Antibody data
- Antibody Data
- Antigen structure
- References [3]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [3]
- Immunohistochemistry [6]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-52369 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- RAB7 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Recombinant protein fragment
- Description
- Immunogen sequence: CVLVFDVTAP NTFKTLDSWR DEFLIQASPR DPENFPFVVL GNKIDLENRQ VATKRAQAWC YSKNNIPYFE TSAKEAINVE QAFQTIARNA LKQETEVELY NEFPEPIKLD KNDRAK Highest antigen sequence identity to the following orthologs: Mouse - 100%, Rat - 99%.
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 μL
- Concentration
- 0.20 mg/mL
- Storage
- Store at 4°C short term. For long term storage, store at -20°C, avoiding freeze/thaw cycles.
Submitted references Dysregulation of endocytic machinery and ACE2 in small airways of smokers and COPD patients can augment their susceptibility to SARS-CoV-2 (COVID-19) infections.
Major Determinants of Airway Epithelial Cell Sensitivity to S. aureus Alpha-Toxin: Disposal of Toxin Heptamers by Extracellular Vesicle Formation and Lysosomal Degradation.
α- Linolenic acid modulates phagocytosis and endosomal pathways of extracellular Tau in microglia.
Eapen MS, Lu W, Hackett TL, Singhera GK, Thompson IE, McAlinden KD, Hardikar A, Weber HC, Haug G, Wark PAB, Chia C, Sohal SS
American journal of physiology. Lung cellular and molecular physiology 2021 Jan 1;320(1):L158-L163
American journal of physiology. Lung cellular and molecular physiology 2021 Jan 1;320(1):L158-L163
Major Determinants of Airway Epithelial Cell Sensitivity to S. aureus Alpha-Toxin: Disposal of Toxin Heptamers by Extracellular Vesicle Formation and Lysosomal Degradation.
Möller N, Ziesemer S, Hentschker C, Völker U, Hildebrandt JP
Toxins 2021 Feb 24;13(3)
Toxins 2021 Feb 24;13(3)
α- Linolenic acid modulates phagocytosis and endosomal pathways of extracellular Tau in microglia.
Desale SE, Chinnathambi S
Cell adhesion & migration 2021 Dec;15(1):84-100
Cell adhesion & migration 2021 Dec;15(1):84-100
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of RAB7 in A-431 cells transfected with control siRNA, target specific siRNA probe #1 and #2, using a RAB7 Polyclonal Antibody (Product # PA5-52369). Remaining relative intensity is presented. Loading control: Anti-GAPDH.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of RAB7 in mouse cell line NIH-3T3 and rat cell line NBT-II using a RAB7 Polyclonal Antibody (Product # PA5-52369).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
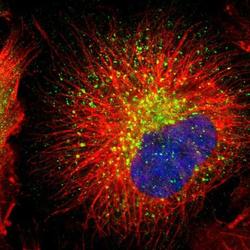
- Experimental details
- Immunofluorescent staining of RAB7 in human cell line U-251 MG shows positivity in vesicles. Samples were probed using a RAB7 Polyclonal Antibody (Product # PA5-52369).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
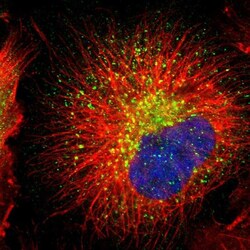
- Experimental details
- Immunofluorescent staining of RAB7 in human cell line U-251 MG using a RAB7 Polyclonal Antibody (Product # PA5-52369) shows localization to lysosomes.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
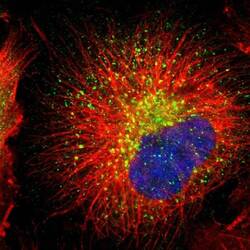
- Experimental details
- Immunofluorescent staining of RAB7 in human cell line U-251 MG using a RAB7 Polyclonal Antibody (Product # PA5-52369) shows localization to lysosomes.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
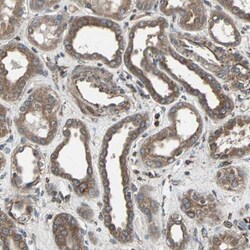
- Experimental details
- Immunohistochemical staining of RAB7 in human kidney using a RAB7 Polyclonal Antibody (Product # PA5-52369) shows moderate to strong cytoplasmic positivity in cells in tubules.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
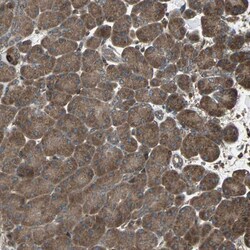
- Experimental details
- Immunohistochemical staining of RAB7 in human pancreas using a RAB7 Polyclonal Antibody (Product # PA5-52369) shows moderate to strong cytoplasmic positivity in exocrine glandular cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
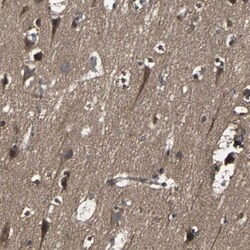
- Experimental details
- Immunohistochemical staining of RAB7 in human cerebral cortex using a RAB7 Polyclonal Antibody (Product # PA5-52369) shows moderate to strong cytoplasmic positivity in neuronal cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
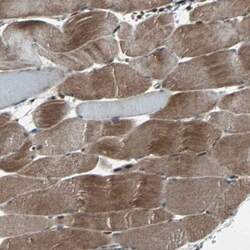
- Experimental details
- Immunohistochemical staining of RAB7 in human skeletal muscle using a RAB7 Polyclonal Antibody (Product # PA5-52369) shows moderate to strong cytoplasmic positivity in myocytes.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
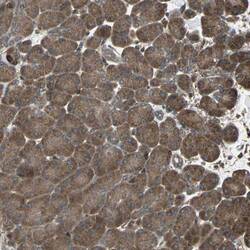
- Experimental details
- Immunohistochemical staining of RAB7 in human pancreas using a RAB7 Polyclonal Antibody (Product # PA5-52369) shows moderate to strong cytoplasmic positivity in exocrine glandular cells.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
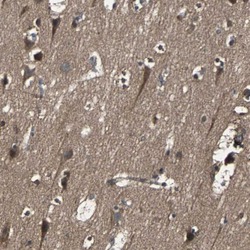
- Experimental details
- Immunohistochemical staining of RAB7 in human cerebral cortex using a RAB7 Polyclonal Antibody (Product # PA5-52369) shows moderate to strong cytoplasmic positivity in neuronal cells.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
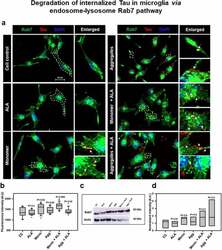
- Experimental details
- Figure 4. Degradation of internalized Tau in microglia via endosome-lysosme Rab7 pathway . Fate of internalized Tau was observed with the help of late endosomal marker Rab7 by fluorescence microscopy. (a) The colocalization of internalized Tau was observed with late endosomal marker Rab7, and the enlarged area indicates the specific area of colocalization inside the cell. The white arrowmarks denote the position and colocalization of internalized Tau with Rab7; scale bar is 20 mum. (b) The intensity analysis of Rab7 plotted as a mean intensity of Rab7; significance is P < 0.05. (c) Expression profile of Rab7 was analyzed by western blot after exposure of extracellular Tau. (d) The quantification of protein bands was carried out by calculating the intensity and normalized with beta-actin as a loading control
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
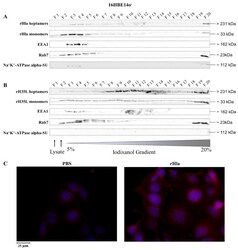
- Experimental details
- Figure 2 Detection of internalized rHla in 16HBE14o - cells. 16HBE14o - cells were treated for 2.5 h with 2000 ng/mL rHla ( A ) or rH35L ( B ). Cells were mechanically disrupted and post-nuclear supernatants were obtained by centrifugation. These were layered on a continuous iodixanol density gradient (5%-20%). Cellular vesicles and organelles were separated by ultracentrifugation according to their density. Fractions (500 uL) were analyzed by Western blotting for rHla heptamers and monomers, for EEA1 as a marker protein for early endosomes or for Rab7 as a marker protein for late endosomes, autophagosomes or lysosomes. Detection of the alpha-subunit of Na + K + -ATPase indicated possible contamination of fractions with plasma membrane. Panel ( C ) shows representative examples of fluorescence microscopy images of 16HBE14o - cells incubated with 2000 ng/mL rHla (right) or with PBS as a control (left) for 2 h. Incubation with a Chromeo 546 antibody conjugate against Strep-tag II of rHla was intended to visualize internalized rHla accumulated in vesicles (red staining of vesicles). The cell nuclei were counterstained with 4'',6-Diamidin-2-phenylindol (DAPI). To better see signals in HBE cells, the contrast and brightness of the respective example image of the rHla-treated and PBS-treated cells were adjusted to the same extent.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
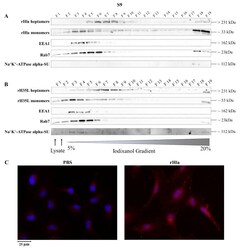
- Experimental details
- Figure 3 Detection of internalized rHla in S9 cells. S9 cells were treated for 2.5 h with 2000 ng/mL rHla ( A ) or rH35L ( B ). Cells were mechanically disrupted and post-nuclear supernatants were obtained by centrifugation. These were layered on a continuous iodixanol density gradient (5%-20%). Cellular vesicles and organelles were separated by ultracentrifugation according to their density. Fractions (500 uL) were analyzed by Western blotting for rHla heptamers and monomers, for EEA1 as a marker protein for early endosomes or for Rab7 as a marker protein for late endosomes, autophagosomes or lysosomes. Detection of the alpha-subunit of Na + K + -ATPase indicated possible contamination of fractions with plasma membrane. Panel ( C ) shows representative examples of fluorescence microscopy images of S9 cells incubated with 2000 ng/mL rHla (right) or with PBS as a control (left) for 2 h. Incubation with a Chromeo 546 antibody conjugate against Strep-tag II of rHla was intended to visualize internalized rHla accumulated in vesicles (red staining of vesicles). The cell nuclei were counterstained with DAPI. Note the strong rHla-specific immunofluorescence signals in intracellular granules in alpha-toxin exposed S9 cells ( Figure 3 C, right panel).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
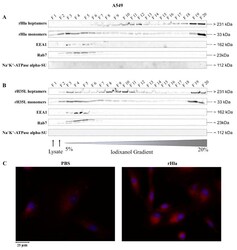
- Figure 4 Detection of internalized rHla in A549 cells. A549 cells were treated for 2.5 h with 2000 ng/mL rHla ( A ) or rH35L ( B ). Cells were mechanically disrupted and post-nuclear supernatants were obtained by centrifugation. These were layered on a continuous iodixanol density gradient (5%-20%). Cellular vesicles and organelles were separated by ultracentrifugation according to their density. Fractions (500 uL) were analyzed by Western blotting for rHla heptamers and monomers, for EEA1 as a marker protein for early endosomes or for Rab7 as a marker protein for late endosomes, autophagosomes or lysosomes. Detection of the alpha-subunit of Na + K + -ATPase indicated possible contamination of fractions with plasma membrane. Panel ( C ) shows representative examples of fluorescence microscopy images of A549 cells incubated with 2000 ng/mL rHla (right) or with PBS as a control (left) for 2 h. Incubation with a Chromeo 546 antibody conjugate against Strep-tag II of rHla was intended to visualize internalized rHla accumulated in vesicles (red staining of vesicles). The cell nuclei were counterstained with DAPI. Note the diffuse rHla-specific immunofluorescence signals in intracellular granules in alpha-toxin exposed A549 cells ( Figure 4 C, right panel).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
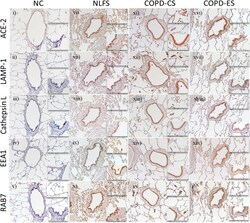
- Figure 3. Representative images of serial sections from surgical lung resections stained for angiotensin-converting enzyme-2 (ACE2) receptor, lysosomal associated membrane protein1 (LAMP-1), cathepsin-L, early endosomal marker 1 (EEA1), and late endosomal marker (RAB7). The observation was done using brightfield microscopy at a magnification of x10 and enlarged insets at x40 magnification for the small airway wall and parenchymal spaces. Normal nonsmoker ( i - v ) SA tissues observed to have a low expression for ACE-2, cathepsin-L, and endocytic markers when compared with NLFS ( vi - x ), chronic obstructive pulmonary disease (COPD)-current smokers (CS) ( xi - xv ), and COPD-ex-smokers (ES) ( xvi - xx ). Compared with NLFS and COPS-CS, the expression levels of ACE-2 and endocytic makers were observed to be comparably lesser in ex-smokers across the 5 markers tested. A similar pattern was also observed in type 2 pneumocytes and macrophages across the patient population ( insets ). LAMP-1 expression in small airway epithelium appeared to be more diffused across the epithelium in COPD-CS ( xii , inset ) while in both normal lung function smokers (NLFS; vii , inset ) and COPD-ES ( xvii , inset ) the LAMP-1 was found to accumulate close to the nucleus. NC, normal control; LP lamina propria.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot