Antibody data
- Antibody Data
- Antigen structure
- References [2]
- Comments [0]
- Validations
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-44961 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- USP17L2 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- Peptide sequence: FYIQKSEWER HSESVSRGRE PRALGAEDTD RRATQGELKR DHPCLQAPEL Sequence homology: Human: 100%
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 μL
- Concentration
- 0.5 mg/mL
- Storage
- -20°C, Avoid Freeze/Thaw Cycles
Submitted references Deubiquitinase DUB3 Regulates Cell Cycle Progression via Stabilizing Cyclin A for Proliferation of Non-Small Cell Lung Cancer Cells.
DUB3 Deubiquitylating Enzymes Regulate Hippo Pathway Activity by Regulating the Stability of ITCH, LATS and AMOT Proteins.
Hu B, Deng T, Ma H, Liu Y, Feng P, Wei D, Ling N, Li L, Qiu S, Zhang L, Peng B, Liu J, Ye M
Cells 2019 Mar 31;8(4)
Cells 2019 Mar 31;8(4)
DUB3 Deubiquitylating Enzymes Regulate Hippo Pathway Activity by Regulating the Stability of ITCH, LATS and AMOT Proteins.
Nguyen HT, Kugler JM, Cohen SM
PloS one 2017;12(1):e0169587
PloS one 2017;12(1):e0169587
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
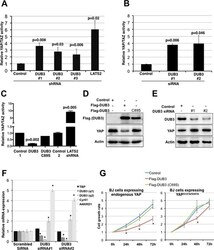
- Experimental details
- Fig 1 DUB3 regulates Hippo activity by mediating YAP turnover. (A) Luciferase reporter assays showing the effects of DUB3 shRNAs on YAP/TAZ activity. HEK293T cells were transfected to express the 8XGTIIC_luc YAP/TAZ reporter, which contains 8 TEAD binding sites to control the expression of firefly luciferase and a vector expressing CMV-Renilla luciferase to normalize for transfection efficiency, together with independent shRNA vectors to deplete DUB3 or with a control shRNA. shRNA to deplete LATS2 was used as a positive control. Data represent the average of three independent transfection experiments +- SD. P values were determined using Student's T-test (2-tailed, unequal variance). (B) Luciferase reporter assays showing the effects of DUB3 siRNAs on YAP/TAZ activity. HEK293T cells were transfected to express the luciferase reporters together with independent siRNAs to deplete DUB3 or with a control scrambled siRNA. Data represent the average of three independent transfection experiments +- SD. P values were determined using Student's T-test (2-tailed, unequal variance). (C) Luciferase reporter assays showing the effect of DUB3 on YAP/TAZ activity. HEK293T cells were transfected to express the luciferase reporters together with a vector expressing Flag-DUB3, the C89S mutant form of DUB3 or relevant controls vectors. shRNA to deplete LATS2 was used as a positive control. Data represent the average of three independent replicates +- SD. P values were determined using
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
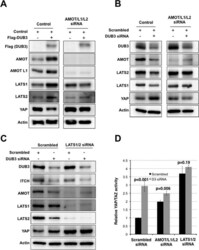
- Experimental details
- Fig 3 LATS and AMOT proteins are required for DUB3-mediated regulation of Hippo signaling. (A) Immunoblots showing the effect of DUB3 expression on LATS kinases, AMOT and YAP. HEK293T cells were transfected with a vector expressing Flag-DUB3 or a control vector together with a mixture of siRNAs targeting AMOT, AMOTL1 and AMOTL2 or a scrambled siRNA control. Blots were probed with antibodies against Flag, AMOT, AMOTL1, LATS1, LATS2 and YAP. Anti-Actin was used to control for loading. Samples were run on the same SDS-acrylamide gels with intervening lanes removed. (B) Immunoblots showing the effect of DUB3 depletion on LATS kinases, AMOT and YAP. HEK293T cells were transfected with a vector expressing Flag-DUB3 or a control vector together with a mixture of siRNAs targeting AMOT, AMOTL1 and AMOTL2 or a scrambled siRNA control. Blots were probed with antibodies against DUB3, AMOT, LATS1, LATS2 and YAP. Anti-Actin was used to control for loading. Samples were run on the same gels with intervening lanes removed. (C) Immunoblots showing the effect of DUB3 depletion on ITCH, LATS kinases and AMOT. HEK293T cells were transfected with a control or DUB3 siRNA in the presence of LATS1 and LATS2 siRNAs or a scrambled siRNA control. Blots were probed with antibodies against DUB3, ITCH, AMOT, LATS1, LATS2 and YAP. Anti-Actin was used to control for loading. (D) Luciferase reporter assays showing the effects of DUB3 siRNAs on YAP/TAZ activity. HEK293T cells were transfected to express the l
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Other assay
Other assay