Antibody data
- Antibody Data
- Antigen structure
- References [7]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 12-5875-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD357 (AITR/GITR) Monoclonal Antibody (eBioAITR), PE, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The monoclonal antibody reacts with human AITR, Activation Inducible TNFR family member, an approximately 25 kDa member of the TNFR superfamily. AITR mRNA is detected in lymph node, peripheral blood leukocytes and weakly in spleen. AITR mRNA expression was upregulated within 24 hour on PMA/ionomycin or PHA stimulated PBMC. At the protein level, AITR is expressed by a small population of activated PBMC. AITR associates with TRAF1, TRAF2 and TRAF3 and induces nuclear factor NF-kappaB activation via TRAF2. Recently TL6 (AITRL) has been reported as the ligand for AITR. Interaction of AITR with AITRL is important for cross-talk between T lymphocytes and endothelial cells. Applications Reported: This eBioAITR antibody has been reported for use in flow cytometric analysis. Applications Tested: This eBioAITR antibody has been pre-titrated and tested by flow cytometric analysis of stimulated human peripheral blood mononuclear cells. This can be used at 5 µL (0.5 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488-561 nm; Emission: 578 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Yellow dye
- Isotype
- IgG
- Antibody clone number
- eBioAITR
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Optimal target saturation of ligand-blocking anti-GITR antibody IBI37G5 dictates FcγR-independent GITR agonism and antitumor activity.
Follicular Regulatory CD8 T Cells Impair the Germinal Center Response in SIV and Ex Vivo HIV Infection.
Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection.
Highly heterogeneous, activated, and short-lived regulatory T cells during chronic filarial infection.
Elevated levels of CD4(+)CD25(+)FoxP3(+) T cells in systemic sclerosis patients contribute to the secretion of IL-17 and immunosuppression dysfunction.
Transfer of regulatory properties from tolerogenic to proinflammatory dendritic cells via induced autoreactive regulatory T cells.
CD4(+) regulatory T cells in a cynomolgus macaque model of Mycobacterium tuberculosis infection.
Liu H, Wu W, Sun G, Chia T, Cao L, Liu X, Guan J, Fu F, Yao Y, Wu Z, Zhou S, Wang J, Lu J, Kuang Z, Wu M, He L, Shao Z, Wu D, Chen B, Xu W, Wang Z, He K
Cell reports. Medicine 2022 Jun 21;3(6):100660
Cell reports. Medicine 2022 Jun 21;3(6):100660
Follicular Regulatory CD8 T Cells Impair the Germinal Center Response in SIV and Ex Vivo HIV Infection.
Miles B, Miller SM, Folkvord JM, Levy DN, Rakasz EG, Skinner PJ, Connick E
PLoS pathogens 2016 Oct;12(10):e1005924
PLoS pathogens 2016 Oct;12(10):e1005924
Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection.
Miles B, Miller SM, Folkvord JM, Kimball A, Chamanian M, Meditz AL, Arends T, McCarter MD, Levy DN, Rakasz EG, Skinner PJ, Connick E
Nature communications 2015 Oct 20;6:8608
Nature communications 2015 Oct 20;6:8608
Highly heterogeneous, activated, and short-lived regulatory T cells during chronic filarial infection.
Metenou S, Coulibaly YI, Sturdevant D, Dolo H, Diallo AA, Soumaoro L, Coulibaly ME, Kanakabandi K, Porcella SF, Klion AD, Nutman TB
European journal of immunology 2014 Jul;44(7):2036-47
European journal of immunology 2014 Jul;44(7):2036-47
Elevated levels of CD4(+)CD25(+)FoxP3(+) T cells in systemic sclerosis patients contribute to the secretion of IL-17 and immunosuppression dysfunction.
Liu X, Gao N, Li M, Xu D, Hou Y, Wang Q, Zhang G, Sun Q, Zhang H, Zeng X
PloS one 2013;8(6):e64531
PloS one 2013;8(6):e64531
Transfer of regulatory properties from tolerogenic to proinflammatory dendritic cells via induced autoreactive regulatory T cells.
Kleijwegt FS, Laban S, Duinkerken G, Joosten AM, Koeleman BP, Nikolic T, Roep BO
Journal of immunology (Baltimore, Md. : 1950) 2011 Dec 15;187(12):6357-64
Journal of immunology (Baltimore, Md. : 1950) 2011 Dec 15;187(12):6357-64
CD4(+) regulatory T cells in a cynomolgus macaque model of Mycobacterium tuberculosis infection.
Green AM, Mattila JT, Bigbee CL, Bongers KS, Lin PL, Flynn JL
The Journal of infectious diseases 2010 Aug 15;202(4):533-41
The Journal of infectious diseases 2010 Aug 15;202(4):533-41
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of 3-day Anti-Human CD3 and Anti-Human CD28 Functional Grade Purified (Product # 16-0039-81 and Product # 16-0289-81)-stimulated normal human peripheral blood cells with Mouse IgG1 K Isotype Control PE (Product # 12-4714-81) (blue histogram) or Anti-Human CD357 (AITR/GITR) PE (purple histogram). Total viable cells, as determined by Fixable Viability Dye eFluor® 780 (Product # 65-0865-14), were used for analysis.
- Conjugate
- Yellow dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
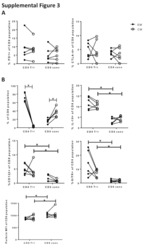
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
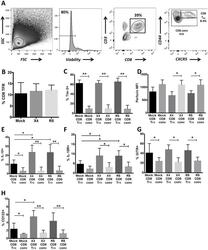
- Experimental details
- Fig 1 Human tonsil CD8 T FR are distinct from conventional CD8 T cells. Disaggregated tonsil cell cultures were mock-spinoculated or spinoculated with X4- or R5-tropic HIV and cultured for 2 days (n = 8). (A) Flow gating strategy to determine viable, CD8 T FR (CD3+CD8+CXCR5 hi CD44 hi ) and all other CD3+CD8+ cells (CD8 conv). (B) The percentage of CD8 T FR in mock- or HIV-spinoculated samples. The percent or MFI of CD8 T FR and CD8 conv expressing (C) Tim-3, (D) perforin, (E) IL-10, (F) IL-15 receptor, (G) GITR, and (H) CD122 (IL-2Rbeta). Graphs depict median and range. Statistical significance was determined by non-parametric one-way ANOVA (Friedman test) and is displayed as * = p
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
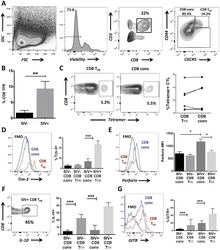
- Experimental details
- Fig 6 CD8 T FR are higher in SIV-infected than uninfected rhesus macaques. Disaggregated cells from lymph nodes and spleen of SIV-uninfected (n = 6) and SIV-infected (n = 6) rhesus macaques were analyzed for CD8 T FR by flow cytometry. (A) Flow gating strategy to determine viable CD8 T FR (CD3+CD8+CXCR5 hi CD44 hi ) and CD8 conv. (B) Percent CD8 T FR in SIV-uninfected compared to SIV-infected rhesus macaques. (C) Percent of SIV-Gag tetramer+ CD8 T FR compared to CD8 conv. CD8 T FR and CD8 conv from SIV-uninfected and SIV-infected rhesus macaques were analyzed for percent or MFI expression of (D) Tim-3, (E) perforin, (F) IL-10, and (G) GITR. Graphs depict median and range. Statistical significance was determined by non-parametric one-way ANOVA (Friedman test) and is displayed as * = p
- Conjugate
- Yellow dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry