates, children, and adults.
Arra A, Pech M, Fu H, Lingel H, Braun F, Beyer C, Spiliopoulou M, Bröker BM, Lampe K, Arens C, Vogel K, Pierau M, Brunner-Weinzierl MC
Oncoimmunology 2021 Jun 14;10(1):1938475
Oncoimmunology 2021 Jun 14;10(1):1938475
The neutrophil antimicrobial peptide cathelicidin promotes Th17 differentiation.
Minns D, Smith KJ, Alessandrini V, Hardisty G, Melrose L, Jackson-Jones L, MacDonald AS, Davidson DJ, Gwyer Findlay E
Nature communications 2021 Feb 24;12(1):1285
Nature communications 2021 Feb 24;12(1):1285
Small-molecule antagonist of VLA-4 (GW559090) attenuated neuro-inflammation by targeting Th17 cell trafficking across the blood-retinal barrier in experimental autoimmune uveitis.
Chen YH, Eskandarpour M, Zhang X, Galatowicz G, Greenwood J, Lightman S, Calder V
Journal of neuroinflammation 2021 Feb 18;18(1):49
Journal of neuroinflammation 2021 Feb 18;18(1):49
IL-12-Induced Immune Suppressive Deficit During CD8+ T-Cell Differentiation.
Renavikar PS, Sinha S, Brate AA, Borcherding N, Crawford MP, Steward-Tharp SM, Karandikar NJ
Frontiers in immunology 2020;11:568630
Frontiers in immunology 2020;11:568630
Robust T Cell Immunity in Convalescent Individuals with Asymptomatic or Mild COVID-19.
Sekine T, Perez-Potti A, Rivera-Ballesteros O, Strålin K, Gorin JB, Olsson A, Llewellyn-Lacey S, Kamal H, Bogdanovic G, Muschiol S, Wullimann DJ, Kammann T, Emgård J, Parrot T, Folkesson E, Karolinska COVID-19 Study Group, Rooyackers O, Eriksson LI, Henter JI, Sönnerborg A, Allander T, Albert J, Nielsen M, Klingström J, Gredmark-Russ S, Björkström NK, Sandberg JK, Price DA, Ljunggren HG, Aleman S, Buggert M
Cell 2020 Oct 1;183(1):158-168.e14
Cell 2020 Oct 1;183(1):158-168.e14
Th17/Treg imbalance modulates rat myocardial fibrosis and heart failure by regulating LOX expression.
Lu M, Qin X, Yao J, Yang Y, Zhao M, Sun L
Acta physiologica (Oxford, England) 2020 Nov;230(3):e13537
Acta physiologica (Oxford, England) 2020 Nov;230(3):e13537
Paradoxical psoriasis induced by TNF-α blockade shows immunological features typical of the early phase of psoriasis development.
Fania L, Morelli M, Scarponi C, Mercurio L, Scopelliti F, Cattani C, Scaglione GL, Tonanzi T, Pilla MA, Pagnanelli G, Mazzanti C, Girolomoni G, Cavani A, Madonna S, Albanesi C
The journal of pathology. Clinical research 2020 Jan;6(1):55-68
The journal of pathology. Clinical research 2020 Jan;6(1):55-68
Serum Amyloid A Proteins Induce Pathogenic Th17 Cells and Promote Inflammatory Disease.
Lee JY, Hall JA, Kroehling L, Wu L, Najar T, Nguyen HH, Lin WY, Yeung ST, Silva HM, Li D, Hine A, Loke P, Hudesman D, Martin JC, Kenigsberg E, Merad M, Khanna KM, Littman DR
Cell 2020 Jan 9;180(1):79-91.e16
Cell 2020 Jan 9;180(1):79-91.e16
Differential influences of the BPA, BPS and BPF on in vitro IL-17 secretion by mouse and human T cells.
Malaisé Y, Le Mentec H, Sparfel L, Guzylack-Piriou L
Toxicology in vitro : an international journal published in association with BIBRA 2020 Dec;69:104993
Toxicology in vitro : an international journal published in association with BIBRA 2020 Dec;69:104993
In Vitro Induction of T Helper 17 Cells by Synergistic Activation of Human Monocyte-Derived Langerhans Cell-Like Cells with Bacterial Agonists.
Gramlich R, Aliahmadi E, Peiser M
International journal of molecular sciences 2019 Mar 19;20(6)
International journal of molecular sciences 2019 Mar 19;20(6)
IL-2 Inhibition of Th17 Generation Rather Than Induction of Treg Cells Is Impaired in Primary Sjögren's Syndrome Patients.
Luo J, Ming B, Zhang C, Deng X, Li P, Wei Z, Xia Y, Jiang K, Ye H, Ma W, Liu Z, Li H, Yang XP, Dong L
Frontiers in immunology 2018;9:1755
Frontiers in immunology 2018;9:1755
Effect of Equipotent Doses of Propofol versus Sevoflurane Anesthesia on Regulatory T Cells after Breast Cancer Surgery.
Oh CS, Lee J, Yoon TG, Seo EH, Park HJ, Piao L, Lee SH, Kim SH
Anesthesiology 2018 Nov;129(5):921-931
Anesthesiology 2018 Nov;129(5):921-931
The NRF2 pathway as potential biomarker for dimethyl fumarate treatment in multiple sclerosis.
Hammer A, Waschbisch A, Kuhbandner K, Bayas A, Lee DH, Duscha A, Haghikia A, Gold R, Linker RA
Annals of clinical and translational neurology 2018 Jun;5(6):668-676
Annals of clinical and translational neurology 2018 Jun;5(6):668-676
The transcription factor CREM drives an inflammatory phenotype of T cells in oligoarticular juvenile idiopathic arthritis.
Ohl K, Nickel H, Moncrieffe H, Klemm P, Scheufen A, Föll D, Wixler V, Schippers A, Wagner N, Wedderburn LR, Tenbrock K
Pediatric rheumatology online journal 2018 Jun 20;16(1):39
Pediatric rheumatology online journal 2018 Jun 20;16(1):39
Accumulation of T-helper 22 cells, interleukin-22 and myeloid-derived suppressor cells promotes gastric cancer progression in elderly patients.
Chen X, Wang Y, Wang J, Wen J, Jia X, Wang X, Zhang H
Oncology letters 2018 Jul;16(1):253-261
Oncology letters 2018 Jul;16(1):253-261
The Translational Machinery of Human CD4(+) T Cells Is Poised for Activation and Controls the Switch from Quiescence to Metabolic Remodeling.
Ricciardi S, Manfrini N, Alfieri R, Calamita P, Crosti MC, Gallo S, Müller R, Pagani M, Abrignani S, Biffo S
Cell metabolism 2018 Dec 4;28(6):895-906.e5
Cell metabolism 2018 Dec 4;28(6):895-906.e5
CD39(+) regulatory T cells accumulate in colon adenocarcinomas and display markers of increased suppressive function.
Ahlmanner F, Sundström P, Akeus P, Eklöf J, Börjesson L, Gustavsson B, Lindskog EB, Raghavan S, Quiding-Järbrink M
Oncotarget 2018 Dec 11;9(97):36993-37007
Oncotarget 2018 Dec 11;9(97):36993-37007
Age-associated alteration in Th17 cell response is related to endothelial cell senescence and atherosclerotic cerebral infarction.
Li Q, Ding S, Wang YM, Xu X, Shen Z, Fu R, Liu M, Hu C, Zhang C, Cao Q, Wang Y
American journal of translational research 2017;9(11):5160-5168
American journal of translational research 2017;9(11):5160-5168
Protective effect of 1α,25-dihydroxyvitamin D3 on effector CD4+ T cell induced injury in human renal proximal tubular epithelial cells.
Chung BH, Kim BM, Doh KC, Cho ML, Kim KW, Yang CW
PloS one 2017;12(2):e0172536
PloS one 2017;12(2):e0172536
Transcriptional signature of human pro-inflammatory T(H)17 cells identifies reduced IL10 gene expression in multiple sclerosis.
Hu D, Notarbartolo S, Croonenborghs T, Patel B, Cialic R, Yang TH, Aschenbrenner D, Andersson KM, Gattorno M, Pham M, Kivisakk P, Pierre IV, Lee Y, Kiani K, Bokarewa M, Tjon E, Pochet N, Sallusto F, Kuchroo VK, Weiner HL
Nature communications 2017 Nov 17;8(1):1600
Nature communications 2017 Nov 17;8(1):1600
Oncostatin M drives intestinal inflammation and predicts response to tumor necrosis factor-neutralizing therapy in patients with inflammatory bowel disease.
West NR, Hegazy AN, Owens BMJ, Bullers SJ, Linggi B, Buonocore S, Coccia M, Görtz D, This S, Stockenhuber K, Pott J, Friedrich M, Ryzhakov G, Baribaud F, Brodmerkel C, Cieluch C, Rahman N, Müller-Newen G, Owens RJ, Kühl AA, Maloy KJ, Plevy SE, Oxford IBD Cohort Investigators, Keshav S, Travis SPL, Powrie F
Nature medicine 2017 May;23(5):579-589
Nature medicine 2017 May;23(5):579-589
Cytochrome P450s in human immune cells regulate IL-22 and c-Kit via an AHR feedback loop.
Effner R, Hiller J, Eyerich S, Traidl-Hoffmann C, Brockow K, Triggiani M, Behrendt H, Schmidt-Weber CB, Buters JT
Scientific reports 2017 Mar 9;7:44005
Scientific reports 2017 Mar 9;7:44005
IL-1β induced HIF-1α inhibits the differentiation of human FOXP3(+) T cells.
Feldhoff LM, Rueda CM, Moreno-Fernandez ME, Sauer J, Jackson CM, Chougnet CA, Rupp J
Scientific reports 2017 Mar 28;7(1):465
Scientific reports 2017 Mar 28;7(1):465
Berberine exerts an anti-inflammatory role in ocular Behcet's disease.
Yang Y, Wang Q, Xie M, Liu P, Qi X, Liu X, Li Z
Molecular medicine reports 2017 Jan;15(1):97-102
Molecular medicine reports 2017 Jan;15(1):97-102
Single-cell profiling reveals GPCR heterogeneity and functional patterning during neuroinflammation.
Tischner D, Grimm M, Kaur H, Staudenraus D, Carvalho J, Looso M, Günther S, Wanke F, Moos S, Siller N, Breuer J, Schwab N, Zipp F, Waisman A, Kurschus FC, Offermanns S, Wettschureck N
JCI insight 2017 Aug 3;2(15)
JCI insight 2017 Aug 3;2(15)
Enhancing the toolbox to study IL-17A in cattle and sheep.
Wattegedera SR, Corripio-Miyar Y, Pang Y, Frew D, McNeilly TN, Palarea-Albaladejo J, McInnes CJ, Hope JC, Glass EJ, Entrican G
Veterinary research 2017 Apr 8;48(1):20
Veterinary research 2017 Apr 8;48(1):20
TGF-β Affects the Differentiation of Human GM-CSF(+) CD4(+) T Cells in an Activation- and Sodium-Dependent Manner.
Éliás S, Schmidt A, Kannan V, Andersson J, Tegnér J
Frontiers in immunology 2016;7:603
Frontiers in immunology 2016;7:603
CD16(+) Monocyte Subset Was Enriched and Functionally Exacerbated in Driving T-Cell Activation and B-Cell Response in Systemic Lupus Erythematosus.
Zhu H, Hu F, Sun X, Zhang X, Zhu L, Liu X, Li X, Xu L, Shi L, Gan Y, Su Y
Frontiers in immunology 2016;7:512
Frontiers in immunology 2016;7:512
Booster vaccination against tetanus and diphtheria: insufficient protection against diphtheria in young and elderly adults.
Grasse M, Meryk A, Schirmer M, Grubeck-Loebenstein B, Weinberger B
Immunity & ageing : I & A 2016;13(1):26
Immunity & ageing : I & A 2016;13(1):26
CCR6(-) regulatory T cells blunt the restoration of gut Th17 cells along the CCR6-CCL20 axis in treated HIV-1-infected individuals.
Loiseau C, Requena M, Mavigner M, Cazabat M, Carrere N, Suc B, Barange K, Alric L, Marchou B, Massip P, Izopet J, Delobel P
Mucosal immunology 2016 Sep;9(5):1137-50
Mucosal immunology 2016 Sep;9(5):1137-50
FOXP3(+) associated with the pro-inflammatory regulatory T and T helper 17 effector cells in asthma patients.
Zhang JG, Chen XJ, Liu T, Jiang SJ
Experimental and therapeutic medicine 2016 Oct;12(4):2753-2758
Experimental and therapeutic medicine 2016 Oct;12(4):2753-2758
IL-21 and probiotic therapy improve Th17 frequencies, microbial translocation, and microbiome in ARV-treated, SIV-infected macaques.
Ortiz AM, Klase ZA, DiNapoli SR, Vujkovic-Cvijin I, Carmack K, Perkins MR, Calantone N, Vinton CL, Riddick NE, Gallagher J, Klatt NR, McCune JM, Estes JD, Paiardini M, Brenchley JM
Mucosal immunology 2016 Mar;9(2):458-67
Mucosal immunology 2016 Mar;9(2):458-67
Interaction among activated lymphocytes and mesenchymal cells through podoplanin is critical for a high IL-17 secretion.
Noack M, Ndongo-Thiam N, Miossec P
Arthritis research & therapy 2016 Jun 23;18:148
Arthritis research & therapy 2016 Jun 23;18:148
Impact of immunosuppressive drugs on the therapeutic efficacy of ex vivo expanded human regulatory T cells.
Scottà C, Fanelli G, Hoong SJ, Romano M, Lamperti EN, Sukthankar M, Guggino G, Fazekasova H, Ratnasothy K, Becker PD, Afzali B, Lechler RI, Lombardi G
Haematologica 2016 Jan;101(1):91-100
Haematologica 2016 Jan;101(1):91-100
ILC3 GM-CSF production and mobilisation orchestrate acute intestinal inflammation.
Pearson C, Thornton EE, McKenzie B, Schaupp AL, Huskens N, Griseri T, West N, Tung S, Seddon BP, Uhlig HH, Powrie F
eLife 2016 Jan 18;5:e10066
eLife 2016 Jan 18;5:e10066
IL-17A- and IFNγ-Producing T Cells in Healthy Skin.
Dyring-Andersen B, Nielsen MM, Geisler C, Bonefeld CM, Skov L
Scandinavian journal of immunology 2016 Apr;83(4):297-9
Scandinavian journal of immunology 2016 Apr;83(4):297-9
Vaccination against canine leishmaniosis increases the phagocytic activity, nitric oxide production and expression of cell activation/migration molecules in neutrophils and monocytes.
Moreira ML, Costa-Pereira C, Alves ML, Marteleto BH, Ribeiro VM, Peruhype-Magalhães V, Giunchetti RC, Martins-Filho OA, Araújo MS
Veterinary parasitology 2016 Apr 15;220:33-45
Veterinary parasitology 2016 Apr 15;220:33-45
Increase of Th17 Cell Phenotype in Kidney Transplant Recipients with Chronic Allograft Dysfunction.
Chung BH, Kim KW, Kim BM, Doh KC, Cho ML, Yang CW
PloS one 2015;10(12):e0145258
PloS one 2015;10(12):e0145258
Interleukin-25 Mediates Transcriptional Control of PD-L1 via STAT3 in Multipotent Human Mesenchymal Stromal Cells (hMSCs) to Suppress Th17 Responses.
Wang WB, Yen ML, Liu KJ, Hsu PJ, Lin MH, Chen PM, Sudhir PR, Chen CH, Chen CH, Sytwu HK, Yen BL
Stem cell reports 2015 Sep 8;5(3):392-404
Stem cell reports 2015 Sep 8;5(3):392-404
Cross-reactivity of commercially available anti-human monoclonal antibodies with canine cytokines: establishment of a reliable panel to detect the functional profile of peripheral blood lymphocytes by intracytoplasmic staining.
Moreira ML, Dorneles EM, Soares RP, Magalhães CP, Costa-Pereira C, Lage AP, Teixeira-Carvalho A, Martins-Filho OA, Araújo MS
Acta veterinaria Scandinavica 2015 Sep 11;57(1):51
Acta veterinaria Scandinavica 2015 Sep 11;57(1):51
Optimization of adipose tissue-derived mesenchymal stem cells by rapamycin in a murine model of acute graft-versus-host disease.
Kim KW, Moon SJ, Park MJ, Kim BM, Kim EK, Lee SH, Lee EJ, Chung BH, Yang CW, Cho ML
Stem cell research & therapy 2015 Oct 23;6:202
Stem cell research & therapy 2015 Oct 23;6:202
Human Mucosa-Associated Invariant T Cells Accumulate in Colon Adenocarcinomas but Produce Reduced Amounts of IFN-γ.
Sundström P, Ahlmanner F, Akéus P, Sundquist M, Alsén S, Yrlid U, Börjesson L, Sjöling Å, Gustavsson B, Wong SB, Quiding-Järbrink M
Journal of immunology (Baltimore, Md. : 1950) 2015 Oct 1;195(7):3472-81
Journal of immunology (Baltimore, Md. : 1950) 2015 Oct 1;195(7):3472-81
NY-ESO-1 specific antibody and cellular responses in melanoma patients primed with NY-ESO-1 protein in ISCOMATRIX and boosted with recombinant NY-ESO-1 fowlpox virus.
Chen JL, Dawoodji A, Tarlton A, Gnjatic S, Tajar A, Karydis I, Browning J, Pratap S, Verfaille C, Venhaus RR, Pan L, Altman DG, Cebon JS, Old LL, Nathan P, Ottensmeier C, Middleton M, Cerundolo V
International journal of cancer 2015 Mar 15;136(6):E590-601
International journal of cancer 2015 Mar 15;136(6):E590-601
Effector Vγ9Vδ2 T cells dominate the human fetal γδ T-cell repertoire.
Dimova T, Brouwer M, Gosselin F, Tassignon J, Leo O, Donner C, Marchant A, Vermijlen D
Proceedings of the National Academy of Sciences of the United States of America 2015 Feb 10;112(6):E556-65
Proceedings of the National Academy of Sciences of the United States of America 2015 Feb 10;112(6):E556-65
DCLRE1C (ARTEMIS) mutations causing phenotypes ranging from atypical severe combined immunodeficiency to mere antibody deficiency.
Volk T, Pannicke U, Reisli I, Bulashevska A, Ritter J, Björkman A, Schäffer AA, Fliegauf M, Sayar EH, Salzer U, Fisch P, Pfeifer D, Di Virgilio M, Cao H, Yang F, Zimmermann K, Keles S, Caliskaner Z, Güner SÜ, Schindler D, Hammarström L, Rizzi M, Hummel M, Pan-Hammarström Q, Schwarz K, Grimbacher B
Human molecular genetics 2015 Dec 20;24(25):7361-72
Human molecular genetics 2015 Dec 20;24(25):7361-72
Identification of novel HIV-1 dependency factors in primary CCR4(+)CCR6(+)Th17 cells via a genome-wide transcriptional approach.
Cleret-Buhot A, Zhang Y, Planas D, Goulet JP, Monteiro P, Gosselin A, Wacleche VS, Tremblay CL, Jenabian MA, Routy JP, El-Far M, Chomont N, Haddad EK, Sekaly RP, Ancuta P
Retrovirology 2015 Dec 10;12:102
Retrovirology 2015 Dec 10;12:102
Whole CMV proteome pattern recognition analysis after HSCT identifies unique epitope targets associated with the CMV status.
Pérez-Bercoff L, Valentini D, Gaseitsiwe S, Mahdavifar S, Schutkowski M, Poiret T, Pérez-Bercoff Å, Ljungman P, Maeurer MJ
PloS one 2014;9(4):e89648
PloS one 2014;9(4):e89648
Immunomodulation of human intestinal T cells by the synthetic CD80 antagonist RhuDex®.
Heninger AK, Wentrup S, Al-Saeedi M, Schiessling S, Giese T, Wartha F, Meuer S, Schröder-Braunstein J
Immunity, inflammation and disease 2014 Nov;2(3):166-80
Immunity, inflammation and disease 2014 Nov;2(3):166-80
Agonistic induction of PPARγ reverses cigarette smoke-induced emphysema.
Shan M, You R, Yuan X, Frazier MV, Porter P, Seryshev A, Hong JS, Song LZ, Zhang Y, Hilsenbeck S, Whitehead L, Zarinkamar N, Perusich S, Corry DB, Kheradmand F
The Journal of clinical investigation 2014 Mar;124(3):1371-81
The Journal of clinical investigation 2014 Mar;124(3):1371-81
Decreases in colonic and systemic inflammation in chronic HIV infection after IL-7 administration.
Sereti I, Estes JD, Thompson WL, Morcock DR, Fischl MA, Croughs T, Beq S, Lafaye de Micheaux S, Yao MD, Ober A, Wilson EM, Natarajan V, Imamichi H, Boulassel MR, Lederman MM, Routy JP
PLoS pathogens 2014 Jan;10(1):e1003890
PLoS pathogens 2014 Jan;10(1):e1003890
Patients with the most advanced rheumatoid arthritis remain with Th1 systemic defects after TNF inhibitors treatment despite clinical improvement.
Kosmaczewska A, Swierkot J, Ciszak L, Szteblich A, Chrobak A, Karabon L, Partyka A, Szechinski J, Wiland P, Frydecka I
Rheumatology international 2014 Feb;34(2):243-53
Rheumatology international 2014 Feb;34(2):243-53
The disturbance of TH17-Treg cell balance in adenomyosis.
Gui T, Chen C, Zhang Z, Tang W, Qian R, Ma X, Cao P, Wan G
Fertility and sterility 2014 Feb;101(2):506-14
Fertility and sterility 2014 Feb;101(2):506-14
Elevated frequencies of circulating Th22 cell in addition to Th17 cell and Th17/Th1 cell in patients with acute coronary syndrome.
Zhang L, Wang T, Wang XQ, Du RZ, Zhang KN, Liu XG, Ma DX, Yu S, Su GH, Li ZH, Guan YQ, Du NL
PloS one 2013;8(12):e71466
PloS one 2013;8(12):e71466
Peripheral Th17/Treg imbalance in patients with atherosclerotic cerebral infarction.
Li Q, Wang Y, Yu F, Wang YM, Zhang C, Hu C, Wu Z, Xu X, Hu S
International journal of clinical and experimental pathology 2013;6(6):1015-27
International journal of clinical and experimental pathology 2013;6(6):1015-27
An ACT1 mutation selectively abolishes interleukin-17 responses in humans with chronic mucocutaneous candidiasis.
Boisson B, Wang C, Pedergnana V, Wu L, Cypowyj S, Rybojad M, Belkadi A, Picard C, Abel L, Fieschi C, Puel A, Li X, Casanova JL
Immunity 2013 Oct 17;39(4):676-86
Immunity 2013 Oct 17;39(4):676-86
Induction and stability of human Th17 cells require endogenous NOS2 and cGMP-dependent NO signaling.
Obermajer N, Wong JL, Edwards RP, Chen K, Scott M, Khader S, Kolls JK, Odunsi K, Billiar TR, Kalinski P
The Journal of experimental medicine 2013 Jul 1;210(7):1433-445
The Journal of experimental medicine 2013 Jul 1;210(7):1433-445
Association of a NOD2 gene polymorphism and T-helper 17 cells with presumed ocular toxoplasmosis.
Dutra MS, Béla SR, Peixoto-Rangel AL, Fakiola M, Cruz AG, Gazzinelli A, Quites HF, Bahia-Oliveira LM, Peixe RG, Campos WR, Higino-Rocha AC, Miller NE, Blackwell JM, Antonelli LR, Gazzinelli RT
The Journal of infectious diseases 2013 Jan 1;207(1):152-63
The Journal of infectious diseases 2013 Jan 1;207(1):152-63
Patients with cystic fibrosis have inducible IL-17+IL-22+ memory cells in lung draining lymph nodes.
Chan YR, Chen K, Duncan SR, Lathrop KL, Latoche JD, Logar AJ, Pociask DA, Wahlberg BJ, Ray P, Ray A, Pilewski JM, Kolls JK
The Journal of allergy and clinical immunology 2013 Apr;131(4):1117-29, 1129.e1-5
The Journal of allergy and clinical immunology 2013 Apr;131(4):1117-29, 1129.e1-5
Frequency of Th17 CD4+ T cells in early rheumatoid arthritis: a marker of anti-CCP seropositivity.
Arroyo-Villa I, Bautista-Caro MB, Balsa A, Aguado-Acín P, Nuño L, Bonilla-Hernán MG, Puig-Kröger A, Martín-Mola E, Miranda-Carús ME
PloS one 2012;7(8):e42189
PloS one 2012;7(8):e42189
Dysregulation of Th17 cells during the early post-transplant period in patients under calcineurin inhibitor based immunosuppression.
Chung BH, Kim KW, Kim BM, Piao SG, Lim SW, Choi BS, Park CW, Kim YS, Cho ML, Yang CW
PloS one 2012;7(7):e42011
PloS one 2012;7(7):e42011
Increased frequencies of Th22 cells as well as Th17 cells in the peripheral blood of patients with ankylosing spondylitis and rheumatoid arthritis.
Zhang L, Li YG, Li YH, Qi L, Liu XG, Yuan CZ, Hu NW, Ma DX, Li ZF, Yang Q, Li W, Li JM
PloS one 2012;7(4):e31000
PloS one 2012;7(4):e31000
The colocalization potential of HIV-specific CD8+ and CD4+ T-cells is mediated by integrin β7 but not CCR6 and regulated by retinoic acid.
Wacleche VS, Chomont N, Gosselin A, Monteiro P, Goupil M, Kared H, Tremblay C, Bernard N, Boulassel MR, Routy JP, Ancuta P
PloS one 2012;7(3):e32964
PloS one 2012;7(3):e32964
Characterization of Th17 responses to Streptococcus pneumoniae in humans: comparisons between adults and children in a developed and a developing country.
Lundgren A, Bhuiyan TR, Novak D, Kaim J, Reske A, Lu YJ, Qadri F, Malley R
Vaccine 2012 Jun 6;30(26):3897-907
Vaccine 2012 Jun 6;30(26):3897-907
Cigarette smoke induction of osteopontin (SPP1) mediates T(H)17 inflammation in human and experimental emphysema.
Shan M, Yuan X, Song LZ, Roberts L, Zarinkamar N, Seryshev A, Zhang Y, Hilsenbeck S, Chang SH, Dong C, Corry DB, Kheradmand F
Science translational medicine 2012 Jan 18;4(117):117ra9
Science translational medicine 2012 Jan 18;4(117):117ra9
Complement component C5a promotes expression of IL-22 and IL-17 from human T cells and its implication in age-related macular degeneration.
Liu B, Wei L, Meyerle C, Tuo J, Sen HN, Li Z, Chakrabarty S, Agron E, Chan CC, Klein ML, Chew E, Ferris F, Nussenblatt RB
Journal of translational medicine 2011 Jul 15;9:1-12
Journal of translational medicine 2011 Jul 15;9:1-12
IL-1β and TGF-β act antagonistically in induction and differentially in propagation of human proinflammatory precursor CD4+ T cells.
Hebel K, Rudolph M, Kosak B, Chang HD, Butzmann J, Brunner-Weinzierl MC
Journal of immunology (Baltimore, Md. : 1950) 2011 Dec 1;187(11):5627-35
Journal of immunology (Baltimore, Md. : 1950) 2011 Dec 1;187(11):5627-35
Memory CCR6+CD4+ T cells are preferential targets for productive HIV type 1 infection regardless of their expression of integrin β7.
Monteiro P, Gosselin A, Wacleche VS, El-Far M, Said EA, Kared H, Grandvaux N, Boulassel MR, Routy JP, Ancuta P
Journal of immunology (Baltimore, Md. : 1950) 2011 Apr 15;186(8):4618-30
Journal of immunology (Baltimore, Md. : 1950) 2011 Apr 15;186(8):4618-30
Generation of IL-23 producing dendritic cells (DCs) by airborne fungi regulates fungal pathogenicity via the induction of T(H)-17 responses.
Chamilos G, Ganguly D, Lande R, Gregorio J, Meller S, Goldman WE, Gilliet M, Kontoyiannis DP
PloS one 2010 Sep 23;5(9):e12955
PloS one 2010 Sep 23;5(9):e12955
Peripheral blood CCR4+CCR6+ and CXCR3+CCR6+CD4+ T cells are highly permissive to HIV-1 infection.
Gosselin A, Monteiro P, Chomont N, Diaz-Griffero F, Said EA, Fonseca S, Wacleche V, El-Far M, Boulassel MR, Routy JP, Sekaly RP, Ancuta P
Journal of immunology (Baltimore, Md. : 1950) 2010 Feb 1;184(3):1604-16
Journal of immunology (Baltimore, Md. : 1950) 2010 Feb 1;184(3):1604-16
Human plasmacytoid dendritic cells support Th17 cell effector function in response to TLR7 ligation.
Yu CF, Peng WM, Oldenburg J, Hoch J, Bieber T, Limmer A, Hartmann G, Barchet W, Eis-Hübinger AM, Novak N
Journal of immunology (Baltimore, Md. : 1950) 2010 Feb 1;184(3):1159-67
Journal of immunology (Baltimore, Md. : 1950) 2010 Feb 1;184(3):1159-67
Interleukin-12 is the optimum cytokine to expand human Th17 cells in vitro.
Nady S, Ignatz-Hoover J, Shata MT
Clinical and vaccine immunology : CVI 2009 Jun;16(6):798-805
Clinical and vaccine immunology : CVI 2009 Jun;16(6):798-805
The effects of trastuzumab on the CD4+CD25+FoxP3+ and CD4+IL17A+ T-cell axis in patients with breast cancer.
Horlock C, Stott B, Dyson PJ, Morishita M, Coombes RC, Savage P, Stebbing J
British journal of cancer 2009 Apr 7;100(7):1061-7
British journal of cancer 2009 Apr 7;100(7):1061-7
Prolonged, granulocyte-macrophage colony-stimulating factor-dependent, neutrophil survival following rheumatoid synovial fibroblast activation by IL-17 and TNFalpha.
Parsonage G, Filer A, Bik M, Hardie D, Lax S, Howlett K, Church LD, Raza K, Wong SH, Trebilcock E, Scheel-Toellner D, Salmon M, Lord JM, Buckley CD
Arthritis research & therapy 2008;10(2):R47
Arthritis research & therapy 2008;10(2):R47
Phenotypic analysis of prostate-infiltrating lymphocytes reveals TH17 and Treg skewing.
Sfanos KS, Bruno TC, Maris CH, Xu L, Thoburn CJ, DeMarzo AM, Meeker AK, Isaacs WB, Drake CG
Clinical cancer research : an official journal of the American Association for Cancer Research 2008 Jun 1;14(11):3254-61
Clinical cancer research : an official journal of the American Association for Cancer Research 2008 Jun 1;14(11):3254-61
Interleukins 1beta and 6 but not transforming growth factor-beta are essential for the differentiation of interleukin 17-producing human T helper cells.
Acosta-Rodriguez EV, Napolitani G, Lanzavecchia A, Sallusto F
Nature immunology 2007 Sep;8(9):942-9
Nature immunology 2007 Sep;8(9):942-9
Distinct regulation of interleukin-17 in human T helper lymphocytes.
Chen Z, Tato CM, Muul L, Laurence A, O'Shea JJ
Arthritis and rheumatism 2007 Sep;56(9):2936-46
Arthritis and rheumatism 2007 Sep;56(9):2936-46
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
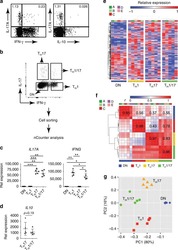
- CD4-enriched human peripheral blood cells were polarized under Th17 conditions (with Human IL-23 Recombinant Protein (Product # 14-8239-63) for 10 days. Cells were restimulated with Protein Transport Inhibitor Cocktail (Product # 00-4980-03) (left) or Cell Stimulation Cocktail (plus protein transport inhibitors) (Product # 00-4975-03) (right) for 6 hours. Cells were intracellular stained with Anti-Human CD4 APC (Product # 17-0049-42) and Anti-Human IL-17A FITC using the Fixation & Permeabilization Buffers (Product # 88-8824-00). Viable cells, as determined by Fixable Viability Dye eFluor® 450 (Product # 65-0863-14), were used for analysis.
- Conjugate
- Yellow dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
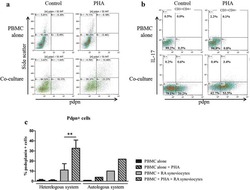
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
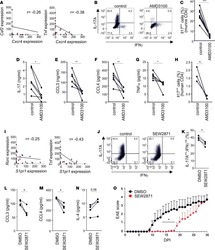
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
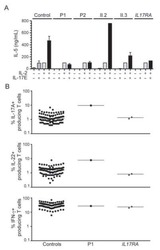
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
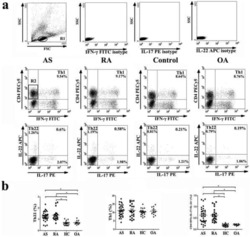
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
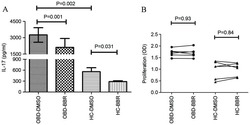
- Figure 1. Effect of BBR on PBMC IL-17 production and cell viability. (A) The expression of IL-17 in PBMCs derived from HC (n=6) and patients with active OBD (n=6) was significantly decreased in the presence of BBR (5 uM) compared with DMSO, as determined by flow cytometry analysis. (B) No significant difference in PBMC cell viability was observed between HC (n=6) and OBD (n=6) groups. Data are presented as the mean +- standard deviation. BBR, berberine; IL-17, interleukin 17; PBMC, peripheral blood mononuclear cells; HC, healthy control; OBD, ocular Behcet's disease; DMSO, dimethyl sulfoxide.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Evaluation of commercial antibodies for the intracellular detection of recombinant bovine and ovine IL-17A. The eight commercial antibodies listed in Table 1 were tested against fixed, permeabilised untransfected (UTF) CHO cells and CHO cells transfected with cDNA encoding bovIL-17A or ovIL-17A for their capacity to detect intracellular recombinant IL-17A by flow cytometry. Results are shown for one polyclonal antibody (pab) produced against bovIL-17A ( A ) and seven monoclonal antibodies (mabs) produced against human or mouse IL-17A ( B - D ). Profiles of the relevant control antibodies listed in Table 2 are included in the overlapping histograms. Events were acquired on the MacsQuant according to the gating strategy described previously (in brief) and shown in Additional file 2 . Line colours representing different antibody treatments are given in parentheses: A Primary rabbit anti-bovine IL-17A pab PB0274B-100 at 1 mug/mL (A.1, red) or negative control primary anti-bovine CD34 pab (in-house) at an estimated 1 mug/mL equivalent (a, black) then detected with a secondary goat anti-rabbit alexafluor 488 at 1 mug/mL; B Directly conjugated mouse anti-human IL-17A eBio64DEC17-phycoerythrin (PE) mab (IgG1) at 2.5 mug/mL (B.1, red) and control IgG1 VPM21 mab (in-house) at an estimated 2.5 mug/mL equivalent (b, black) and detected with goat anti-mouse PE at 1 mug/mL; C Primary mouse anti-human IL-17A mabs MT44.6 (C.1, blue), MT241 (C.2, green), MT2770 (C.3, brown) and MT504
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
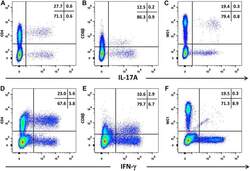
- Figure 5 Intracellular expression of IL-17A and IFN-gamma by activated bovine T cell subsets. PBMC from four cattle were stimulated with phorbol 12-myristate 13 acetate, ionomycin and brefeldin A in RPMI culture medium for 4 h. Cells were harvested and stained for viability and with mabs specific for cell-surface phenotypic markers and intracellular cytokines as described in Table 3 and "" Expression of intracellular IL-17A and IFN-gamma by bovine and ovine T cell subsets section "". Cells were stained for CD4 with mab CC8-PE at 1:20 dilution ( A , D ), for CD8beta with mab CC58-PE at 1:20 dilution ( B , D ) and for WC-1 (gammadelta T cells) with mab CC15-PE at 1:200 ( C , E ). Intracellular cytokine staining for IL-17A was conducted using mab eBioDEC17-APC at a 1:20 dilution ( A - C ) and for IFN-gamma using mab CC302-Alexafluor 647 at a 1:200 dilution ( D - F ). Data are shown for PBMC from one representative animal of four.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
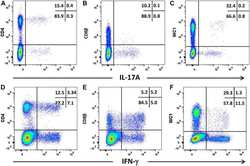
- Figure 6 Intracellular expression of IL-17A and IFN-gamma by activated ovine T cell subsets. PBMC from four sheep were stimulated with phorbol 12-myristate 13 acetate, ionomycin and brefeldin A in RPMI culture medium for 4 h. Cells were harvested and stained for viability and with mabs specific for cell-surface phenotypic markers and intracellular cytokines as described in Table 3 and "" Expression of intracellular IL-17A and IFN-gamma by bovine and ovine T cell subsets section "". Cells were then stained for CD4 with mab 44.38-PE at 1:20 dilution ( A , D ), CD8beta with mab CC58-PE at 1:20 dilution ( B , D ) and WC-1 (gammadelta) with mab CC15-PE at 1:200 ( C , E ). Intracellular cytokine staining for IL-17A was conducted using mab eBio64DEC17-APC a 1:20 dilution ( A - C ) and for IFN-gamma using mab CC302-alexafluor 647 at a 1:200 dilution ( D - F ). Data shown is for one representative animal out of four.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Additional file 2. Gating strategy used for the evaluation of commercial antibodies to bind intracellular recombinant bovine and ovine IL-17A in fixed cells. Cells were acquired for flow cytometric analyses using the MacsQuant flow cytometer and analysed using the MacsQuantify Software. 20 000-50 000 events were collected and the following gating strategy was followed. Cells in the plot of Forward Scatter-Area (FSC-A) against the high dynamic range over time (HDR-T) are gated in P1 to exclude any non-specific artefacts ( A ). The P1/P2 gate represents Side Scatter-Area (SSC-A) plotted against FSC-A set to identify the main cell population and exclude debris ( B ). Single cells were gated (P1/P2/P3) using FSC-Height (H) vs FSC-A for doublet discrimination ( C ). Finally, the cells of interest were identified in the phycoerythrin or alexafluor 488 channel vs SSC-A (P1/P2/P3/P4) where regions were set using the isotype or equivalent control for each CHO cell line to establish threshold gates ( D ). Overlaying histogram plots of phycoerythrin or alexafluor 488 using (P1/P2/P3) gating strategy selecting for all cells in the region (equivalent to cells above and below region boundary in plot D ) ( E ) were used to compare anti-IL-17A antibodies with appropriate isotype or equivalent controls presented in Figure 4 . Gated percentage numbers above the region boundary (P1/P2/P3/P4) and median fluorescence region values (P1/P2/P3) were measured for each antibody in the relevant fluoroc
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
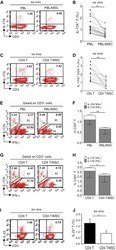
- Figure 1 Multipotent Human Mesenchymal Stromal Cells (hMSCs) Suppress Th17 Responses (A-D) Human peripheral blood CD3 + leukocytes (PBLs) (A, representative data; B, pooled data of 17 PBL donors co-cultured with all three hMSC donors) or CD3 + CD4 T cells (C, representative data; D, pooled data of 11 PBL donors co-cultured with all three hMSC donors) were co-cultured without (left) or with (right) hMSCs ex vivo, followed by PMA/ionomycin stimulation for 6 hr. (E-H) IL-17A production in ex-vivo-cultured CD3 + T cells was assessed by intracellular staining. IL-17A and IFN-gamma production in CD3 + PBLs (E, representative data; F, pooled data) or CD3 + CD4 T cells (G, representative data; H, pooled data) without and with co-culture of hMSCs was analyzed by flow cytometry. Representative intracellular staining is shown for IL-17A + IFN-gamma - - CD3 + T cells (R3 region) and IL-17A + IFN-gamma + (R5 region) CD3 + T cells, and pooled data from PBLs (n = 4) or CD4 T cells (n = 4) co-cultured with two hMSC donors (donors A and B) are provided in (F) and (H), respectively. Gray bars represent the percentages of IL-17A + IFN-gamma - - CD3 + T cells, whereas white bars represent the percentages of IL-17A + IFN-gamma + T cells. (I and J) IL-22 production in four donors of CD3 + CD4 T cells (I, representative data; J, pooled data) without and with co-culture of two donors of hMSCs (donors A and B) was assessed by intracellular staining. Cell percentages are denoted in the dotplot quadran
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
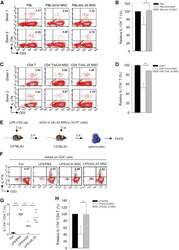
- Figure 3 IL-25 Silencing in hMSCs Reverses Th17 Responses In Vitro and In Vivo (A-D) Freshly isolated human PBLs (A) or CD4 T cells (C) were co-cultured without (left) or with either siCtrl hMSCs (middle) or siIL-25 hMSCs (right) for 3 days, followed by PMA/ionomycin stimulation for 6 hr. IL-17A production in CD3 + T cells was assessed by intracellular staining. Numbers in the top right quadrants represent the percentages of IL-17A-producing CD3 + T cells. Pooled data from PBLs (n = 3) or CD4 T cells (n = 3) and two hMSC donors (donors A and B) are provided in (B) and (D), respectively. Data are shown as mean +- SD. * p < 0.05, ** p < 0.01. (E) Experimental strategy for establishing in vivo inflammatory conditions in wild-type C57BL/6J mice with expansion of Th17 cells and adoptive transfer of hMSCs is shown. (F) On day 3 after LPS (100 mug/mouse) challenge, IL-17A production in activated CD4 T cells in splenocytes from control mice, PBS-treated mice, siCtrl-hMSC-treated mice, or siIL-25-hMSC-treated mice was assessed by intracellular staining. (G and H) Calculated (G) and relative (H) mean percentage of IL-17A-expressing CD4 T cells among control mice, PBS-treated mice, siCtrl-hMSC-treated mice, or siIL-25-hMSC-treated mice (n = 6). Data are shown as mean +- SD. * p < 0.05, ** p < 0.01, *** p < 0.005.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
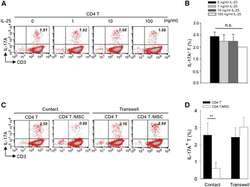
- Figure 4 Exogenous IL-25 Alone Is Insufficient to Significantly Suppress Th17 Responses, with Cell Contact Required as well for hMSC-Mediated Inhibition of Th17 Responses (A) Human CD4 T cells were treated with indicated doses of rhIL-25 for 18 hr, followed by PMA/ionomycin stimulation for 6 hr. IL-17A production in CD3 + T cells was assessed by intracellular staining. Numbers in the top right quadrants represent the percentages of IL-17A-producing CD3 + T cells. (B) Pooled data of five PBL donors are shown. (C) Human CD4 T cells (n = 4) were co-cultured without or with hMSCs (two donors, B and C) in the absence or presence of transwell barriers. (D) Pooled data from healthy donors are shown. Data are shown as mean +- SD. ** p < 0.01; n.s., not significant.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
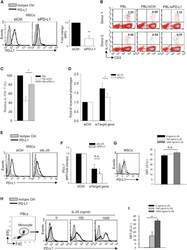
- Figure 5 IL-25 Induces PD-L1 Surface Expression on hMSCs and Human Monocytes (A) PD-L1 in siCtrl MSCs (left) and siPD-L1 MSCs (right) was analyzed by surface staining. (B) Freshly isolated human PBLs were co-cultured without (left) or with siCtrl MSCs (middle) or siPD-L1 MSCs (right) for 3 days, followed by PMA/ionomycin stimulation for 6 hr. IL-17A production in CD3 + T cells was assessed by intracellular staining. Representative data are shown with numbers in the top right quadrants representing the percentages of IL-17A-producing CD3 + T cells. (C) Pooled data from PBLs (n = 4) and two hMSC donors (donors A and B) are shown. (D) Folds of reversed phenotypes of siIL-25 and siPD-L1 are shown. (E) PD-L1 expression on siCtrl hMSCs (left) and siIL-25 hMSCs (right) was assessed by cell surface staining. Filled histograms represent isotype control; unfilled histograms represent PD-L1 antibody staining. (F) Pooled data of PD-L1 expression (indicated by fold change in MFI) on siIL-25 hMSCs and siPD-L1 hMSCs (all three donors) are shown. PD-L1 expression levels were compared between hMSCs silenced for the target gene (IL-25 or PD-L1) and the respective siCtrl. (G) hMSCs were treated with the indicated doses of rhIL-25 for 18 hr and assessed for cell surface PD-L1 expression by cell surface staining. Pooled data (all three donors) are shown in chart to the right with bars representing MFI. (H) Human PBLs were treated with the indicated doses of rhIL-25 for 18 hr and assessed for cell
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
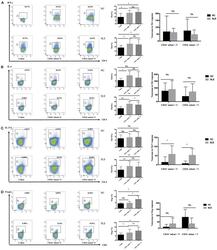
- Experimental details
- Figure 4 CD16 + monocytes promoted T-cell-mediated inflammation in SLE . CD16 + or CD16 - monocytes were cocultured with CD4 + T cells isolated from freshly collected SLE blood or blood bank collected HC blood buffy coat for 5 days in the presence of anti-CD3 (1 mug/mL) and anti-CD28 (1 mug/mL) antibodies and M-CSF (50 ng/mL). Intracellular IFN-gamma, IL-4, and IL-17A expression was detected by flow cytometry after PMA (50 ng/mL), ionomycin (1 mug/mL), and BFA (10 mug/mL) stimulation for 5 h on day 5. The percentage of Treg (CD4 + Foxp3 + ) was also analyzed. Representative pseudocolor dots depicted Th1 (A) , Th2 (B) , Th17 (C) , and Treg (D) frequencies in CD4 + T cells after coculture with each monocyte subset from one patient and one control donor. The proportion of Th1, Th2, Th17, and Treg cells was calculated after coculture of CD4 + T cells with each monocyte. The percentage increases in T-cell subsets in cocultures of monocytes and T cells compared with CD4 + T cells cultured alone were compared between 7 healthy individuals and 10 patients with SLE. Data were expressed as mean +- SD and analyzed by non-parametric paired t test and Mann-Whitney U test. * P < 0.05, ** P < 0.01; NS, no significance.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
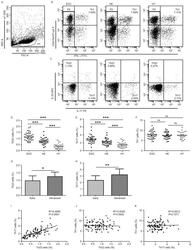
- Experimental details
- Figure 1. Flow cytometric analysis was used to determine the distribution of Th22, Th17 and Th1 cells in EGC, HE and HY ( Fig. 1 ). Flow cytometric analysis of Th22, Th17 and Th1 cells in peripheral whole blood from EGC (n=39), HE (n=32) and HY (n=31). (A) Lymphocytes were gated in P1 using flow cytometry. CD4 + IFN-gamma - lymphocytes were gated in P2 using flow cytometry, and representative results of flow cytometric analyses for (B) Th1 (CD4 + IFN-gamma + ), (C) Th22 (CD4 + IFN-gamma - IL-17 - IL-22 + ) and Th17 (CD4 + IFN-gamma - IL-17 + IL-22 - ) cells in the three groups of subjects are presented. The number of cells stained in EGC, HE and HY in P2 were 2,654, 4,696 and 5,185, respectively. The proportion of (D) Th22, (E) Th17 and (F) Th1 cells in the three groups of subjects. The proportion of (G) Th22 and (H) Th17 cells in peripheral whole blood derived from patients with early (n=13) or advanced (n=26) gastric cancer. The association between the proportion of (I) Th22 and Th17 cells, (J) Th22 and Th1 cells, and (K) Th17 and Th1 cells, in peripheral whole blood of all subjects. *P
- Conjugate
- Yellow dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry