12-8899-41
antibody from Invitrogen Antibodies
Targeting: GZMB
CCPI, CGL-1, CGL1, CSP-B, CSPB, CTLA1, CTSGL1, HLP, SECT
Antibody data
- Antibody Data
- Antigen structure
- References [21]
- Comments [0]
- Validations
- Flow cytometry [2]
- Other assay [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 12-8899-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Granzyme B Monoclonal Antibody (GB11), PE, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The GB11 antibody reacts with human Granzyme B (GrB). GrB is one of the family of serine proteases, known as granzymes, that is located in the granules of cytotoxic T cells and NK cells. Granzyme B has been described also as CGL1 (cathepsin G-like-1), a serine protease expressed only in cytotoxic T-lymphocytes after cell activation. GrB has been called CTLA-1 (cytotoxic T lymphocyte-associated serine esterase 1) based on identification of mRNA in various cytotoxic T cells, but not observed in non-cytotoxic lymphoid cells. GrB is crucial for the rapid induction of target cell death by apoptosis, induced by interaction with cytotoxic T cells. The receptor involved has been identified as mannose 6-phosphate receptor. This receptor functions as a death receptor for granzyme B during cytotoxic T cell-induced apoptosis. Applications Reported: The GB11 antibody has been reported useful for intracellular staining followed by flow cytometric analysis. Applications Tested: This GB11 antibody has been pre-titrated and tested on by intracellular staining and flow cytometric analysis of both cultured normal human peripheral blood cells and mouse splenocytes. This can be used at test size: 5 µL (0.06 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488-561 nm; Emission: 578 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Yellow dye
- Isotype
- IgG
- Antibody clone number
- GB11
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references GPR116 receptor regulates the antitumor function of NK cells via Gαq/HIF1α/NF-κB signaling pathway as a potential immune checkpoint.
Suppression of 4.1R enhances the potency of NKG2D-CAR T cells against pancreatic carcinoma via activating ERK signaling pathway.
The killing effect of Tanshinol on breast cancer cells: insight into the reversion of TGF-β1-mediated suppression of NK cell functions.
Human Thymic CD10(+) PD-1(+) Intraepithelial Lymphocyte Precursors Acquire Interleukin-15 Responsiveness at the CD1a(-) CD95(+) CD28(-) CCR7(-) Developmental Stage.
Targets of T Cell Responses to SARS-CoV-2 Coronavirus in Humans with COVID-19 Disease and Unexposed Individuals.
Autophagy in T cells from aged donors is maintained by spermidine and correlates with function and vaccine responses.
CXCR3 chemokine receptor guides Trypanosoma cruzi-specific T-cells triggered by DNA/adenovirus ASP2 vaccine to heart tissue after challenge.
Enhanced Anti-lymphoma Activity of CAR19-iNKT Cells Underpinned by Dual CD19 and CD1d Targeting.
Effect of IL-7 Therapy on Phospho-Ribosomal Protein S6 and TRAF1 Expression in HIV-Specific CD8 T Cells in Patients Receiving Antiretroviral Therapy.
Enhancing adoptive cancer immunotherapy with Vγ2Vδ2 T cells through pulse zoledronate stimulation.
Low-dose interleukin-2 promotes STAT-5 phosphorylation, T(reg) survival and CTLA-4-dependent function in autoimmune liver diseases.
Blocking the recruitment of naive CD4(+) T cells reverses immunosuppression in breast cancer.
Skewed distribution of IL-7 receptor-α-expressing effector memory CD8+ T cells with distinct functional characteristics in oral squamous cell carcinoma.
PD-1(+) CD8(+) T cells are exhausted in tumours and functional in draining lymph nodes of colorectal cancer patients.
Differentiation and functional regulation of human fetal NK cells.
A polymorphism in TIM1 is associated with susceptibility to severe hepatitis A virus infection in humans.
Impaired NK cytolytic activity and enhanced tumor growth in NK lytic-associated molecule-deficient mice.
The Granzyme B ELISPOT assay: an alternative to the 51Cr-release assay for monitoring cell-mediated cytotoxicity.
Granzyme B ELISPOT assay for ex vivo measurements of T cell immunity.
Granzymes: exogenous proteinases that induce target cell apoptosis.
Human natural killer cells: biologic and pathologic aspects.
Guo D, Jin C, Gao Y, Lin H, Zhang L, Zhou Y, Yao J, Duan Y, Ren Y, Hui X, Ge Y, Yang R, Jiang W
Cell & bioscience 2023 Mar 9;13(1):51
Cell & bioscience 2023 Mar 9;13(1):51
Suppression of 4.1R enhances the potency of NKG2D-CAR T cells against pancreatic carcinoma via activating ERK signaling pathway.
Gao Y, Lin H, Guo D, Cheng S, Zhou Y, Zhang L, Yao J, Farooq MA, Ajmal I, Duan Y, He C, Tao L, Wu S, Liu M, Jiang W
Oncogenesis 2021 Sep 21;10(9):62
Oncogenesis 2021 Sep 21;10(9):62
The killing effect of Tanshinol on breast cancer cells: insight into the reversion of TGF-β1-mediated suppression of NK cell functions.
Yang C, Qian C, Zhang T, Zheng W, Zhang S, Wang S, Wang A, Zhao Y, Lu Y
Frontiers in bioscience (Landmark edition) 2021 Nov 30;26(11):1106-1118
Frontiers in bioscience (Landmark edition) 2021 Nov 30;26(11):1106-1118
Human Thymic CD10(+) PD-1(+) Intraepithelial Lymphocyte Precursors Acquire Interleukin-15 Responsiveness at the CD1a(-) CD95(+) CD28(-) CCR7(-) Developmental Stage.
Billiet L, Goetgeluk G, Bonte S, De Munter S, De Cock L, Pille M, Ingels J, Jansen H, Weening K, Van Nieuwerburgh F, Kerre T, Taghon T, Leclercq G, Vandekerckhove B
International journal of molecular sciences 2020 Nov 20;21(22)
International journal of molecular sciences 2020 Nov 20;21(22)
Targets of T Cell Responses to SARS-CoV-2 Coronavirus in Humans with COVID-19 Disease and Unexposed Individuals.
Grifoni A, Weiskopf D, Ramirez SI, Mateus J, Dan JM, Moderbacher CR, Rawlings SA, Sutherland A, Premkumar L, Jadi RS, Marrama D, de Silva AM, Frazier A, Carlin AF, Greenbaum JA, Peters B, Krammer F, Smith DM, Crotty S, Sette A
Cell 2020 Jun 25;181(7):1489-1501.e15
Cell 2020 Jun 25;181(7):1489-1501.e15
Autophagy in T cells from aged donors is maintained by spermidine and correlates with function and vaccine responses.
Alsaleh G, Panse I, Swadling L, Zhang H, Richter FC, Meyer A, Lord J, Barnes E, Klenerman P, Green C, Simon AK
eLife 2020 Dec 15;9
eLife 2020 Dec 15;9
CXCR3 chemokine receptor guides Trypanosoma cruzi-specific T-cells triggered by DNA/adenovirus ASP2 vaccine to heart tissue after challenge.
Pontes Ferreira C, Cariste LM, Ferri Moraschi B, Ferrarini Zanetti B, Won Han S, Araki Ribeiro D, Vieira Machado A, Lannes-Vieira J, Gazzinelli RT, Vasconcelos JRC
PLoS neglected tropical diseases 2019 Jul;13(7):e0007597
PLoS neglected tropical diseases 2019 Jul;13(7):e0007597
Enhanced Anti-lymphoma Activity of CAR19-iNKT Cells Underpinned by Dual CD19 and CD1d Targeting.
Rotolo A, Caputo VS, Holubova M, Baxan N, Dubois O, Chaudhry MS, Xiao X, Goudevenou K, Pitcher DS, Petevi K, Kachramanoglou C, Iles S, Naresh K, Maher J, Karadimitris A
Cancer cell 2018 Oct 8;34(4):596-610.e11
Cancer cell 2018 Oct 8;34(4):596-610.e11
Effect of IL-7 Therapy on Phospho-Ribosomal Protein S6 and TRAF1 Expression in HIV-Specific CD8 T Cells in Patients Receiving Antiretroviral Therapy.
Wang C, Edilova MI, Wagar LE, Mujib S, Singer M, Bernard NF, Croughs T, Lederman MM, Sereti I, Fischl MA, Kremmer E, Ostrowski M, Routy JP, Watts TH
Journal of immunology (Baltimore, Md. : 1950) 2018 Jan 15;200(2):558-564
Journal of immunology (Baltimore, Md. : 1950) 2018 Jan 15;200(2):558-564
Enhancing adoptive cancer immunotherapy with Vγ2Vδ2 T cells through pulse zoledronate stimulation.
Nada MH, Wang H, Workalemahu G, Tanaka Y, Morita CT
Journal for immunotherapy of cancer 2017;5:9
Journal for immunotherapy of cancer 2017;5:9
Low-dose interleukin-2 promotes STAT-5 phosphorylation, T(reg) survival and CTLA-4-dependent function in autoimmune liver diseases.
Jeffery HC, Jeffery LE, Lutz P, Corrigan M, Webb GJ, Hirschfield GM, Adams DH, Oo YH
Clinical and experimental immunology 2017 Jun;188(3):394-411
Clinical and experimental immunology 2017 Jun;188(3):394-411
Blocking the recruitment of naive CD4(+) T cells reverses immunosuppression in breast cancer.
Su S, Liao J, Liu J, Huang D, He C, Chen F, Yang L, Wu W, Chen J, Lin L, Zeng Y, Ouyang N, Cui X, Yao H, Su F, Huang JD, Lieberman J, Liu Q, Song E
Cell research 2017 Apr;27(4):461-482
Cell research 2017 Apr;27(4):461-482
Skewed distribution of IL-7 receptor-α-expressing effector memory CD8+ T cells with distinct functional characteristics in oral squamous cell carcinoma.
Lee JJ, Yeh CY, Jung CJ, Chen CW, Du MK, Yu HM, Yang CJ, Lin HY, Sun A, Ko JY, Cheng SJ, Chang YL, Chia JS
PloS one 2014;9(1):e85521
PloS one 2014;9(1):e85521
PD-1(+) CD8(+) T cells are exhausted in tumours and functional in draining lymph nodes of colorectal cancer patients.
Wu X, Zhang H, Xing Q, Cui J, Li J, Li Y, Tan Y, Wang S
British journal of cancer 2014 Sep 23;111(7):1391-9
British journal of cancer 2014 Sep 23;111(7):1391-9
Differentiation and functional regulation of human fetal NK cells.
Ivarsson MA, Loh L, Marquardt N, Kekäläinen E, Berglin L, Björkström NK, Westgren M, Nixon DF, Michaëlsson J
The Journal of clinical investigation 2013 Sep;123(9):3889-901
The Journal of clinical investigation 2013 Sep;123(9):3889-901
A polymorphism in TIM1 is associated with susceptibility to severe hepatitis A virus infection in humans.
Kim HY, Eyheramonho MB, Pichavant M, Gonzalez Cambaceres C, Matangkasombut P, Cervio G, Kuperman S, Moreiro R, Konduru K, Manangeeswaran M, Freeman GJ, Kaplan GG, DeKruyff RH, Umetsu DT, Rosenzweig SD
The Journal of clinical investigation 2011 Mar;121(3):1111-8
The Journal of clinical investigation 2011 Mar;121(3):1111-8
Impaired NK cytolytic activity and enhanced tumor growth in NK lytic-associated molecule-deficient mice.
Hoover RG, Gullickson G, Kornbluth J
Journal of immunology (Baltimore, Md. : 1950) 2009 Dec 1;183(11):6913-21
Journal of immunology (Baltimore, Md. : 1950) 2009 Dec 1;183(11):6913-21
The Granzyme B ELISPOT assay: an alternative to the 51Cr-release assay for monitoring cell-mediated cytotoxicity.
Shafer-Weaver K, Sayers T, Strobl S, Derby E, Ulderich T, Baseler M, Malyguine A
Journal of translational medicine 2003 Dec 29;1(1):14
Journal of translational medicine 2003 Dec 29;1(1):14
Granzyme B ELISPOT assay for ex vivo measurements of T cell immunity.
Rininsland FH, Helms T, Asaad RJ, Boehm BO, Tary-Lehmann M
Journal of immunological methods 2000 Jun 23;240(1-2):143-55
Journal of immunological methods 2000 Jun 23;240(1-2):143-55
Granzymes: exogenous proteinases that induce target cell apoptosis.
Smyth MJ, Trapani JA
Immunology today 1995 Apr;16(4):202-6
Immunology today 1995 Apr;16(4):202-6
Human natural killer cells: biologic and pathologic aspects.
Trinchieri G, Perussia B
Laboratory investigation; a journal of technical methods and pathology 1984 May;50(5):489-513
Laboratory investigation; a journal of technical methods and pathology 1984 May;50(5):489-513
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Left: Normal human peripheral blood cells were stimulated for 2 days with Human IL-2 Recombinant Protein (Product # 14-8029-81). The cells were surface stained with Anti-Human CD56 (NCAM) FITC and stained intracellularly with Anti-Human Granzyme B PE. Quadrants demarcate boundary for isotype controls. Right: Mouse splenocytes were stimulated for 3 days with Mouse IL-2 Recombinant Protein (Product # 14-8021-64). Cells were surface stained with Anti-Mouse CD49b (Integrin alpha 2) APC (Product # 17-5971-82) and subsequently stained intracellularly with Anti-Human Granzyme B PE.
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Human naïve CD8 T cells from 4 healthy donors were stimulated with PMA (50ng/ml) plus ionomycin (500ng/ml) for 2 hrs, and stained with PE anti-human Granzyme B antibody (Product # 12-8899-41) at the concentration of 1:100. Data courtesy of the antibody data exchange program.
- Conjugate
- Yellow dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
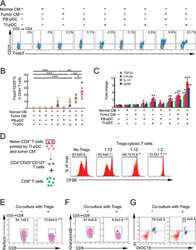
- Experimental details
- Figure 3 Naive CD4 + T cells are converted to functional Tregs by tumor-infiltrating DCs and tumor conditioned medium (CM). (A-C) Naive CD4 + T cells from peripheral blood of patients with invasive breast carcinoma were co-cultured with or without autologous pDCs isolated from tumor (TI) or peripheral blood (PB) for 9 days in the presence or absence of 30% CM from autologous tumor slices or adjacent normal tissue slices. (A , B) Non-adherent cells from co-cultures were stained for CD3, CD4, CD25 and intracellular Foxp3, and analyzed by flow cytometry. Representative plots of gated CD3 + CD4 + cells (A) and quantification of percentage of Foxp3 + CD25 + cells among CD3 + CD4 + cells (B) are shown (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 by Student's t -test). (C) Expression of Treg-associated genes, assessed by qRT-PCR normalized to GAPDH , in sorted CD4 + T cells, relative to expression in cultures without DCs or CM (mean +- SEM, n = 19; * P < 0.05, ** P < 0.01, *** P < 0.001 compared with naive CD4 + T cells cultured alone by Student's t -test). (D-G) Effect of naive CD4 + T cell-derived Tregs, obtained by co-culture with TI pDCs and tumor CM as above, on function of autologous tumor-specific CD8 + T cells. Tumor-specific CD8 + T cells were generated for each subject by stimulating autologous PB CD8 + T cells with autologous tumor lysate-pulsed autologous DCs. Tregs were recovered from co-cultures by magnetic sorting. (D) CFSE-labeled CD8 + T ce
- Conjugate
- Yellow dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry