Antibody data
- Antibody Data
- Antigen structure
- References [15]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [10]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 17-9185-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD185 (CXCR5) Monoclonal Antibody (MU5UBEE), APC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The MU5UBEE monoclonal antibody reacts with human and non-human primate CD185. CD185, which is also known as C-X-C chemokine receptor 5 (CXCR5) and Burkitt lymphoma receptor 1 (BLR1), is a seven transmembrane G protein-coupled receptor originally identified in Burkitt's lymphoma. In peripheral blood, CD185 is expressed on B cells, CD4+ T cells (but not Th1 or Th2 cells), as well as on a subpopulation of memory (CD45RO+) T cells. Circulating CD185+ T cells are in a resting state and migrate to the lymph nodes due to expression of CCR7 and CD62L. In tonsil, CD185 is expressed on nearly all CD4+ cells together with CD45RO and activation markers such as CD69 and ICOS. Tonsillar CD185+ cells have been shown to induce antibody production when co-cultured with B cells, thus supporting their role in providing help to B cells. Furthermore, this chemokine receptor plays a critical role in lymphocyte trafficking, in particular T cell migration into the B cell follicles of germinal centers in response to CXCL13, making CD185 an established marker of follicular helper T cells. The MU5UBEE monoclonal antibody also crossreacts with Rhesus macaque. Applications Reported: This MU5UBEE antibody has been reported for use in flow cytometric analysis. Applications Tested: This MU5UBEE antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.125 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 633-647 nm; Emission: 660 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- MU5UBEE
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references In Situ Characterization of Human Lymphoid Tissue Immune Cells by Multispectral Confocal Imaging and Quantitative Image Analysis; Implications for HIV Reservoir Characterization.
NK-B cell cross talk induces CXCR5 expression on natural killer cells.
Th2 Biased Immunity With Altered B Cell Profiles in Circulation of Patients With Sporotrichosis Caused by Sporothrix globosa.
Rapid Transduction and Expansion of Transduced T Cells with Maintenance of Central Memory Populations.
E4BP4-mediated inhibition of T follicular helper cell differentiation is compromised in autoimmune diseases.
Dynamics of NK, CD8 and Tfh cell mediated the production of cytokines and antiviral antibodies in Chinese patients with moderate COVID-19.
S-Geranylgeranyl-L-glutathione is a ligand for human B cell-confinement receptor P2RY8.
Vaccine induction of antibodies and tissue-resident CD8+ T cells enhances protection against mucosal SHIV-infection in young macaques.
Distribution of circulating follicular helper T cells and expression of interleukin-21 and chemokine C-X-C ligand 13 in gastric cancer.
Elicitation of Robust Tier 2 Neutralizing Antibody Responses in Nonhuman Primates by HIV Envelope Trimer Immunization Using Optimized Approaches.
Cytokine-secreting follicular T cells shape the antibody repertoire.
Systemic immunoregulatory and pathogenic functions of homeostatic chemokine receptors.
A putative chemokine receptor, BLR1, directs B cell migration to defined lymphoid organs and specific anatomic compartments of the spleen.
Sequence variation of a novel heptahelical leucocyte receptor through alternative transcript formation.
Differentiation-specific expression of a novel G protein-coupled receptor from Burkitt's lymphoma.
Moysi E, Del Rio Estrada PM, Torres-Ruiz F, Reyes-Terán G, Koup RA, Petrovas C
Frontiers in immunology 2021;12:683396
Frontiers in immunology 2021;12:683396
NK-B cell cross talk induces CXCR5 expression on natural killer cells.
Rascle P, Jacquelin B, Petitdemange C, Contreras V, Planchais C, Lazzerini M, Dereuddre-Bosquet N, Le Grand R, Mouquet H, Huot N, Müller-Trutwin M
iScience 2021 Oct 22;24(10):103109
iScience 2021 Oct 22;24(10):103109
Th2 Biased Immunity With Altered B Cell Profiles in Circulation of Patients With Sporotrichosis Caused by Sporothrix globosa.
Zu J, Yao L, Song Y, Cui Y, Guan M, Chen R, Zhen Y, Li S
Frontiers in immunology 2020;11:570888
Frontiers in immunology 2020;11:570888
Rapid Transduction and Expansion of Transduced T Cells with Maintenance of Central Memory Populations.
Pampusch MS, Haran KP, Hart GT, Rakasz EG, Rendahl AK, Berger EA, Connick E, Skinner PJ
Molecular therapy. Methods & clinical development 2020 Mar 13;16:1-10
Molecular therapy. Methods & clinical development 2020 Mar 13;16:1-10
E4BP4-mediated inhibition of T follicular helper cell differentiation is compromised in autoimmune diseases.
Wang Z, Zhao M, Yin J, Liu L, Hu L, Huang Y, Liu A, Ouyang J, Min X, Rao S, Zhou W, Wu H, Yoshimura A, Lu Q
The Journal of clinical investigation 2020 Jul 1;130(7):3717-3733
The Journal of clinical investigation 2020 Jul 1;130(7):3717-3733
Dynamics of NK, CD8 and Tfh cell mediated the production of cytokines and antiviral antibodies in Chinese patients with moderate COVID-19.
Yan L, Cai B, Li Y, Wang MJ, An YF, Deng R, Li DD, Wang LC, Xu H, Gao XD, Wang LL
Journal of cellular and molecular medicine 2020 Dec;24(24):14270-14279
Journal of cellular and molecular medicine 2020 Dec;24(24):14270-14279
S-Geranylgeranyl-L-glutathione is a ligand for human B cell-confinement receptor P2RY8.
Lu E, Wolfreys FD, Muppidi JR, Xu Y, Cyster JG
Nature 2019 Mar;567(7747):244-248
Nature 2019 Mar;567(7747):244-248
Vaccine induction of antibodies and tissue-resident CD8+ T cells enhances protection against mucosal SHIV-infection in young macaques.
Petitdemange C, Kasturi SP, Kozlowski PA, Nabi R, Quarnstrom CF, Reddy PBJ, Derdeyn CA, Spicer LM, Patel P, Legere T, Kovalenkov YO, Labranche CC, Villinger F, Tomai M, Vasilakos J, Haynes B, Kang CY, Gibbs JS, Yewdell JW, Barouch D, Wrammert J, Montefiori D, Hunter E, Amara RR, Masopust D, Pulendran B
JCI insight 2019 Feb 21;4(4)
JCI insight 2019 Feb 21;4(4)
Distribution of circulating follicular helper T cells and expression of interleukin-21 and chemokine C-X-C ligand 13 in gastric cancer.
Meng X, Yu X, Dong Q, Xu X, Li J, Xu Q, Ma J, Zhou C
Oncology letters 2018 Sep;16(3):3917-3922
Oncology letters 2018 Sep;16(3):3917-3922
Elicitation of Robust Tier 2 Neutralizing Antibody Responses in Nonhuman Primates by HIV Envelope Trimer Immunization Using Optimized Approaches.
Pauthner M, Havenar-Daughton C, Sok D, Nkolola JP, Bastidas R, Boopathy AV, Carnathan DG, Chandrashekar A, Cirelli KM, Cottrell CA, Eroshkin AM, Guenaga J, Kaushik K, Kulp DW, Liu J, McCoy LE, Oom AL, Ozorowski G, Post KW, Sharma SK, Steichen JM, de Taeye SW, Tokatlian T, Torrents de la Peña A, Butera ST, LaBranche CC, Montefiori DC, Silvestri G, Wilson IA, Irvine DJ, Sanders RW, Schief WR, Ward AB, Wyatt RT, Barouch DH, Crotty S, Burton DR
Immunity 2017 Jun 20;46(6):1073-1088.e6
Immunity 2017 Jun 20;46(6):1073-1088.e6
Cytokine-secreting follicular T cells shape the antibody repertoire.
Reinhardt RL, Liang HE, Locksley RM
Nature immunology 2009 Apr;10(4):385-93
Nature immunology 2009 Apr;10(4):385-93
Systemic immunoregulatory and pathogenic functions of homeostatic chemokine receptors.
Müller G, Höpken UE, Stein H, Lipp M
Journal of leukocyte biology 2002 Jul;72(1):1-8
Journal of leukocyte biology 2002 Jul;72(1):1-8
A putative chemokine receptor, BLR1, directs B cell migration to defined lymphoid organs and specific anatomic compartments of the spleen.
Förster R, Mattis AE, Kremmer E, Wolf E, Brem G, Lipp M
Cell 1996 Dec 13;87(6):1037-47
Cell 1996 Dec 13;87(6):1037-47
Sequence variation of a novel heptahelical leucocyte receptor through alternative transcript formation.
Barella L, Loetscher M, Tobler A, Baggiolini M, Moser B
The Biochemical journal 1995 Aug 1;309 ( Pt 3):773-9
The Biochemical journal 1995 Aug 1;309 ( Pt 3):773-9
Differentiation-specific expression of a novel G protein-coupled receptor from Burkitt's lymphoma.
Dobner T, Wolf I, Emrich T, Lipp M
European journal of immunology 1992 Nov;22(11):2795-9
European journal of immunology 1992 Nov;22(11):2795-9
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
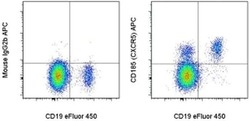
- Experimental details
- Staining of normal human peripheral blood cells with Anti-Human CD19 eFluor® 450 (Product # 48-0199-42) and Mouse IgG2b K Isotype Control APC (Product # 17-4732-81) (left) or Anti-Human CD185 (CXCR5) APC (right). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
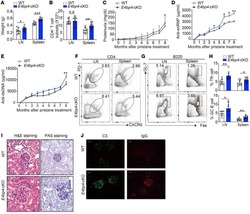
- Figure 4 Extended Immunogen Release Induces Higher nAb Titers Than Conventional Immunization (A-E) Immunogen doses of 100 or 20 mug s.c. immunizations of BG505 SOSIP.664. (A) BG505 nAb titers at week 26 (n = 6 or 12). (B) BG505 SOSIP binding titers at week 26 (n = 6 or 12). (C) Kinetics of BG505 nAb titers. (D and E) GC B cell (D) and GC Tfh cell (E) frequencies after the first, second, and third immunizations. (F-L) Bolus (conventional) versus continuous immunogen delivery of BG505 SOSIP.v5.2 immunogen. (F) Immunization schedule and sampling for continuous antigen delivery using osmotic pumps. (G) BG505 nAb titers in animals immunized by osmotic pump (red) or conventional bolus (Conv, black) ( * p < 0.05; ** p < 0.01; n = 6). (H) Peak BG505 nAb titers after the third immunization (n = 6). (I and J) GC B cell (I) and GC Tfh cell (J) frequencies after the first, second, and third immunizations. (K) Proliferation of GC Tfh cells at week 11. Flow cytometry was gated on CXCR5 hi PD-1 hi GC Tfh cells. (L) Frequency of Ki67 + GC Tfh cells at week 11 (n = 12). All nAb titer and ELISA binding Ab data represent geometric mean titers with geometric SD. All cell-frequency data represent the mean and SD. See also Figure S4 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
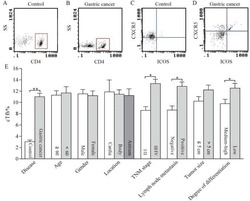
- Experimental details
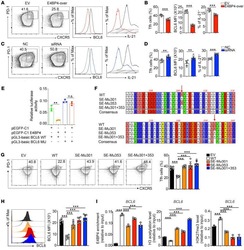
- Figure 4 Unchanged cTfh cells and altered Ig profile in sporotrichosis patients. (A) Statistical graphs in the upper row were for CD4 + CXCR5 + Tfh while the lower row were for CD4 + CXCR5 + PD1 + Tfh. The comparison of cTfh percentages were between HC (n = 24) and whole patients (n = 50), patients with different duration (< 6 mon, n = 24; > 6 mon, n = 26) and clinical types (FF, n = 33; LF, n = 17). (B) Comparison of serum levels of total IgG, IgM, and IgE between patients (n = 46) and HC (n = 24). (C, D) Distribution of IgG subtypes (IgG1, IgG2, and IgG3 and IgG4) in patients (in whole: n = 20; SD: n = 7; LD: n = 13) and HC (n = 19). Error bars represent mean+-SD. * P < 0.05, ** P < 0.01, and NS P >= 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
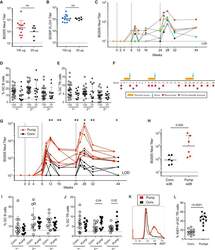
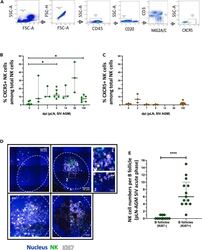
- Figure 1 Temporal and spatial dynamics of CXCR5+ NK cells in peripheral lymph nodes during primary SIVagm infection (A) Gating strategy of CXCR5+ NK cells in pLN. NK cells were gated as CD45 + CD3 - CD20 - NKG2A/C + CXCR5 + cells as previously reported (; , p. 80). A representative image is shown. The example corresponds to pLN cells from an African Green Monkey infected by SIVagm at day 9 post-infection. (B) Kinetic of CXCR5 + NK cell percentages in pLN during SIVagm infection, dpi = days post-infection. Each green circle indicates an individual animal (depending on the time point, n = 3 or 6 AGM); p value = *
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
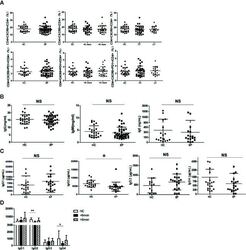
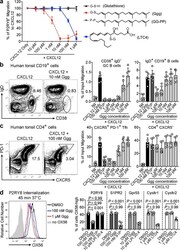
- Figure 2. Ggg inhibits the migration of P2RY8-expressing cells. (a) P2RY8 ligand bioassay using the indicated concentrations of Ggg, glutathione (GSH), GG-PP, or LTC4 with 50 ng/mL CXCL12 (n=4 biological replicates). (b, c) Transwell migration assays of tonsil cells towards CXCL12 mixed with the indicated concentrations of Ggg. Left plots, representative flow cytometry of CD19 + cells showing the gate for CD38 + IgD - GC B cells (b) or of CD4 + cells showing the gate for PD-1 + , CXCR5 + Tfh cells (c). Right graphs show summary data for indicated cell types. (n=3 tonsils, 2 technical replicates each). (d) Internalization assay using cells expressing OX56 epitope-tagged P2RY8, read by measuring surface OX56 levels (representative flow cytometry histogram, left). Right graphs show summarized data for the indicated receptors (n=6 biological replicates, one-way ANOVA with Bonferroni's multiple comparisons test). Data are pooled from 3 experiments (a,b,c,d). Graphs depict mean with s.d.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
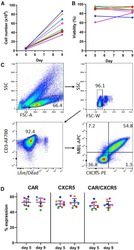
- Figure 2 9-Day Transduction and Expansion Protocol Yields Sufficient Cells for Infusion, Which Are Viable and Maintain Co-expression of Two Transduced Genes (A) Total number of cells in culture on days 5 and 9 of the expansion protocol and (B) viability of the transduced cells using PBMCs from seven different animals. Cell number and viability was monitored by trypan blue exclusion counting on the countess cell counter. (C) Representative flow plots showing gating strategy and expression of both the CD4-MBL CAR and CXCR5 in transduced cells on day 9. (D) Expression of CAR, CXCR5, and co-expression of CAR and CXCR5 on days 5 and 9 in cells from seven different animals. The bars represent the mean value and 95% confidence interval.
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry