Antibody data
- Antibody Data
- Antigen structure
- References [4]
- Comments [0]
- Validations
- Immunocytochemistry [6]
- Flow cytometry [6]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA2607 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD26 Monoclonal Antibody (236.3)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- MA2607 targets CD26 in FACS, IHC, IF/ICC and IP applications and shows reactivity with Human and Rat samples. The MA2607 immunogen is rat 110-120 kD single chain glycoprotein Depeptidyl Peptidase IV (CD26). MA2607 detects CD26 which has a predicted molecular weight of approximately 88 kDa. This product is a Low Endotoxin formulation. This product has been tested for endotoxins by limulus amoebocyte lysate (LAL) assay and contains an endotoxin concentration of less than or equal to 10 endotoxin units per milligram (EU/mg).
- Reactivity
- Human, Rat
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 236.3
- Vial size
- 500 μg
- Concentration
- 1.0 mg/mL
- Storage
- -20°C
Submitted references TMPRSS2 and TMPRSS4 promote SARS-CoV-2 infection of human small intestinal enterocytes.
Insulin-dependent phosphorylation of DPP IV in liver. Evidence for a role of compartmentalized c-Src.
A Fischer rat substrain deficient in dipeptidyl peptidase IV activity makes normal steady-state RNA levels and an altered protein. Use as a liver-cell transplantation model.
A Fischer rat substrain deficient in dipeptidyl peptidase IV activity makes normal steady-state RNA levels and an altered protein. Use as a liver-cell transplantation model.
Zang R, Gomez Castro MF, McCune BT, Zeng Q, Rothlauf PW, Sonnek NM, Liu Z, Brulois KF, Wang X, Greenberg HB, Diamond MS, Ciorba MA, Whelan SPJ, Ding S
Science immunology 2020 May 13;5(47)
Science immunology 2020 May 13;5(47)
Insulin-dependent phosphorylation of DPP IV in liver. Evidence for a role of compartmentalized c-Src.
Bilodeau N, Fiset A, Poirier GG, Fortier S, Gingras MC, Lavoie JN, Faure RL
The FEBS journal 2006 Mar;273(5):992-1003
The FEBS journal 2006 Mar;273(5):992-1003
A Fischer rat substrain deficient in dipeptidyl peptidase IV activity makes normal steady-state RNA levels and an altered protein. Use as a liver-cell transplantation model.
Thompson NL, Hixson DC, Callanan H, Panzica M, Flanagan D, Faris RA, Hong WJ, Hartel-Schenk S, Doyle D
The Biochemical journal 1991 Feb 1;273 ( Pt 3)(Pt 3):497-502
The Biochemical journal 1991 Feb 1;273 ( Pt 3)(Pt 3):497-502
A Fischer rat substrain deficient in dipeptidyl peptidase IV activity makes normal steady-state RNA levels and an altered protein. Use as a liver-cell transplantation model.
Thompson NL, Hixson DC, Callanan H, Panzica M, Flanagan D, Faris RA, Hong WJ, Hartel-Schenk S, Doyle D
The Biochemical journal 1991 Feb 1;273 ( Pt 3):497-502
The Biochemical journal 1991 Feb 1;273 ( Pt 3):497-502
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
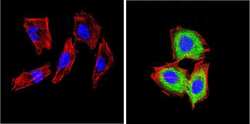
- Experimental details
- Immunofluorescent analysis of CD26 (green) showing positive staining in the cytoplasm of H-4-II-E cells (right) compared with a negative control in the absence of primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes, blocked with 3% BSA-PBS for 30 minutes at room temperature and probed with a CD26 monoclonal antibody (Product # MA2607) in 3% BSA-PBS at a dilution of 1:20 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight 488-conjugated goat-anti-mouse IgG (H+L) secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with DAPI for 5-10 minutes in the dark. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
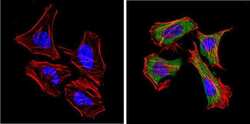
- Experimental details
- Immunofluorescent analysis of CD26 (green) showing positive staining in the cytoplasm of Hela cells (right) compared with a negative control in the absence of primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes, blocked with 3% BSA-PBS for 30 minutes at room temperature and probed with a CD26 monoclonal antibody (Product # MA2607) in 3% BSA-PBS at a dilution of 1:20 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight 488-conjugated goat-anti-mouse IgG (H+L) secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with DAPI for 5-10 minutes in the dark. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
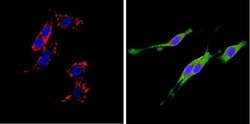
- Experimental details
- Immunofluorescent analysis of CD26 (green) showing positive staining in the cytoplasm of PC12 cells (right) compared with a negative control in the absence of primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes, blocked with 3% BSA-PBS for 30 minutes at room temperature and probed with a CD26 monoclonal antibody (Product # MA2607) in 3% BSA-PBS at a dilution of 1:20 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight 488-conjugated goat-anti-mouse IgG (H+L) secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with DAPI for 5-10 minutes in the dark. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
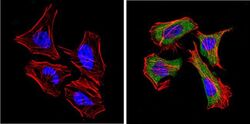
- Experimental details
- Immunofluorescent analysis of CD26 (green) showing positive staining in the cytoplasm of Hela cells (right) compared with a negative control in the absence of primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes, blocked with 3% BSA-PBS for 30 minutes at room temperature and probed with a CD26 monoclonal antibody (Product # MA2607) in 3% BSA-PBS at a dilution of 1:20 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight 488-conjugated goat-anti-mouse IgG (H+L) secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with DAPI for 5-10 minutes in the dark. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
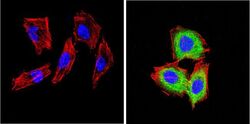
- Experimental details
- Immunofluorescent analysis of CD26 (green) showing positive staining in the cytoplasm of H-4-II-E cells (right) compared with a negative control in the absence of primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes, blocked with 3% BSA-PBS for 30 minutes at room temperature and probed with a CD26 monoclonal antibody (Product # MA2607) in 3% BSA-PBS at a dilution of 1:20 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight 488-conjugated goat-anti-mouse IgG (H+L) secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with DAPI for 5-10 minutes in the dark. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
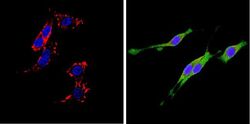
- Experimental details
- Immunofluorescent analysis of CD26 (green) showing positive staining in the cytoplasm of PC12 cells (right) compared with a negative control in the absence of primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes, blocked with 3% BSA-PBS for 30 minutes at room temperature and probed with a CD26 monoclonal antibody (Product # MA2607) in 3% BSA-PBS at a dilution of 1:20 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight 488-conjugated goat-anti-mouse IgG (H+L) secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with DAPI for 5-10 minutes in the dark. Images were taken at a magnification of 60x.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
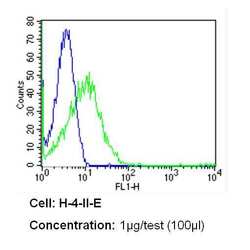
- Experimental details
- Flow cytometry analysis of CD26 in H-4-II-E cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a CD26 monoclonal antibody (Product # MA2607) at a dilution of 1 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
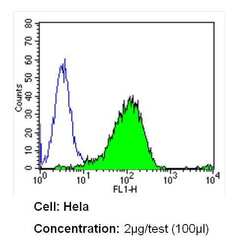
- Experimental details
- Flow cytometry analysis of CD26 in Hela cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a CD26 monoclonal antibody (Product # MA2607) at a dilution of 2 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
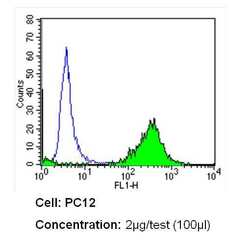
- Experimental details
- Flow cytometry analysis of CD26 in PC12 cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a CD26 monoclonal antibody (Product # MA2607) at a dilution of 2 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
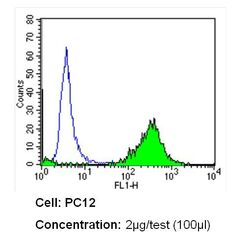
- Experimental details
- Flow cytometry analysis of CD26 in PC12 cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a CD26 monoclonal antibody (Product # MA2607) at a dilution of 2 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
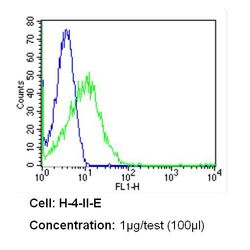
- Experimental details
- Flow cytometry analysis of CD26 in H-4-II-E cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a CD26 monoclonal antibody (Product # MA2607) at a dilution of 1 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated secondary antibody and re-suspended in PBS for FACS analysis.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
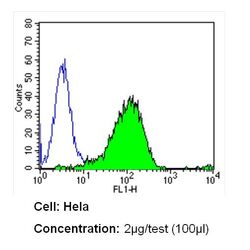
- Experimental details
- Flow cytometry analysis of CD26 in Hela cells (green) compared to an isotype control (blue). Cells were harvested, adjusted to a concentration of 1-5x10^6 cells/mL, fixed with 2% paraformaldehyde and washed with PBS. Cells were blocked with a 2% solution of BSA-PBS for 30 min at room temperature and incubated with a CD26 monoclonal antibody (Product # MA2607) at a dilution of 2 µg/test for 60 min at room temperature. Cells were then incubated for 40 min at room temperature in the dark using a Dylight 488-conjugated secondary antibody and re-suspended in PBS for FACS analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
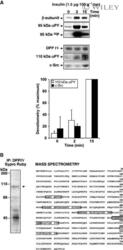
- Experimental details
- 2 Insulin-dependent tyrosine phosphorylation of DPP IV and its association with c-Src in the G/E fraction. (A) Rats were injected with insulin [1.5 ug*100 g -1 body weight (bw)]. The G/E fraction was isolated at the indicated times postinjection. (Upper panels) Proteins were separated by SDS/PAGE (80 ug, 7.5% resolving gel); the IR was detected by using either the anti-IR beta-subunit IgG or the anti-PY IgG (4G10). Autophosphorylation of the IR (95 kDa 32 P panel) was achieved by incubating aliquots (30 ug protein) with [gamma- 32 P]ATP. Following centrifugation, the pellet was solubilized and proteins immunoprecipitated using the anti-IR beta-subunit IgG. Immunoprecipitates were separated by SDS/PAGE and gels were subjected to alkali treatment and autoradiography. (Lower panels) DPP IV immunoprecipitation: Aliquots of G/E fraction (200 ug protein) were immunoprecipitated using an anti-DPP IV IgG (MA-2607). Immunoprecipitated proteins were separated by SDS/PAGE (10% resolving gel). Membranes were incubated with anti-DPP IV (26C), anti-PY (4G10) or anti-c-Src IgG (pieces of the same membrane). The signals were submitted to densitometric analysis, and the results were expressed as a percentage of the maximum signal. Each value represents the mean +- SD of three independent experiments. (B) Amino acid sequence of rat DPP IV (NCBI accession number NP_36921, Swiss-Prot P14740 ). The immunoprecipitated 110-kDa band, stained with SYPRO Ruby (left panel), was excised and subject
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
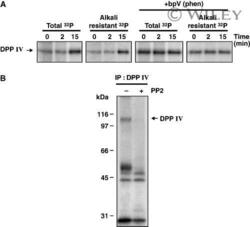
- Experimental details
- 3 In vitro phosphorylation of DPP IV. (A) Rats were injected with insulin (1.5 ug*100 g -1 body weight). The G/E fraction was isolated at the indicated times postinjection and aliquots (100 ug protein) were incubated with [gamma- 32 P]ATP in the presence or absence of 100 u m bpV(phen). They were solubilized and proteins immunoprecipitated using the anti-DPP IV IgG (MA-2607). The immunoprecipitates were separated by SDS/PAGE (10% resolving gel) and gels were subjected to autoradiography before and after alkali treatment. (B) The G/E fraction was isolated 15 min following insulin injection and was subjected to phosphorylation, in the presence or absence of the c-Src inhibitor PP2 (10 u m ), and then immunoprecipitated as above. Results are representative of three independent experiments.
 Explore
Explore Validate
Validate Learn
Learn Immunocytochemistry
Immunocytochemistry Immunoprecipitation
Immunoprecipitation