Antibody data
- Antibody Data
- Antigen structure
- References [38]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [18]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 17-7179-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IL-17A Monoclonal Antibody (eBio64DEC17), APC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The eBio64DEC17 antibody reacts with human IL-17A. The eBio64DEC17 antibody is a neutralizing antibody. Interleukin-17A (IL-17A) is a CD4+ T cell-derived cytokine that promotes inflammatory responses in cell lines and is elevated in rheumatoid arthritis, asthma, multiple sclerosis, psoriasis, and transplant rejection. The cDNA encoding human IL-17A was isolated from a library of CD4+ T cells; the encoded protein exhibits 72 percent amino acid identity with HVS13 , an open reading frame from a T lymphotropic Herpesvirus saimiri, and 63 percent with mouse CTLA-8 (cytotoxic T-lymphocyte associated antigen-8). Human IL-17A exists as glycosylated 20-30 kD homodimers. High levels of IL-17A homodimer are produced by activated peripheral blood CD4+ T-cells. IL-17A enhances expression of the intracellular adhesion molecule-1 (ICAM-1) in human fibroblasts. Human IL-17A also stimulates epithelial, endothelial, or fibroblastic cells to secrete IL-6, IL-8, G-CSF, and PGE2. In the presence of human IL-17A, fibroblasts can sustain the proliferation of CD34+ hematopoietic progenitors and induce maturation into neutrophils. Mouse, rat, and human IL-17A can induce IL-6 secretion in mouse stromal cells, indicating that all homologs can recognize the mouse IL-17A receptor. IL-23-dependent, IL-17A-producing CD4+ T cells (Th-17 cells) have been identified as a unique subset of Th cells that develops along a pathway that is distinct from the Th1- and Th2- cell differentiation pathways. The hallmark effector molecules of Th1 and Th2 cells, e.g., IFN gamma and IL-4, have each been found to negatively regulate the generation of these Th-17 cells. Intracellular staining by eBio64DEC17 antibody identifies the same cell population as the eBio64CAP17 antibody, as can be seen in co-staining experiments using both antibodies. Applications Reported: This eBio64DEC17 antibody has been reported for use in intracellular staining followed by flow cytometric analysis. Applications Tested: This eBio64DEC17 antibody has been pre-titrated and tested by intracellular staining and flow cytometric analysis of stimulated normal human peripheral blood cells. This can be used at 5 µL (0.06 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 633-647 nm; Emission: 660 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- eBio64DEC17
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Chronic morphine administration differentially modulates viral reservoirs in SIVmac251 infected rhesus macaque model.
IFN-γ promoted exosomes from mesenchymal stem cells to attenuate colitis via miR-125a and miR-125b.
Tumors induce de novo steroid biosynthesis in T cells to evade immunity.
Respiratory Syncytial Virus Infection Reduces Kynurenic Acid Production and Reverses Th17/Treg Balance by Modulating Indoleamine 2,3-Dioxygenase (IDO) Molecules in Plasmacytoid Dendritic Cells.
Single-cell RNA sequencing identifies shared differentiation paths of mouse thymic innate T cells.
Interleukin-24 Regulates T Cell Activity in Patients With Colorectal Adenocarcinoma.
Two phase kinetics of the inflammatory response from hepatocyte-peripheral blood mononuclear cell interactions.
CD4 T cell loss and Th2 and Th17 bias are associated with the severity of severe fever with thrombocytopenia syndrome (SFTS).
Accumulation of T-helper 22 cells, interleukin-22 and myeloid-derived suppressor cells promotes gastric cancer progression in elderly patients.
Azacytidine prevents experimental xenogeneic graft-versus-host disease without abrogating graft-versus-leukemia effects.
Transcriptional signature of human pro-inflammatory T(H)17 cells identifies reduced IL10 gene expression in multiple sclerosis.
Antigen-Presenting Human γδ T Cells Promote Intestinal CD4(+) T Cell Expression of IL-22 and Mucosal Release of Calprotectin.
High frequency of intestinal T(H)17 cells correlates with microbiota alterations and disease activity in multiple sclerosis.
The microvascular niche instructs T cells in large vessel vasculitis via the VEGF-Jagged1-Notch pathway.
Berberine exerts an anti-inflammatory role in ocular Behcet's disease.
Single-cell profiling reveals GPCR heterogeneity and functional patterning during neuroinflammation.
Enhancing the toolbox to study IL-17A in cattle and sheep.
CD16(+) Monocyte Subset Was Enriched and Functionally Exacerbated in Driving T-Cell Activation and B-Cell Response in Systemic Lupus Erythematosus.
Interaction among activated lymphocytes and mesenchymal cells through podoplanin is critical for a high IL-17 secretion.
Reduced pro-inflammatory profile of γδT cells in pregnant patients with rheumatoid arthritis.
The Clinical Relevance of IL-17-Producing CD4+CD161+ Cell and Its Subpopulations in Primary Sjögren's Syndrome.
Administration of Panobinostat Is Associated with Increased IL-17A mRNA in the Intestinal Epithelium of HIV-1 Patients.
Polycyclic aromatic hydrocarbons reciprocally regulate IL-22 and IL-17 cytokines in peripheral blood mononuclear cells from both healthy and asthmatic subjects.
Interleukin-25 Mediates Transcriptional Control of PD-L1 via STAT3 in Multipotent Human Mesenchymal Stromal Cells (hMSCs) to Suppress Th17 Responses.
Th17-related cytokines contribute to recall-like expansion/effector function of HMBPP-specific Vγ2Vδ2 T cells after Mycobacterium tuberculosis infection or vaccination.
Vitamin D up-regulates the vitamin D receptor by protecting it from proteasomal degradation in human CD4+ T cells.
Hypercytotoxicity and rapid loss of NKp44+ innate lymphoid cells during acute SIV infection.
Elevated levels of CD4(+)CD25(+)FoxP3(+) T cells in systemic sclerosis patients contribute to the secretion of IL-17 and immunosuppression dysfunction.
An ACT1 mutation selectively abolishes interleukin-17 responses in humans with chronic mucocutaneous candidiasis.
IL-21 regulates the differentiation of a human γδ T cell subset equipped with B cell helper activity.
Tumour cell generation of inducible regulatory T-cells in multiple myeloma is contact-dependent and antigen-presenting cell-independent.
Increased frequencies of Th22 cells as well as Th17 cells in the peripheral blood of patients with ankylosing spondylitis and rheumatoid arthritis.
Peripherally circulating CD4⁺ FOXP3⁺ CXCR3⁺ T regulatory cells correlate with renal allograft function.
Differentiation, phenotype, and function of interleukin-17-producing human Vγ9Vδ2 T cells.
Prostaglandin E2 and IL-23 plus IL-1β differentially regulate the Th1/Th17 immune response of human CD161(+) CD4(+) memory T cells.
Interleukin-12 is the optimum cytokine to expand human Th17 cells in vitro.
Interleukins 1beta and 6 but not transforming growth factor-beta are essential for the differentiation of interleukin 17-producing human T helper cells.
Distinct regulation of interleukin-17 in human T helper lymphocytes.
Acharya A, Olwenyi OA, Thurman M, Pandey K, Morsey BM, Lamberty B, Ferguson N, Callen S, Fang Q, Buch SJ, Fox HS, Byrareddy SN
Journal of virology 2021 Mar 1;95(5)
Journal of virology 2021 Mar 1;95(5)
IFN-γ promoted exosomes from mesenchymal stem cells to attenuate colitis via miR-125a and miR-125b.
Yang R, Huang H, Cui S, Zhou Y, Zhang T, Zhou Y
Cell death & disease 2020 Jul 30;11(7):603
Cell death & disease 2020 Jul 30;11(7):603
Tumors induce de novo steroid biosynthesis in T cells to evade immunity.
Mahata B, Pramanik J, van der Weyden L, Polanski K, Kar G, Riedel A, Chen X, Fonseca NA, Kundu K, Campos LS, Ryder E, Duddy G, Walczak I, Okkenhaug K, Adams DJ, Shields JD, Teichmann SA
Nature communications 2020 Jul 17;11(1):3588
Nature communications 2020 Jul 17;11(1):3588
Respiratory Syncytial Virus Infection Reduces Kynurenic Acid Production and Reverses Th17/Treg Balance by Modulating Indoleamine 2,3-Dioxygenase (IDO) Molecules in Plasmacytoid Dendritic Cells.
Jin L, Hu Q, Hu Y, Chen Z, Liao W
Medical science monitor : international medical journal of experimental and clinical research 2020 Dec 2;26:e926763
Medical science monitor : international medical journal of experimental and clinical research 2020 Dec 2;26:e926763
Single-cell RNA sequencing identifies shared differentiation paths of mouse thymic innate T cells.
Lee M, Lee E, Han SK, Choi YH, Kwon DI, Choi H, Lee K, Park ES, Rha MS, Joo DJ, Shin EC, Kim S, Kim JK, Lee YJ
Nature communications 2020 Aug 31;11(1):4367
Nature communications 2020 Aug 31;11(1):4367
Interleukin-24 Regulates T Cell Activity in Patients With Colorectal Adenocarcinoma.
Zhang Y, Liu Y, Xu Y
Frontiers in oncology 2019;9:1401
Frontiers in oncology 2019;9:1401
Two phase kinetics of the inflammatory response from hepatocyte-peripheral blood mononuclear cell interactions.
Beringer A, Molle J, Bartosch B, Miossec P
Scientific reports 2019 Jun 10;9(1):8378
Scientific reports 2019 Jun 10;9(1):8378
CD4 T cell loss and Th2 and Th17 bias are associated with the severity of severe fever with thrombocytopenia syndrome (SFTS).
Li MM, Zhang WJ, Weng XF, Li MY, Liu J, Xiong Y, Xiong SE, Zou CC, Wang H, Lu MJ, Yang DL, Peng C, Zheng X
Clinical immunology (Orlando, Fla.) 2018 Oct;195:8-17
Clinical immunology (Orlando, Fla.) 2018 Oct;195:8-17
Accumulation of T-helper 22 cells, interleukin-22 and myeloid-derived suppressor cells promotes gastric cancer progression in elderly patients.
Chen X, Wang Y, Wang J, Wen J, Jia X, Wang X, Zhang H
Oncology letters 2018 Jul;16(1):253-261
Oncology letters 2018 Jul;16(1):253-261
Azacytidine prevents experimental xenogeneic graft-versus-host disease without abrogating graft-versus-leukemia effects.
Ehx G, Fransolet G, de Leval L, D'Hondt S, Lucas S, Hannon M, Delens L, Dubois S, Drion P, Beguin Y, Humblet-Baron S, Baron F
Oncoimmunology 2017;6(5):e1314425
Oncoimmunology 2017;6(5):e1314425
Transcriptional signature of human pro-inflammatory T(H)17 cells identifies reduced IL10 gene expression in multiple sclerosis.
Hu D, Notarbartolo S, Croonenborghs T, Patel B, Cialic R, Yang TH, Aschenbrenner D, Andersson KM, Gattorno M, Pham M, Kivisakk P, Pierre IV, Lee Y, Kiani K, Bokarewa M, Tjon E, Pochet N, Sallusto F, Kuchroo VK, Weiner HL
Nature communications 2017 Nov 17;8(1):1600
Nature communications 2017 Nov 17;8(1):1600
Antigen-Presenting Human γδ T Cells Promote Intestinal CD4(+) T Cell Expression of IL-22 and Mucosal Release of Calprotectin.
Tyler CJ, McCarthy NE, Lindsay JO, Stagg AJ, Moser B, Eberl M
Journal of immunology (Baltimore, Md. : 1950) 2017 May 1;198(9):3417-3425
Journal of immunology (Baltimore, Md. : 1950) 2017 May 1;198(9):3417-3425
High frequency of intestinal T(H)17 cells correlates with microbiota alterations and disease activity in multiple sclerosis.
Cosorich I, Dalla-Costa G, Sorini C, Ferrarese R, Messina MJ, Dolpady J, Radice E, Mariani A, Testoni PA, Canducci F, Comi G, Martinelli V, Falcone M
Science advances 2017 Jul;3(7):e1700492
Science advances 2017 Jul;3(7):e1700492
The microvascular niche instructs T cells in large vessel vasculitis via the VEGF-Jagged1-Notch pathway.
Wen Z, Shen Y, Berry G, Shahram F, Li Y, Watanabe R, Liao YJ, Goronzy JJ, Weyand CM
Science translational medicine 2017 Jul 19;9(399)
Science translational medicine 2017 Jul 19;9(399)
Berberine exerts an anti-inflammatory role in ocular Behcet's disease.
Yang Y, Wang Q, Xie M, Liu P, Qi X, Liu X, Li Z
Molecular medicine reports 2017 Jan;15(1):97-102
Molecular medicine reports 2017 Jan;15(1):97-102
Single-cell profiling reveals GPCR heterogeneity and functional patterning during neuroinflammation.
Tischner D, Grimm M, Kaur H, Staudenraus D, Carvalho J, Looso M, Günther S, Wanke F, Moos S, Siller N, Breuer J, Schwab N, Zipp F, Waisman A, Kurschus FC, Offermanns S, Wettschureck N
JCI insight 2017 Aug 3;2(15)
JCI insight 2017 Aug 3;2(15)
Enhancing the toolbox to study IL-17A in cattle and sheep.
Wattegedera SR, Corripio-Miyar Y, Pang Y, Frew D, McNeilly TN, Palarea-Albaladejo J, McInnes CJ, Hope JC, Glass EJ, Entrican G
Veterinary research 2017 Apr 8;48(1):20
Veterinary research 2017 Apr 8;48(1):20
CD16(+) Monocyte Subset Was Enriched and Functionally Exacerbated in Driving T-Cell Activation and B-Cell Response in Systemic Lupus Erythematosus.
Zhu H, Hu F, Sun X, Zhang X, Zhu L, Liu X, Li X, Xu L, Shi L, Gan Y, Su Y
Frontiers in immunology 2016;7:512
Frontiers in immunology 2016;7:512
Interaction among activated lymphocytes and mesenchymal cells through podoplanin is critical for a high IL-17 secretion.
Noack M, Ndongo-Thiam N, Miossec P
Arthritis research & therapy 2016 Jun 23;18:148
Arthritis research & therapy 2016 Jun 23;18:148
Reduced pro-inflammatory profile of γδT cells in pregnant patients with rheumatoid arthritis.
Tham M, Schlör GR, Yerly D, Mueller C, Surbek D, Villiger PM, Förger F
Arthritis research & therapy 2016 Jan 22;18:26
Arthritis research & therapy 2016 Jan 22;18:26
The Clinical Relevance of IL-17-Producing CD4+CD161+ Cell and Its Subpopulations in Primary Sjögren's Syndrome.
Li L, He J, Zhu L, Yang Y, Jin Y, Jia R, Liu X, Liu Y, Sun X, Li Z
Journal of immunology research 2015;2015:307453
Journal of immunology research 2015;2015:307453
Administration of Panobinostat Is Associated with Increased IL-17A mRNA in the Intestinal Epithelium of HIV-1 Patients.
Bjerg Christensen A, Dige A, Vad-Nielsen J, Brinkmann CR, Bendix M, Østergaard L, Tolstrup M, Søgaard OS, Rasmussen TA, Randel Nyengaard J, Agnholt J, Denton PW
Mediators of inflammation 2015;2015:120605
Mediators of inflammation 2015;2015:120605
Polycyclic aromatic hydrocarbons reciprocally regulate IL-22 and IL-17 cytokines in peripheral blood mononuclear cells from both healthy and asthmatic subjects.
Plé C, Fan Y, Ait Yahia S, Vorng H, Everaere L, Chenivesse C, Balsamelli J, Azzaoui I, de Nadai P, Wallaert B, Lazennec G, Tsicopoulos A
PloS one 2015;10(4):e0122372
PloS one 2015;10(4):e0122372
Interleukin-25 Mediates Transcriptional Control of PD-L1 via STAT3 in Multipotent Human Mesenchymal Stromal Cells (hMSCs) to Suppress Th17 Responses.
Wang WB, Yen ML, Liu KJ, Hsu PJ, Lin MH, Chen PM, Sudhir PR, Chen CH, Chen CH, Sytwu HK, Yen BL
Stem cell reports 2015 Sep 8;5(3):392-404
Stem cell reports 2015 Sep 8;5(3):392-404
Th17-related cytokines contribute to recall-like expansion/effector function of HMBPP-specific Vγ2Vδ2 T cells after Mycobacterium tuberculosis infection or vaccination.
Shen H, Wang Y, Chen CY, Frencher J, Huang D, Yang E, Ryan-Payseur B, Chen ZW
European journal of immunology 2015 Feb;45(2):442-51
European journal of immunology 2015 Feb;45(2):442-51
Vitamin D up-regulates the vitamin D receptor by protecting it from proteasomal degradation in human CD4+ T cells.
Kongsbak M, von Essen MR, Boding L, Levring TB, Schjerling P, Lauritsen JP, Woetmann A, Ødum N, Bonefeld CM, Geisler C
PloS one 2014;9(5):e96695
PloS one 2014;9(5):e96695
Hypercytotoxicity and rapid loss of NKp44+ innate lymphoid cells during acute SIV infection.
Li H, Richert-Spuhler LE, Evans TI, Gillis J, Connole M, Estes JD, Keele BF, Klatt NR, Reeves RK
PLoS pathogens 2014 Dec;10(12):e1004551
PLoS pathogens 2014 Dec;10(12):e1004551
Elevated levels of CD4(+)CD25(+)FoxP3(+) T cells in systemic sclerosis patients contribute to the secretion of IL-17 and immunosuppression dysfunction.
Liu X, Gao N, Li M, Xu D, Hou Y, Wang Q, Zhang G, Sun Q, Zhang H, Zeng X
PloS one 2013;8(6):e64531
PloS one 2013;8(6):e64531
An ACT1 mutation selectively abolishes interleukin-17 responses in humans with chronic mucocutaneous candidiasis.
Boisson B, Wang C, Pedergnana V, Wu L, Cypowyj S, Rybojad M, Belkadi A, Picard C, Abel L, Fieschi C, Puel A, Li X, Casanova JL
Immunity 2013 Oct 17;39(4):676-86
Immunity 2013 Oct 17;39(4):676-86
IL-21 regulates the differentiation of a human γδ T cell subset equipped with B cell helper activity.
Caccamo N, Todaro M, La Manna MP, Sireci G, Stassi G, Dieli F
PloS one 2012;7(7):e41940
PloS one 2012;7(7):e41940
Tumour cell generation of inducible regulatory T-cells in multiple myeloma is contact-dependent and antigen-presenting cell-independent.
Feyler S, Scott GB, Parrish C, Jarmin S, Evans P, Short M, McKinley K, Selby PJ, Cook G
PloS one 2012;7(5):e35981
PloS one 2012;7(5):e35981
Increased frequencies of Th22 cells as well as Th17 cells in the peripheral blood of patients with ankylosing spondylitis and rheumatoid arthritis.
Zhang L, Li YG, Li YH, Qi L, Liu XG, Yuan CZ, Hu NW, Ma DX, Li ZF, Yang Q, Li W, Li JM
PloS one 2012;7(4):e31000
PloS one 2012;7(4):e31000
Peripherally circulating CD4⁺ FOXP3⁺ CXCR3⁺ T regulatory cells correlate with renal allograft function.
Hoerning A, Köhler S, Jun C, Tebbe B, Fu J, Menke J, Wilde B, Dolff S, Feldkamp T, Briscoe DM, Kribben A, Hoyer PF, Witzke O
Scandinavian journal of immunology 2012 Sep;76(3):320-8
Scandinavian journal of immunology 2012 Sep;76(3):320-8
Differentiation, phenotype, and function of interleukin-17-producing human Vγ9Vδ2 T cells.
Caccamo N, La Mendola C, Orlando V, Meraviglia S, Todaro M, Stassi G, Sireci G, Fournié JJ, Dieli F
Blood 2011 Jul 7;118(1):129-38
Blood 2011 Jul 7;118(1):129-38
Prostaglandin E2 and IL-23 plus IL-1β differentially regulate the Th1/Th17 immune response of human CD161(+) CD4(+) memory T cells.
Barrie A, Khare A, Henkel M, Zhang Y, Barmada MM, Duerr R, Ray A
Clinical and translational science 2011 Aug;4(4):268-73
Clinical and translational science 2011 Aug;4(4):268-73
Interleukin-12 is the optimum cytokine to expand human Th17 cells in vitro.
Nady S, Ignatz-Hoover J, Shata MT
Clinical and vaccine immunology : CVI 2009 Jun;16(6):798-805
Clinical and vaccine immunology : CVI 2009 Jun;16(6):798-805
Interleukins 1beta and 6 but not transforming growth factor-beta are essential for the differentiation of interleukin 17-producing human T helper cells.
Acosta-Rodriguez EV, Napolitani G, Lanzavecchia A, Sallusto F
Nature immunology 2007 Sep;8(9):942-9
Nature immunology 2007 Sep;8(9):942-9
Distinct regulation of interleukin-17 in human T helper lymphocytes.
Chen Z, Tato CM, Muul L, Laurence A, O'Shea JJ
Arthritis and rheumatism 2007 Sep;56(9):2936-46
Arthritis and rheumatism 2007 Sep;56(9):2936-46
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
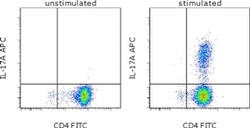
- Experimental details
- CD4-enriched human peripheral blood cells were polarized under Th17 conditions (with Human IL-23 Recombinant Protein (Product # 14-8239-63) for 10 days. Cells were restimulated with Protein Transport Inhibitor Cocktail (Product # 00-4980-03) (left) or Cell Stimulation Cocktail plus protein transport inhibitors (Product # 00-4975-03) (right) for 6 hours. Cells were intracellularly stained with Anti-Human CD4 FITC (Product # 11-0047-42) and Anti-Human IL-17A APC using the Intracellular Fixation & Permeabilization Buffer Set (Product # 88-8824-00). Viable cells, as determined by Fixable Viability Dye eFluor® 450 (Product # 65-0863-14), were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
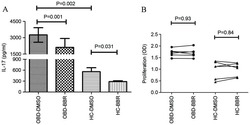
- Figure 1. Effect of BBR on PBMC IL-17 production and cell viability. (A) The expression of IL-17 in PBMCs derived from HC (n=6) and patients with active OBD (n=6) was significantly decreased in the presence of BBR (5 uM) compared with DMSO, as determined by flow cytometry analysis. (B) No significant difference in PBMC cell viability was observed between HC (n=6) and OBD (n=6) groups. Data are presented as the mean +- standard deviation. BBR, berberine; IL-17, interleukin 17; PBMC, peripheral blood mononuclear cells; HC, healthy control; OBD, ocular Behcet's disease; DMSO, dimethyl sulfoxide.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
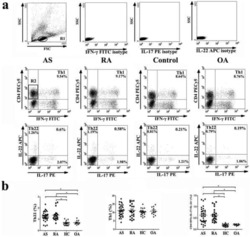
- Figure 4 Evaluation of commercial antibodies for the intracellular detection of recombinant bovine and ovine IL-17A. The eight commercial antibodies listed in Table 1 were tested against fixed, permeabilised untransfected (UTF) CHO cells and CHO cells transfected with cDNA encoding bovIL-17A or ovIL-17A for their capacity to detect intracellular recombinant IL-17A by flow cytometry. Results are shown for one polyclonal antibody (pab) produced against bovIL-17A ( A ) and seven monoclonal antibodies (mabs) produced against human or mouse IL-17A ( B - D ). Profiles of the relevant control antibodies listed in Table 2 are included in the overlapping histograms. Events were acquired on the MacsQuant according to the gating strategy described previously (in brief) and shown in Additional file 2 . Line colours representing different antibody treatments are given in parentheses: A Primary rabbit anti-bovine IL-17A pab PB0274B-100 at 1 mug/mL (A.1, red) or negative control primary anti-bovine CD34 pab (in-house) at an estimated 1 mug/mL equivalent (a, black) then detected with a secondary goat anti-rabbit alexafluor 488 at 1 mug/mL; B Directly conjugated mouse anti-human IL-17A eBio64DEC17-phycoerythrin (PE) mab (IgG1) at 2.5 mug/mL (B.1, red) and control IgG1 VPM21 mab (in-house) at an estimated 2.5 mug/mL equivalent (b, black) and detected with goat anti-mouse PE at 1 mug/mL; C Primary mouse anti-human IL-17A mabs MT44.6 (C.1, blue), MT241 (C.2, green), MT2770 (C.3, brown) and MT504
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
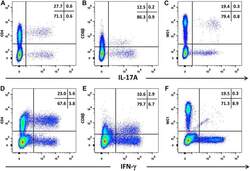
- Figure 5 Intracellular expression of IL-17A and IFN-gamma by activated bovine T cell subsets. PBMC from four cattle were stimulated with phorbol 12-myristate 13 acetate, ionomycin and brefeldin A in RPMI culture medium for 4 h. Cells were harvested and stained for viability and with mabs specific for cell-surface phenotypic markers and intracellular cytokines as described in Table 3 and "" Expression of intracellular IL-17A and IFN-gamma by bovine and ovine T cell subsets section "". Cells were stained for CD4 with mab CC8-PE at 1:20 dilution ( A , D ), for CD8beta with mab CC58-PE at 1:20 dilution ( B , D ) and for WC-1 (gammadelta T cells) with mab CC15-PE at 1:200 ( C , E ). Intracellular cytokine staining for IL-17A was conducted using mab eBioDEC17-APC at a 1:20 dilution ( A - C ) and for IFN-gamma using mab CC302-Alexafluor 647 at a 1:200 dilution ( D - F ). Data are shown for PBMC from one representative animal of four.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
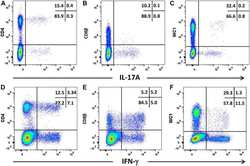
- Figure 6 Intracellular expression of IL-17A and IFN-gamma by activated ovine T cell subsets. PBMC from four sheep were stimulated with phorbol 12-myristate 13 acetate, ionomycin and brefeldin A in RPMI culture medium for 4 h. Cells were harvested and stained for viability and with mabs specific for cell-surface phenotypic markers and intracellular cytokines as described in Table 3 and "" Expression of intracellular IL-17A and IFN-gamma by bovine and ovine T cell subsets section "". Cells were then stained for CD4 with mab 44.38-PE at 1:20 dilution ( A , D ), CD8beta with mab CC58-PE at 1:20 dilution ( B , D ) and WC-1 (gammadelta) with mab CC15-PE at 1:200 ( C , E ). Intracellular cytokine staining for IL-17A was conducted using mab eBio64DEC17-APC a 1:20 dilution ( A - C ) and for IFN-gamma using mab CC302-alexafluor 647 at a 1:200 dilution ( D - F ). Data shown is for one representative animal out of four.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Additional file 2. Gating strategy used for the evaluation of commercial antibodies to bind intracellular recombinant bovine and ovine IL-17A in fixed cells. Cells were acquired for flow cytometric analyses using the MacsQuant flow cytometer and analysed using the MacsQuantify Software. 20 000-50 000 events were collected and the following gating strategy was followed. Cells in the plot of Forward Scatter-Area (FSC-A) against the high dynamic range over time (HDR-T) are gated in P1 to exclude any non-specific artefacts ( A ). The P1/P2 gate represents Side Scatter-Area (SSC-A) plotted against FSC-A set to identify the main cell population and exclude debris ( B ). Single cells were gated (P1/P2/P3) using FSC-Height (H) vs FSC-A for doublet discrimination ( C ). Finally, the cells of interest were identified in the phycoerythrin or alexafluor 488 channel vs SSC-A (P1/P2/P3/P4) where regions were set using the isotype or equivalent control for each CHO cell line to establish threshold gates ( D ). Overlaying histogram plots of phycoerythrin or alexafluor 488 using (P1/P2/P3) gating strategy selecting for all cells in the region (equivalent to cells above and below region boundary in plot D ) ( E ) were used to compare anti-IL-17A antibodies with appropriate isotype or equivalent controls presented in Figure 4 . Gated percentage numbers above the region boundary (P1/P2/P3/P4) and median fluorescence region values (P1/P2/P3) were measured for each antibody in the relevant fluoroc
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
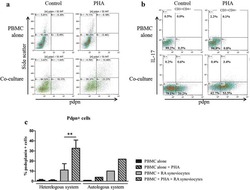
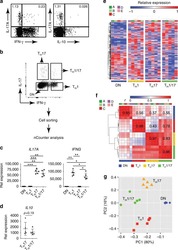
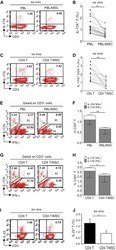
- Figure 1 Multipotent Human Mesenchymal Stromal Cells (hMSCs) Suppress Th17 Responses (A-D) Human peripheral blood CD3 + leukocytes (PBLs) (A, representative data; B, pooled data of 17 PBL donors co-cultured with all three hMSC donors) or CD3 + CD4 T cells (C, representative data; D, pooled data of 11 PBL donors co-cultured with all three hMSC donors) were co-cultured without (left) or with (right) hMSCs ex vivo, followed by PMA/ionomycin stimulation for 6 hr. (E-H) IL-17A production in ex-vivo-cultured CD3 + T cells was assessed by intracellular staining. IL-17A and IFN-gamma production in CD3 + PBLs (E, representative data; F, pooled data) or CD3 + CD4 T cells (G, representative data; H, pooled data) without and with co-culture of hMSCs was analyzed by flow cytometry. Representative intracellular staining is shown for IL-17A + IFN-gamma - - CD3 + T cells (R3 region) and IL-17A + IFN-gamma + (R5 region) CD3 + T cells, and pooled data from PBLs (n = 4) or CD4 T cells (n = 4) co-cultured with two hMSC donors (donors A and B) are provided in (F) and (H), respectively. Gray bars represent the percentages of IL-17A + IFN-gamma - - CD3 + T cells, whereas white bars represent the percentages of IL-17A + IFN-gamma + T cells. (I and J) IL-22 production in four donors of CD3 + CD4 T cells (I, representative data; J, pooled data) without and with co-culture of two donors of hMSCs (donors A and B) was assessed by intracellular staining. Cell percentages are denoted in the dotplot quadran
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
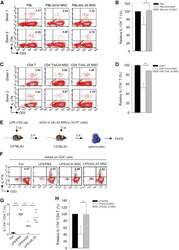
- Figure 3 IL-25 Silencing in hMSCs Reverses Th17 Responses In Vitro and In Vivo (A-D) Freshly isolated human PBLs (A) or CD4 T cells (C) were co-cultured without (left) or with either siCtrl hMSCs (middle) or siIL-25 hMSCs (right) for 3 days, followed by PMA/ionomycin stimulation for 6 hr. IL-17A production in CD3 + T cells was assessed by intracellular staining. Numbers in the top right quadrants represent the percentages of IL-17A-producing CD3 + T cells. Pooled data from PBLs (n = 3) or CD4 T cells (n = 3) and two hMSC donors (donors A and B) are provided in (B) and (D), respectively. Data are shown as mean +- SD. * p < 0.05, ** p < 0.01. (E) Experimental strategy for establishing in vivo inflammatory conditions in wild-type C57BL/6J mice with expansion of Th17 cells and adoptive transfer of hMSCs is shown. (F) On day 3 after LPS (100 mug/mouse) challenge, IL-17A production in activated CD4 T cells in splenocytes from control mice, PBS-treated mice, siCtrl-hMSC-treated mice, or siIL-25-hMSC-treated mice was assessed by intracellular staining. (G and H) Calculated (G) and relative (H) mean percentage of IL-17A-expressing CD4 T cells among control mice, PBS-treated mice, siCtrl-hMSC-treated mice, or siIL-25-hMSC-treated mice (n = 6). Data are shown as mean +- SD. * p < 0.05, ** p < 0.01, *** p < 0.005.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
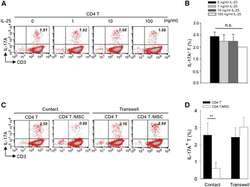
- Experimental details
- Figure 4 Exogenous IL-25 Alone Is Insufficient to Significantly Suppress Th17 Responses, with Cell Contact Required as well for hMSC-Mediated Inhibition of Th17 Responses (A) Human CD4 T cells were treated with indicated doses of rhIL-25 for 18 hr, followed by PMA/ionomycin stimulation for 6 hr. IL-17A production in CD3 + T cells was assessed by intracellular staining. Numbers in the top right quadrants represent the percentages of IL-17A-producing CD3 + T cells. (B) Pooled data of five PBL donors are shown. (C) Human CD4 T cells (n = 4) were co-cultured without or with hMSCs (two donors, B and C) in the absence or presence of transwell barriers. (D) Pooled data from healthy donors are shown. Data are shown as mean +- SD. ** p < 0.01; n.s., not significant.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
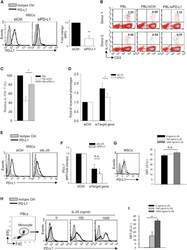
- Experimental details
- Figure 5 IL-25 Induces PD-L1 Surface Expression on hMSCs and Human Monocytes (A) PD-L1 in siCtrl MSCs (left) and siPD-L1 MSCs (right) was analyzed by surface staining. (B) Freshly isolated human PBLs were co-cultured without (left) or with siCtrl MSCs (middle) or siPD-L1 MSCs (right) for 3 days, followed by PMA/ionomycin stimulation for 6 hr. IL-17A production in CD3 + T cells was assessed by intracellular staining. Representative data are shown with numbers in the top right quadrants representing the percentages of IL-17A-producing CD3 + T cells. (C) Pooled data from PBLs (n = 4) and two hMSC donors (donors A and B) are shown. (D) Folds of reversed phenotypes of siIL-25 and siPD-L1 are shown. (E) PD-L1 expression on siCtrl hMSCs (left) and siIL-25 hMSCs (right) was assessed by cell surface staining. Filled histograms represent isotype control; unfilled histograms represent PD-L1 antibody staining. (F) Pooled data of PD-L1 expression (indicated by fold change in MFI) on siIL-25 hMSCs and siPD-L1 hMSCs (all three donors) are shown. PD-L1 expression levels were compared between hMSCs silenced for the target gene (IL-25 or PD-L1) and the respective siCtrl. (G) hMSCs were treated with the indicated doses of rhIL-25 for 18 hr and assessed for cell surface PD-L1 expression by cell surface staining. Pooled data (all three donors) are shown in chart to the right with bars representing MFI. (H) Human PBLs were treated with the indicated doses of rhIL-25 for 18 hr and assessed for cell
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
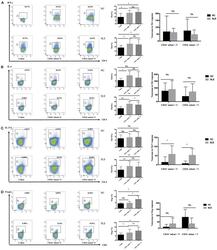
- Experimental details
- Figure 4 CD16 + monocytes promoted T-cell-mediated inflammation in SLE . CD16 + or CD16 - monocytes were cocultured with CD4 + T cells isolated from freshly collected SLE blood or blood bank collected HC blood buffy coat for 5 days in the presence of anti-CD3 (1 mug/mL) and anti-CD28 (1 mug/mL) antibodies and M-CSF (50 ng/mL). Intracellular IFN-gamma, IL-4, and IL-17A expression was detected by flow cytometry after PMA (50 ng/mL), ionomycin (1 mug/mL), and BFA (10 mug/mL) stimulation for 5 h on day 5. The percentage of Treg (CD4 + Foxp3 + ) was also analyzed. Representative pseudocolor dots depicted Th1 (A) , Th2 (B) , Th17 (C) , and Treg (D) frequencies in CD4 + T cells after coculture with each monocyte subset from one patient and one control donor. The proportion of Th1, Th2, Th17, and Treg cells was calculated after coculture of CD4 + T cells with each monocyte. The percentage increases in T-cell subsets in cocultures of monocytes and T cells compared with CD4 + T cells cultured alone were compared between 7 healthy individuals and 10 patients with SLE. Data were expressed as mean +- SD and analyzed by non-parametric paired t test and Mann-Whitney U test. * P < 0.05, ** P < 0.01; NS, no significance.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
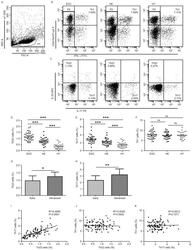
- Experimental details
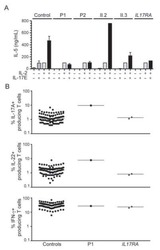
- Figure 1. Flow cytometric analysis was used to determine the distribution of Th22, Th17 and Th1 cells in EGC, HE and HY ( Fig. 1 ). Flow cytometric analysis of Th22, Th17 and Th1 cells in peripheral whole blood from EGC (n=39), HE (n=32) and HY (n=31). (A) Lymphocytes were gated in P1 using flow cytometry. CD4 + IFN-gamma - lymphocytes were gated in P2 using flow cytometry, and representative results of flow cytometric analyses for (B) Th1 (CD4 + IFN-gamma + ), (C) Th22 (CD4 + IFN-gamma - IL-17 - IL-22 + ) and Th17 (CD4 + IFN-gamma - IL-17 + IL-22 - ) cells in the three groups of subjects are presented. The number of cells stained in EGC, HE and HY in P2 were 2,654, 4,696 and 5,185, respectively. The proportion of (D) Th22, (E) Th17 and (F) Th1 cells in the three groups of subjects. The proportion of (G) Th22 and (H) Th17 cells in peripheral whole blood derived from patients with early (n=13) or advanced (n=26) gastric cancer. The association between the proportion of (I) Th22 and Th17 cells, (J) Th22 and Th1 cells, and (K) Th17 and Th1 cells, in peripheral whole blood of all subjects. *P
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry