Antibody data
- Antibody Data
- Antigen structure
- References [17]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [21]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 50-7178-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IL-17A Monoclonal Antibody (eBio64CAP17), eFluor™ 660, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The eBio64CAP17 antibody reacts with human IL-17A; the antibody has been reported to cross react with Rhesus monkey IL-17A, as verified by intracellular staining experiments. The eBio64CAP17 antibody is a neutralizing antibody. This antibody has been shown to have no reactivity to human IL-17F. Reactivity to other members of the IL-17 family has not been evaluated. Interleukin-17A (IL-17A) is a CD4+ T cell-derived cytokine that promotes inflammatory responses in cell lines and is elevated in rheumatoid arthritis, asthma, multiple sclerosis, psoriasis, and transplant rejection. The cDNA encoding human IL-17A was isolated from a library of CD4+ T cells; the encoded protein exhibits 72 percent amino acid identity with HVS13 , an open reading frame from a T lymphotropic Herpesvirus saimiri, and 63 percent with mouse CTLA-8 (cytotoxic T-lymphocyte associated antigen-8). Human IL-17A exists as glycosylated 20-30 kD homodimers. High levels of IL-17A homodimer are produced by activated peripheral blood CD4+ T-cells. IL-17A enhances expression of the intracellular adhesion molecule-1 (ICAM-1) in human fibroblasts. Human IL-17A also stimulates epithelial, endothelial, or fibroblastic cells to secrete IL-6, IL-8, G-CSF, and PGE2. In the presence of human IL-17A, fibroblasts can sustain the proliferation of CD34+ hematopoietic progenitors and induce maturation into neutrophils. Mouse, rat, and human IL-17A can induce IL-6 secretion in mouse stromal cells, indicating that all homologs can recognize the mouse IL-17A receptor. IL-23-dependent, IL-17A-producing CD4+ T cells (Th-17 cells) have been identified as a unique subset of Th cells that develops along a pathway that is distinct from the Th1- and Th2- cell differentiation pathways. The hallmark effector molecules of Th1 and Th2 cells, e.g., IFN-g and IL-4, have each been found to negatively regulate the generation of these Th-17 cells. Additionally, activated human CD4+ T cells have been found to produce the IL-17A/F heterodimer, as well as the corresponding homodimers. In comparing the relative potency of IL-17A, IL-17F, and IL-17A/F, all three were found to induce GRO-a secretion; IL-17A was most potent, followed by IL-17A/F heterodimer, then IL-17F (100fold lower than IL-17A). eBio64CAP17 can be used to detect IL-17 heterodimers by immunoprecipitation followed by immunoblot withH17F10A7 anti-IL17F monoclonal antibody. The eBio64CAP17 has been shown to react to rhesus and marmoset primates. Applications Reported: This eBio64CAP17 antibody has been reported for use in intracellular staining followed by flow cytometric analysis. Applications Tested: This eBio64CAP17 antibody has been pre-titrated and tested by intracellular staining and flow cytometric analysis of stimulated human peripheral blood cells. This can be used at 5 µL (0.125 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. eFluor® 660 is a replacement for Alexa Fluor® 647. eFluor® 660 emits at 659 nm and is excited with the red laser (633 nm). Please make sure that your instrument is capable of detecting this fluorochome. Excitation: 633-647 nm; Emission: 668 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- eBio64CAP17
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references FABP4 facilitates inflammasome activation to induce the Treg/Th17 imbalance in preeclampsia via forming a positive feedback with IL-17A.
Immunological evaluation of an inactivated SARS-CoV-2 vaccine in rhesus macaques.
Th2 Biased Immunity With Altered B Cell Profiles in Circulation of Patients With Sporotrichosis Caused by Sporothrix globosa.
Allogeneic ADSCs Induce the Production of Alloreactive Memory-CD8 T Cells through HLA-ABC Antigens.
Allogeneic ADSCs induce CD8 T cell-mediated cytotoxicity and faster cell death after exposure to xenogeneic serum or proinflammatory cytokines.
The Effects of High Mobility Group Box-1 Protein on Peripheral Treg/Th17 Balance in Patients with Atherosclerosis.
RNA-seq and flow-cytometry of conventional, scalp, and palmoplantar psoriasis reveal shared and distinct molecular pathways.
P2X7R mutation disrupts the NLRP3-mediated Th program and predicts poor cardiac allograft outcomes.
Application of a whole blood mycobacterial growth inhibition assay to study immunity against Mycobacterium tuberculosis in a high tuberculosis burden population.
Downregulation of RUNX3 moderates the frequency of Th17 and Th22 cells in patients with psoriasis.
Altered expression of miR-92a correlates with Th17 cell frequency in patients with primary biliary cirrhosis.
Fiber composite slices for multiplexed immunoassays.
Impaired Th17 polarization of phenotypically naive CD4(+) T-cells during chronic HIV-1 infection and potential restoration with early ART.
Differential requirement for P2X7R function in IL-17 dependent vs. IL-17 independent cellular immune responses.
CCL20 Secretion from the Nucleus Pulposus Improves the Recruitment of CCR6-Expressing Th17 Cells to Degenerated IVD Tissues.
A monoclonal antibody selection for immunohistochemical examination of lymphoid tissues from non-human primates.
Interleukin-17 stimulates C-reactive protein expression in hepatocytes and smooth muscle cells via p38 MAPK and ERK1/2-dependent NF-kappaB and C/EBPbeta activation.
Chang GP, Yang XL, Liu W, Lin S, Yang SL, Zhao MY
Molecular therapy. Nucleic acids 2021 Jun 4;24:743-754
Molecular therapy. Nucleic acids 2021 Jun 4;24:743-754
Immunological evaluation of an inactivated SARS-CoV-2 vaccine in rhesus macaques.
Chen H, Xie Z, Long R, Fan S, Li H, He Z, Xu K, Liao Y, Wang L, Zhang Y, Li X, Dong X, Mou T, Zhou X, Yang Y, Guo L, Yang J, Zheng H, Xu X, Li J, Liang Y, Li D, Zhao Z, Hong C, Zhao H, Jiang G, Che Y, Yang F, Hu Y, Wang X, Pu J, Ma K, Wang L, Cheng C, Duan W, Shen D, Zhao H, Jiang R, Deng X, Li Y, Zhu H, Zhou J, Yu L, Xu M, Yang H, Yi L, Zhou Z, Yang J, Duan N, Yang H, Zhao W, Yang W, Li C, Liu L, Li Q
Molecular therapy. Methods & clinical development 2021 Dec 10;23:108-118
Molecular therapy. Methods & clinical development 2021 Dec 10;23:108-118
Th2 Biased Immunity With Altered B Cell Profiles in Circulation of Patients With Sporotrichosis Caused by Sporothrix globosa.
Zu J, Yao L, Song Y, Cui Y, Guan M, Chen R, Zhen Y, Li S
Frontiers in immunology 2020;11:570888
Frontiers in immunology 2020;11:570888
Allogeneic ADSCs Induce the Production of Alloreactive Memory-CD8 T Cells through HLA-ABC Antigens.
Chang SH, Kim HJ, Park CG
Cells 2020 May 18;9(5)
Cells 2020 May 18;9(5)
Allogeneic ADSCs induce CD8 T cell-mediated cytotoxicity and faster cell death after exposure to xenogeneic serum or proinflammatory cytokines.
Chang SH, Park CG
Experimental & molecular medicine 2019 Mar 11;51(3):1-10
Experimental & molecular medicine 2019 Mar 11;51(3):1-10
The Effects of High Mobility Group Box-1 Protein on Peripheral Treg/Th17 Balance in Patients with Atherosclerosis.
Ding JW, Zhou T, Zheng XX, Wang XA, Tong XH, Luo CY, Zhang ZQ, Yu B
Acta Cardiologica Sinica 2018 Sep;34(5):399-408
Acta Cardiologica Sinica 2018 Sep;34(5):399-408
RNA-seq and flow-cytometry of conventional, scalp, and palmoplantar psoriasis reveal shared and distinct molecular pathways.
Ahn R, Yan D, Chang HW, Lee K, Bhattarai S, Huang ZM, Nakamura M, Singh R, Afifi L, Taravati K, Munoz-Sandoval P, Pauli M, Rosenblum MD, Liao W
Scientific reports 2018 Jul 27;8(1):11368
Scientific reports 2018 Jul 27;8(1):11368
P2X7R mutation disrupts the NLRP3-mediated Th program and predicts poor cardiac allograft outcomes.
D'Addio F, Vergani A, Potena L, Maestroni A, Usuelli V, Ben Nasr M, Bassi R, Tezza S, Dellepiane S, El Essawy B, Iascone M, Iacovoni A, Borgese L, Liu K, Visner G, Dhe-Paganon S, Corradi D, Abdi R, Starling RC, Folli F, Zuccotti GV, Sayegh MH, Heeger PS, Chandraker A, Grigioni F, Fiorina P
The Journal of clinical investigation 2018 Aug 1;128(8):3490-3503
The Journal of clinical investigation 2018 Aug 1;128(8):3490-3503
Application of a whole blood mycobacterial growth inhibition assay to study immunity against Mycobacterium tuberculosis in a high tuberculosis burden population.
Baguma R, Penn-Nicholson A, Smit E, Erasmus M, Day J, Makhethe L, de Kock M, Hughes EJ, van Rooyen M, Pienaar B, Stone L, Hanekom W, Brennan MJ, Wallis RS, Hatherill M, Scriba TJ
PloS one 2017;12(9):e0184563
PloS one 2017;12(9):e0184563
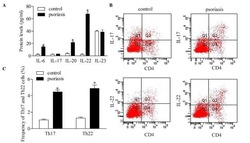
Downregulation of RUNX3 moderates the frequency of Th17 and Th22 cells in patients with psoriasis.
Fu D, Song X, Hu H, Sun M, Li Z, Tian Z
Molecular medicine reports 2016 Jun;13(6):4606-12
Molecular medicine reports 2016 Jun;13(6):4606-12
Altered expression of miR-92a correlates with Th17 cell frequency in patients with primary biliary cirrhosis.
Liang DY, Hou YQ, Luo LJ, Ao L
International journal of molecular medicine 2016 Jul;38(1):131-8
International journal of molecular medicine 2016 Jul;38(1):131-8
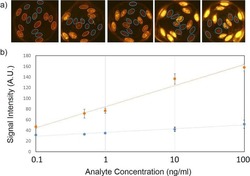
Fiber composite slices for multiplexed immunoassays.
Kim J, Bae S, Song S, Chung K, Kwon S
Biomicrofluidics 2015 Jul;9(4):044109
Biomicrofluidics 2015 Jul;9(4):044109
Impaired Th17 polarization of phenotypically naive CD4(+) T-cells during chronic HIV-1 infection and potential restoration with early ART.
DaFonseca S, Niessl J, Pouvreau S, Wacleche VS, Gosselin A, Cleret-Buhot A, Bernard N, Tremblay C, Jenabian MA, Routy JP, Ancuta P
Retrovirology 2015 Apr 30;12:38
Retrovirology 2015 Apr 30;12:38
Differential requirement for P2X7R function in IL-17 dependent vs. IL-17 independent cellular immune responses.
Sullivan JA, Jankowska-Gan E, Shi L, Roenneburg D, Hegde S, Greenspan DS, Wilkes DS, Denlinger LC, Burlingham WJ
American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 2014 Jul;14(7):1512-22
American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 2014 Jul;14(7):1512-22
CCL20 Secretion from the Nucleus Pulposus Improves the Recruitment of CCR6-Expressing Th17 Cells to Degenerated IVD Tissues.
Zhang W, Nie L, Wang Y, Wang XP, Zhao H, Dongol S, Maharjan S, Cheng L
PloS one 2013;8(6):e66286
PloS one 2013;8(6):e66286
A monoclonal antibody selection for immunohistochemical examination of lymphoid tissues from non-human primates.
Kap YS, van Meurs M, van Driel N, Koopman G, Melief MJ, Brok HP, Laman JD, 't Hart BA
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2009 Dec;57(12):1159-67
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2009 Dec;57(12):1159-67
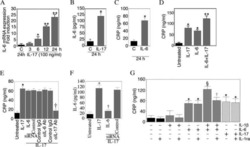
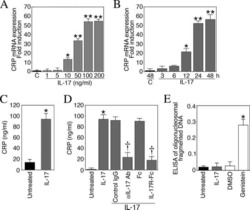
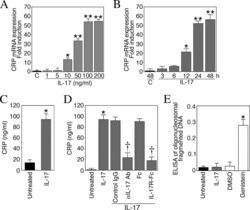
Interleukin-17 stimulates C-reactive protein expression in hepatocytes and smooth muscle cells via p38 MAPK and ERK1/2-dependent NF-kappaB and C/EBPbeta activation.
Patel DN, King CA, Bailey SR, Holt JW, Venkatachalam K, Agrawal A, Valente AJ, Chandrasekar B
The Journal of biological chemistry 2007 Sep 14;282(37):27229-27238
The Journal of biological chemistry 2007 Sep 14;282(37):27229-27238
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- CD4-enriched human peripheral blood cells were polarized under Th17 conditions (with Human IL-23 Recombinant Protein (Product # 14-8239-63) for 8 days. Cells were restimulated with Protein Transport Inhibitor Cocktail (Product # 00-4980-03) (left) or Cell Stimulation Cocktail (plus protein transport inhibitors) (Product # 00-4975-03) (right) for 6 hours. Cells were stained with Fixable Viability Dye eFluor® 780 (Product # 65-0865-14) followed by intracellular staining with Anti-Human CD4 PE (Product # 12-0049-42) and Anti-Human IL-17A eFluor 660 using the Fixation & Permeabilization Buffers (Product # 88-8823-88). Viable cells were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
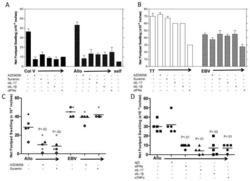
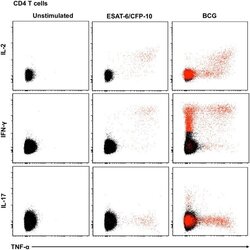
- Fig 5 Representative flow cytometry plots of cytokine expression by CD4 T cells. CD4 T cells expressing IFN-gamma, IL-2, TNF-alpha and/or IL-17 upon stimulation with BCG or ESAT-6/CFP-10 peptide pools for 12 hours, compared to an unstimulated control sample. The plots represent cytokine-positive cells (red) overlayed onto the background of the entire CD4 T cell parent population (black). Similar assessment was also performed for CD8 and gammadelta T cells (data not shown).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
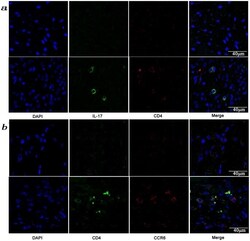
- Figure 3 Presence of IL-17-producing cells in the degenerated IVD tissues. Fifty IVD tissue samples (Group P, n = 20 and Group E, n = 30) from disc degeneration patients and 3 from scoliosis patients were analyzed. In Group P or the control group, there were few or no positive cells (data not shown). The representative results of group E are shown. (a), IL-17-producing cells were detected by a rabbit anti-IL-17 polyclonal antibody and a mouse anti-CD4 monoclonal Ab, followed by secondary staining with an Alexa 488-conjugated donkey anti-rabbit and an Alexa 568-conjugated donkey anti-mouse IgG. DAPI mounting medium was used for nuclear staining. (b), surface CCR6 expression on the cells was detected with a rabbit anti CD4 monoclonal Ab and a mouse anti-CCR6 monoclonal Ab, followed by secondary staining with an Alexa 488-conjugated donkey anti-rabbit and an Alexa 568-conjugated donkey anti-mouse IgG. DAPI mounting medium was used for nuclear staining. In the top panel, green and red represents the expression of IL-17 and CD4, respectively, and the double-stained cells represent the IL-17-producing cells. In the bottom panel, green and red represents the expression of CD4 and CCR6, respectively, and the double-stained cells demonstrate the surface expression of CCR6 on T lymphocytes.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
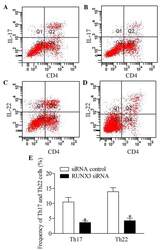
- Figure 5 Inhibition of RUNX3 regulates the frequencies of Th17 and Th22 in CD4 + T cells from patients with psoriasis. The frequencies of Th17 and Th22 cells in CD4 + T cells from patients with psoriasis transfected with RUNX3 siRNA or an siRNA control was detected using flow cytometry. (A and B) Flow cytometric analysis of Th17 in CD4 + T cells from patients with psoriasis. The percentages of cells in the Q2 region represent the percentage of Th17 cells. (C and D) Flow cytometric analysis of Th22 in CD4 + T cells from patients with psoriasis. The percentages of cells in the Q2 region represent the percentage of Th22 cells. (E) Percentages of Th17 and Th22 cells in CD4 + T cells from patients with psoriasis transfected with RUNX3 siRNA or an siRNA control. Data are presented as the mean +- standard deviation of three experiments. * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
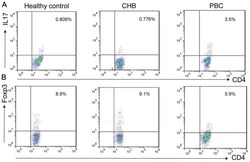
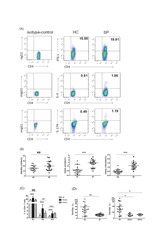
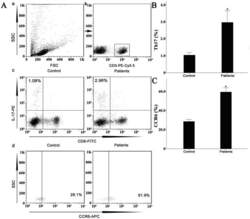
- Figure 7 Circulating percentages of Th17 cells and CCR6-positive cells in peripheral blood are increased in IVD degenerated patients when compared with controls. Heparinized peripheral whole blood cells from 20 patients and 15 healthy controls were stimulated with phorbol myristate acetate (PMA), ionomycin, and monensin for 4 h and subsequently stained with fluorochrome-labeled antibodies as described in Materials and Methods. A(a) Lymphocytes were gated by flow cytometry. A(b) CD3 + T subsets were gated by flow cytometry; the plots in the inset box represent the CD3 + T cells. A(c) Representative IL-17 expression levels in the CD3 + CD8 - T subsets (CD4 + T subsets) from each group are shown. The percentages of positive cells are shown in the upper left panels. A(d) Representative surface CCR6 expression levels on the CD3 + CD8 - IL-17 + subsets from each group are shown. The percentages of positive cells are shown in the right panel. (B) The percentage of circulating Th17 cells was significantly higher in IVD degenerated patients (2.973+-0.689%) than in the control group (1.039+-0.156%; *, p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
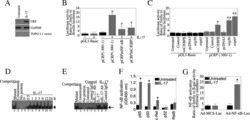
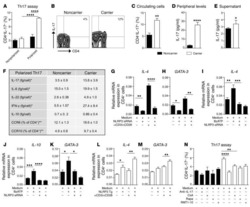
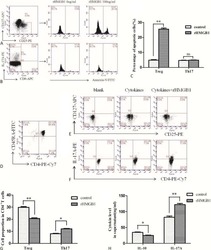
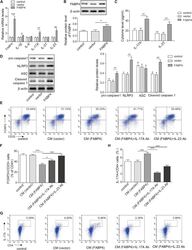
- Figure 4 FABP4 promotes Th17 differentiation of naive T cells via increasing IL-17A/IL-23 release (A) The mRNA levels of cytokines were determined by qRT-PCR. (B) The protein level of FABP4 was detected by western blot. (C) The IL-17A and IL-23 levels in cell culture supernatant were assessed by ELISA. (D) The protein levels of NLRP3 inflammasome components were determined by western blot. Naive T cells were cultured under Treg or Th17 cell-polarizing conditions with or without primary macrophages-CM and neutralizing antibody. (E-H) The percentage of Tregs (E and F) or Th17 cells (G and H) was assessed by FACS. GAPDH and beta-actin were used for normalization in qRT-PCR and western blot, respectively. Data are expressed as representative images or the mean +- SD of n = 3 experiments. *p < 0.05, **p < 0.01, ***p < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
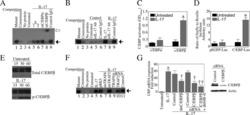
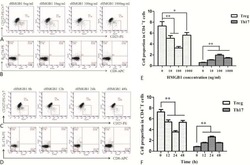
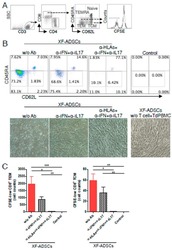
- Figure 6 HLA-blocking antibodies inhibit the production of alloreactive memory-CD8 T cells during allogeneic-antigen stimulation. XF-ADSCs were cultured with CD3 T cells and Td-PBMCs via the direct pathway, after 3 weeks whole cells were harvested and memory-CD8 T cells were analyzed by flow cytometry. ( A ) Scheme for the flow cytometry analysis. ( B ) CD3 T cells were cultured for 3 weeks with or without neutralizing or blocking antibodies during allogeneic-antigen stimulation. A combination of anti-human IL-17A and anti-human IFN-gamma antibodies was used to neutralize pro-inflammatory cytokines. A combination of anti-HLA-ABC, anti-HLA-DR and anti-HLA-DQ antibodies was used to block HLAs. The dot plots showed the population of memory-CD8 T cells on the 21st day after allogeneic-antigen stimulation, compared with the control. ( C ) Graphical representation of the number of CFSE-low memory-CD8 T cells corresponding to (B) ( n = 7 (each CD8 TEM), n = 6 (each CD8 TCM)). The control does not contain XF-ADSCs and consist only of CD3 T cells and Td-PBMCs from healthy donor. The data were repeated at least three times; *, p < 0.05; **, p < 0.01; ***, p < 0.001. alpha-IFN+alpha-IL17: combination of anti-human IFN-gamma and anti-human IL-17A antibodies; alpha-HLAs: combination of anti-HLA-ABC, HLA-DR and anti-HLA-DQ antibodies.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
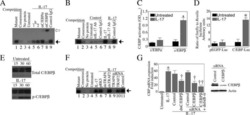
- Fig. 5 Human leukocyte antigens (HLAs) on allogeneic adipose-derived mesenchymal stem cells grown in xeno-free medium (XF-ADSCs) are the major cause of T cell-mediated cytotoxicity. a XF-ADSCs were subjected to allogeneic antigen stimulation for 21-24 days with antibodies against interferon-gamma (IFN-gamma), interleukin (IL)-17A, HLA-ABC, HLA-DR, and HLA-DQ (all at 1 mug/ml) in xeno-free medium containing 5% autologous serum. The effects of the XF-ADSCs with antibodies on T cell-mediated cytotoxicity were evaluated in comparison with those of the XF-ADSCs not containing antibodies. For cell death analysis, the collected XF-ADSCs were stained with anti-CD73 and anti-CD90 antibodies, annexin V, and propidium iodide (PI), and the cell death rate was analyzed by flow cytometry. b Graphical representation of XF-ADSC death corresponding to ( a ) ( n = 21 (untreated), n = 7 (for each sample)); *** p < 0.001. Td-PBMCs T cell-depleted T cell-depleted peripheral blood mononuclear cells
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry