Antibody data
- Antibody Data
- Antigen structure
- References [7]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [9]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 62-9185-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD185 (CXCR5) Monoclonal Antibody (MU5UBEE), Super Bright™ 436, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The MU5UBEE monoclonal antibody reacts with human and non-human primate CD185. CD185, which is also known as C-X-C chemokine receptor 5 (CXCR5) and Burkitt lymphoma receptor 1 (BLR1), is a seven transmembrane G protein-coupled receptor originally identified in Burkitt's lymphoma. In peripheral blood, CD185 is expressed on B cells, CD4+ T cells (but not Th1 or Th2 cells), as well as on a subpopulation of memory (CD45RO+) T cells. Circulating CD185+ T cells are in a resting state and migrate to the lymph nodes due to expression of CCR7 and CD62L. In tonsil, CD185 is expressed on nearly all CD4+ cells together with CD45RO and activation markers such as CD69 and ICOS. Tonsillar CD185+ cells have been shown to induce antibody production when co-cultured with B cells, thus supporting their role in providing help to B cells. Furthermore, this chemokine receptor plays a critical role in lymphocyte trafficking, in particular T cell migration into the B cell follicles of germinal centers in response to CXCL13, making CD185 an established marker of follicular helper T cells. Applications Reported: This MU5UBEE antibody has been reported for use in flow cytometric analysis. Applications Tested: This MU5UBEE antibody has been pre-diluted and tested by flow cytometric analysis of normal human peripheral blood cells. This may be used at 5 µL (1.0 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Super Bright 436 can be excited with the violet laser line (405 nm) and emits at 436 nm. We recommend using a 450/50 bandpass filter, or equivalent. Please make sure that your instrument is capable of detecting this fluorochrome. When using two or more Super Bright dye-conjugated antibodies in a staining panel, it is recommended to use Super Bright Complete Staining Buffer (Product # SB-4401) to minimize any non-specific polymer interactions. Please refer to the datasheet for Super Bright Staining Buffer for more information. Excitation: 405 nm; Emission: 436 nm; Laser: Violet Laser Super Bright Polymer Dyes are sold under license from Becton, Dickinson and Company.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- MU5UBEE
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references SARS-CoV-2 mRNA vaccination elicits a robust and persistent T follicular helper cell response in humans.
In Situ Characterization of Human Lymphoid Tissue Immune Cells by Multispectral Confocal Imaging and Quantitative Image Analysis; Implications for HIV Reservoir Characterization.
NK-B cell cross talk induces CXCR5 expression on natural killer cells.
Th2 Biased Immunity With Altered B Cell Profiles in Circulation of Patients With Sporotrichosis Caused by Sporothrix globosa.
Rapid Transduction and Expansion of Transduced T Cells with Maintenance of Central Memory Populations.
E4BP4-mediated inhibition of T follicular helper cell differentiation is compromised in autoimmune diseases.
Rapid Germinal Center and Antibody Responses in Non-human Primates after a Single Nanoparticle Vaccine Immunization.
Mudd PA, Minervina AA, Pogorelyy MV, Turner JS, Kim W, Kalaidina E, Petersen J, Schmitz AJ, Lei T, Haile A, Kirk AM, Mettelman RC, Crawford JC, Nguyen THO, Rowntree LC, Rosati E, Richards KA, Sant AJ, Klebert MK, Suessen T, Middleton WD, SJTRC Study Team, Wolf J, Teefey SA, O'Halloran JA, Presti RM, Kedzierska K, Rossjohn J, Thomas PG, Ellebedy AH
Cell 2022 Feb 17;185(4):603-613.e15
Cell 2022 Feb 17;185(4):603-613.e15
In Situ Characterization of Human Lymphoid Tissue Immune Cells by Multispectral Confocal Imaging and Quantitative Image Analysis; Implications for HIV Reservoir Characterization.
Moysi E, Del Rio Estrada PM, Torres-Ruiz F, Reyes-Terán G, Koup RA, Petrovas C
Frontiers in immunology 2021;12:683396
Frontiers in immunology 2021;12:683396
NK-B cell cross talk induces CXCR5 expression on natural killer cells.
Rascle P, Jacquelin B, Petitdemange C, Contreras V, Planchais C, Lazzerini M, Dereuddre-Bosquet N, Le Grand R, Mouquet H, Huot N, Müller-Trutwin M
iScience 2021 Oct 22;24(10):103109
iScience 2021 Oct 22;24(10):103109
Th2 Biased Immunity With Altered B Cell Profiles in Circulation of Patients With Sporotrichosis Caused by Sporothrix globosa.
Zu J, Yao L, Song Y, Cui Y, Guan M, Chen R, Zhen Y, Li S
Frontiers in immunology 2020;11:570888
Frontiers in immunology 2020;11:570888
Rapid Transduction and Expansion of Transduced T Cells with Maintenance of Central Memory Populations.
Pampusch MS, Haran KP, Hart GT, Rakasz EG, Rendahl AK, Berger EA, Connick E, Skinner PJ
Molecular therapy. Methods & clinical development 2020 Mar 13;16:1-10
Molecular therapy. Methods & clinical development 2020 Mar 13;16:1-10
E4BP4-mediated inhibition of T follicular helper cell differentiation is compromised in autoimmune diseases.
Wang Z, Zhao M, Yin J, Liu L, Hu L, Huang Y, Liu A, Ouyang J, Min X, Rao S, Zhou W, Wu H, Yoshimura A, Lu Q
The Journal of clinical investigation 2020 Jul 1;130(7):3717-3733
The Journal of clinical investigation 2020 Jul 1;130(7):3717-3733
Rapid Germinal Center and Antibody Responses in Non-human Primates after a Single Nanoparticle Vaccine Immunization.
Havenar-Daughton C, Carnathan DG, Boopathy AV, Upadhyay AA, Murrell B, Reiss SM, Enemuo CA, Gebru EH, Choe Y, Dhadvai P, Viviano F, Kaushik K, Bhiman JN, Briney B, Burton DR, Bosinger SE, Schief WR, Irvine DJ, Silvestri G, Crotty S
Cell reports 2019 Nov 12;29(7):1756-1766.e8
Cell reports 2019 Nov 12;29(7):1756-1766.e8
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
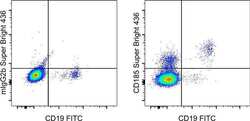
- Experimental details
- Normal human peripheral blood cells cells were stained with CD19 Monoclonal Antibody, FITC (Product # 11-0199-42) and Mouse IgG2b kappa Isotype Control, Super Bright 436 (Product # 62-4732-82) (left) or CD185 Monoclonal Antibody, Super Bright 436 (right). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
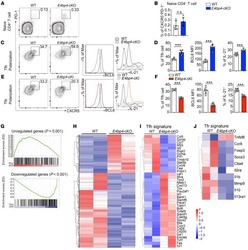
- Figure 4 Unchanged cTfh cells and altered Ig profile in sporotrichosis patients. (A) Statistical graphs in the upper row were for CD4 + CXCR5 + Tfh while the lower row were for CD4 + CXCR5 + PD1 + Tfh. The comparison of cTfh percentages were between HC (n = 24) and whole patients (n = 50), patients with different duration (< 6 mon, n = 24; > 6 mon, n = 26) and clinical types (FF, n = 33; LF, n = 17). (B) Comparison of serum levels of total IgG, IgM, and IgE between patients (n = 46) and HC (n = 24). (C, D) Distribution of IgG subtypes (IgG1, IgG2, and IgG3 and IgG4) in patients (in whole: n = 20; SD: n = 7; LD: n = 13) and HC (n = 19). Error bars represent mean+-SD. * P < 0.05, ** P < 0.01, and NS P >= 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 1 Temporal and spatial dynamics of CXCR5+ NK cells in peripheral lymph nodes during primary SIVagm infection (A) Gating strategy of CXCR5+ NK cells in pLN. NK cells were gated as CD45 + CD3 - CD20 - NKG2A/C + CXCR5 + cells as previously reported (; , p. 80). A representative image is shown. The example corresponds to pLN cells from an African Green Monkey infected by SIVagm at day 9 post-infection. (B) Kinetic of CXCR5 + NK cell percentages in pLN during SIVagm infection, dpi = days post-infection. Each green circle indicates an individual animal (depending on the time point, n = 3 or 6 AGM); p value = *
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 3 CXCR5+ NK cell induction and gene expression profile (A) Staining of NK cells co-cultured with autologous B cells separated by a transwell membrane illustrating CXCR5 expression on NK cells. The CD3 labeling is used here as a negative control. (B) Dot plot representative of CXCR5+ NK cells from uninfected AGM after 6 days of culture: NK cells cultured alone and NK cells co-cultured with autologous B cells separated by a transwell membrane. (C) The fold change of CXCR5+ NK cell frequencies among total NK cells compared with the frequency of CXCR5+ NK cells in NK cells cultured alone is shown after 6 days of co-culture. B and NK cells were isolated from PBMC of 6 non-infected AGMs (p value = *
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
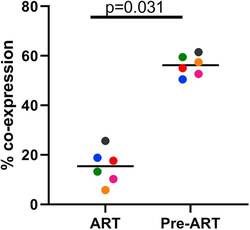
- Experimental details
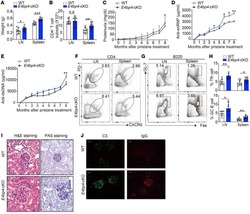
- Figure 3 Treatment of Macaques with Antiretroviral Drugs Prior to Collection of PBMCs Leads to a Reduction of Transduction Efficiency with Gammaretroviral Vectors PBMCs from six animals using cells collected prior to treatment with anti-retroviral drugs (Pre-ART) or during antiretroviral treatment (ART) were used in the 9-day transduction and expansion protocol. On day 9, cells were evaluated for expression of MBL, representing the CAR, and CXCR5 by flow cytometry. A Wilcoxon matched pairs signed rank test was used to determine significance. Colors denote cells from six individual animals. The bar represents the median value.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
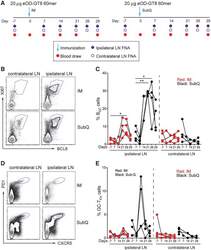
- Experimental details
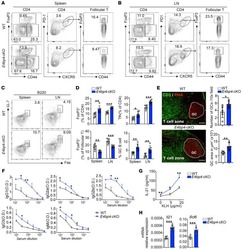
- Figure 2. Longitudinal Sampling of GCs by LN FNA after eOD-GT8 60-mer Immunization (A) Schematic of LN FNA and blood sampling after IM (left) or s.c. (right) immunization. (B) Flow cytometry identification of B GC cell frequencies in the ipsilateral (draining) and contralateral (non-draining) LNs after s.c. and IM immunization. B GC cells are KI67 + BCL6 + . Full gating is Figure S2 . (C) Weekly sampling of B GC cell frequency after one immunization. *p < 0.05, **p < 0.01 (paired t test, one tailed). (D) Flow cytometry identification of GC-T FH cell frequencies in the ipsilateral (draining) and contralateral (non-draining) LNs after s.c. and IM immunization. GC-T FH cells are CXCR5 + PD1 + . (E) Weekly sampling of GC-T FH cell frequency after one immunization. Gated on CD4 + T cells. Each point represents an individual LN FNA sample. n = 8, four LN FNAs per immunization condition at each time point. See also Figures S2 and S3 .
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry