Antibody data
- Antibody Data
- Antigen structure
- References [83]
- Comments [0]
- Validations
- Immunocytochemistry [3]
- Immunohistochemistry [1]
- Flow cytometry [2]
- Other assay [52]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 459200 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- SDHA Monoclonal Antibody (2E3GC12FB2AE2)
- Antibody type
- Monoclonal
- Antigen
- Purifed from natural sources
- Description
- ICC application requires heat-induced antigen retrieval where aldehydes are used as fixatives. Use 20min incubation at 90-100°C in 0.1 M Tris/HCl pH 9.5 with 5% urea (wt/vol). Positive control: Human heart mitochondria.
- Reactivity
- Human, Mouse, Rat, Bovine
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 2E3GC12FB2AE2
- Vial size
- 100 μg
- Concentration
- 1 mg/mL
- Storage
- 4°C
Submitted references Exercise training enhances muscle mitochondrial metabolism in diet-resistant obesity.
Sex-specific genetic regulation of adipose mitochondria and metabolic syndrome by Ndufv2.
Early Dysfunction of Substantia Nigra Dopamine Neurons in the ParkinQ311X Mouse.
Comparative analysis of CI- and CIV-containing respiratory supercomplexes at single-cell resolution.
The mitochondrial carrier SFXN1 is critical for complex III integrity and cellular metabolism.
Augmenter of liver regeneration regulates cellular iron homeostasis by modulating mitochondrial transport of ATP-binding cassette B8.
PPARδ Attenuates Alcohol-Mediated Insulin Resistance by Enhancing Fatty Acid-Induced Mitochondrial Uncoupling and Antioxidant Defense in Skeletal Muscle.
SDHB and SDHA Immunohistochemistry in Canine Pheochromocytomas.
A novel homozygous variant in MICOS13/QIL1 causes hepato-encephalopathy with mitochondrial DNA depletion syndrome.
Na(+) controls hypoxic signalling by the mitochondrial respiratory chain.
A Broad Response to Intracellular Long-Chain Polyphosphate in Human Cells.
Leigh Syndrome Due to NDUFV1 Mutations Initially Presenting as LBSL.
Autophagy inhibition prevents glucocorticoid-increased adiposity via suppressing BAT whitening.
CHCHD10-regulated OPA1-mitofilin complex mediates TDP-43-induced mitochondrial phenotypes associated with frontotemporal dementia.
GLP-1 Receptor Signaling in Astrocytes Regulates Fatty Acid Oxidation, Mitochondrial Integrity, and Function.
A novel approach to measure mitochondrial respiration in frozen biological samples.
Use of in-gel peroxidase assay for cytochrome c to visualize mitochondrial complexes III and IV.
Fine-tuning of the respiratory complexes stability and supercomplexes assembly in cells defective of complex III.
ERdj8 governs the size of autophagosomes during the formation process.
A Scaffold-Free 3-D Co-Culture Mimics the Major Features of the Reverse Warburg Effect In Vitro.
A novel compound heterozygous variant of ECHS1 identified in a Japanese patient with Leigh syndrome.
Chloramphenicol Mitigates Oxidative Stress by Inhibiting Translation of Mitochondrial Complex I in Dopaminergic Neurons of Toxin-Induced Parkinson's Disease Model.
Molecular Characterization of New FBXL4 Mutations in Patients With mtDNA Depletion Syndrome.
Brain pyrimidine nucleotide synthesis and Alzheimer disease.
Mitochondrial supercomplex assembly promotes breast and endometrial tumorigenesis by metabolic alterations and enhanced hypoxia tolerance.
Fascin Controls Metastatic Colonization and Mitochondrial Oxidative Phosphorylation by Remodeling Mitochondrial Actin Filaments.
Effects of prolonged type 2 diabetes on mitochondrial function in cerebral blood vessels.
CerS6-Derived Sphingolipids Interact with Mff and Promote Mitochondrial Fragmentation in Obesity.
Mutations in the mitochondrial complex I assembly factor NDUFAF6 cause isolated bilateral striatal necrosis and progressive dystonia in childhood.
CAPRI enables comparison of evolutionarily conserved RNA interacting regions.
Loss of the mitochondrial i-AAA protease YME1L leads to ocular dysfunction and spinal axonopathy.
Renal cold storage followed by transplantation impairs proteasome function and mitochondrial protein homeostasis.
IL-15 improves skeletal muscle oxidative metabolism and glucose uptake in association with increased respiratory chain supercomplex formation and AMPK pathway activation.
ROMO1 is a constituent of the human presequence translocase required for YME1L protease import.
The failing heart utilizes 3-hydroxybutyrate as a metabolic stress defense.
COX6A2 variants cause a muscle-specific cytochrome c oxidase deficiency.
Exposure of Monocytic Cells to Lipopolysaccharide Induces Coordinated Endotoxin Tolerance, Mitochondrial Biogenesis, Mitophagy, and Antioxidant Defenses.
Mild Impairment of Mitochondrial OXPHOS Promotes Fatty Acid Utilization in POMC Neurons and Improves Glucose Homeostasis in Obesity.
APP promotes osteoblast survival and bone formation by regulating mitochondrial function and preventing oxidative stress.
CLPP deficiency protects against metabolic syndrome but hinders adaptive thermogenesis.
PIMT/NCOA6IP Deletion in the Mouse Heart Causes Delayed Cardiomyopathy Attributable to Perturbation in Energy Metabolism.
Mitochondrial Alterations (Inhibition of Mitochondrial Protein Expression, Oxidative Metabolism, and Ultrastructure) Induced by Linezolid and Tedizolid at Clinically Relevant Concentrations in Cultured Human HL-60 Promyelocytes and THP-1 Monocytes.
Mutations in COQ8B (ADCK4) found in patients with steroid-resistant nephrotic syndrome alter COQ8B function.
Base-excision repair deficiency alone or combined with increased oxidative stress does not increase mtDNA point mutations in mice.
Therapeutic synergy between tigecycline and venetoclax in a preclinical model of MYC/BCL2 double-hit B cell lymphoma.
PARL partitions the lipid transfer protein STARD7 between the cytosol and mitochondria.
Impaired Mitochondrial Respiration in Large Cerebral Arteries of Rats with Type 2 Diabetes.
Dual loss of succinate dehydrogenase (SDH) and complex I activity is necessary to recapitulate the metabolic phenotype of SDH mutant tumors.
Biallelic C1QBP Mutations Cause Severe Neonatal-, Childhood-, or Later-Onset Cardiomyopathy Associated with Combined Respiratory-Chain Deficiencies.
Transcriptomic and proteomic landscape of mitochondrial dysfunction reveals secondary coenzyme Q deficiency in mammals.
CLUH regulates mitochondrial metabolism by controlling translation and decay of target mRNAs.
Age-Associated Loss of OPA1 in Muscle Impacts Muscle Mass, Metabolic Homeostasis, Systemic Inflammation, and Epithelial Senescence.
Burn-induced muscle metabolic derangements and mitochondrial dysfunction are associated with activation of HIF-1α and mTORC1: Role of protein farnesylation.
Post-translational modifications of Annexin A2 are linked to its association with perinuclear nonpolysomal mRNP complexes.
Sixty years old is the breakpoint of human frontal cortex aging.
Mitochondrial respiratory-chain adaptations in macrophages contribute to antibacterial host defense.
Mutant desmin substantially perturbs mitochondrial morphology, function and maintenance in skeletal muscle tissue.
Twinkle overexpression prevents cardiac rupture after myocardial infarction by alleviating impaired mitochondrial biogenesis.
Mitochondrial Protein Synthesis Adapts to Influx of Nuclear-Encoded Protein.
The mitochondrial translation machinery as a therapeutic target in Myc-driven lymphomas.
Control of mitochondrial function and cell growth by the atypical cadherin Fat1.
Amyloid β-peptides interfere with mitochondrial preprotein import competence by a coaggregation process.
Reduction in mitochondrial iron alleviates cardiac damage during injury.
Loss of CLPP alleviates mitochondrial cardiomyopathy without affecting the mammalian UPRmt.
Loss of OMA1 delays neurodegeneration by preventing stress-induced OPA1 processing in mitochondria.
Mitochondrial protein hyperacetylation in the failing heart.
A ketogenic diet accelerates neurodegeneration in mice with induced mitochondrial DNA toxicity in the forebrain.
Downstream effects of plectin mutations in epidermolysis bullosa simplex with muscular dystrophy.
The mitochondrial function of the cerebral vasculature in insulin-resistant Zucker obese rats.
AMP-activated protein kinase controls exercise training- and AICAR-induced increases in SIRT3 and MnSOD.
Overexpression of TFAM or twinkle increases mtDNA copy number and facilitates cardioprotection associated with limited mitochondrial oxidative stress.
Structure and function of the N-terminal domain of the human mitochondrial calcium uniporter.
Dynamics of enhanced mitochondrial respiration in female compared with male rat cerebral arteries.
Cytochrome c oxidase deficiency accelerates mitochondrial apoptosis by activating ceramide synthase 6.
A keratin scaffold regulates epidermal barrier formation, mitochondrial lipid composition, and activity.
Imbalanced OPA1 processing and mitochondrial fragmentation cause heart failure in mice.
CNC-bZIP protein Nrf1-dependent regulation of glucose-stimulated insulin secretion.
Mitochondrial dynamics associated with oxygen-glucose deprivation in rat primary neuronal cultures.
Identification of mitochondrial dysfunction in Hutchinson-Gilford progeria syndrome through use of stable isotope labeling with amino acids in cell culture.
Irgm1 (LRG-47), a regulator of cell-autonomous immunity, does not localize to mycobacterial or listerial phagosomes in IFN-γ-induced mouse cells.
IKKα and alternative NF-κB regulate PGC-1β to promote oxidative muscle metabolism.
Identification of human fumarylacetoacetate hydrolase domain-containing protein 1 (FAHD1) as a novel mitochondrial acylpyruvase.
Mitochondrial DNA toxicity compromises mitochondrial dynamics and induces hippocampal antioxidant defenses.
Pileggi CA, Blondin DP, Hooks BG, Parmar G, Alecu I, Patten DA, Cuillerier A, O'Dwyer C, Thrush AB, Fullerton MD, Bennett SA, Doucet É, Haman F, Cuperlovic-Culf M, McPherson R, Dent RRM, Harper ME
EBioMedicine 2022 Sep;83:104192
EBioMedicine 2022 Sep;83:104192
Sex-specific genetic regulation of adipose mitochondria and metabolic syndrome by Ndufv2.
Chella Krishnan K, Vergnes L, Acín-Pérez R, Stiles L, Shum M, Ma L, Mouisel E, Pan C, Moore TM, Péterfy M, Romanoski CE, Reue K, Björkegren JLM, Laakso M, Liesa M, Lusis AJ
Nature metabolism 2021 Nov;3(11):1552-1568
Nature metabolism 2021 Nov;3(11):1552-1568
Early Dysfunction of Substantia Nigra Dopamine Neurons in the ParkinQ311X Mouse.
Regoni M, Zanetti L, Comai S, Mercatelli D, Novello S, Albanese F, Croci L, Consalez GG, Ciammola A, Valtorta F, Morari M, Sassone J
Biomedicines 2021 May 5;9(5)
Biomedicines 2021 May 5;9(5)
Comparative analysis of CI- and CIV-containing respiratory supercomplexes at single-cell resolution.
Bertan F, Wischhof L, Scifo E, Guranda M, Jackson J, Marsal-Cots A, Piazzesi A, Stork M, Peitz M, Prehn JHM, Ehninger D, Nicotera P, Bano D
Cell reports methods 2021 May 24;1(1):100002
Cell reports methods 2021 May 24;1(1):100002
The mitochondrial carrier SFXN1 is critical for complex III integrity and cellular metabolism.
Acoba MG, Alpergin ESS, Renuse S, Fernández-Del-Río L, Lu YW, Khalimonchuk O, Clarke CF, Pandey A, Wolfgang MJ, Claypool SM
Cell reports 2021 Mar 16;34(11):108869
Cell reports 2021 Mar 16;34(11):108869
Augmenter of liver regeneration regulates cellular iron homeostasis by modulating mitochondrial transport of ATP-binding cassette B8.
Chang HC, Shapiro JS, Jiang X, Senyei G, Sato T, Geier J, Sawicki KT, Ardehali H
eLife 2021 Apr 9;10
eLife 2021 Apr 9;10
PPARδ Attenuates Alcohol-Mediated Insulin Resistance by Enhancing Fatty Acid-Induced Mitochondrial Uncoupling and Antioxidant Defense in Skeletal Muscle.
Koh JH, Kim KH, Park SY, Kim YW, Kim JY
Frontiers in physiology 2020;11:749
Frontiers in physiology 2020;11:749
SDHB and SDHA Immunohistochemistry in Canine Pheochromocytomas.
Abed FM, Brown MA, Al-Mahmood OA, Dark MJ
Animals : an open access journal from MDPI 2020 Sep 17;10(9)
Animals : an open access journal from MDPI 2020 Sep 17;10(9)
A novel homozygous variant in MICOS13/QIL1 causes hepato-encephalopathy with mitochondrial DNA depletion syndrome.
Kishita Y, Shimura M, Kohda M, Akita M, Imai-Okazaki A, Yatsuka Y, Nakajima Y, Ito T, Ohtake A, Murayama K, Okazaki Y
Molecular genetics & genomic medicine 2020 Oct;8(10):e1427
Molecular genetics & genomic medicine 2020 Oct;8(10):e1427
Na(+) controls hypoxic signalling by the mitochondrial respiratory chain.
Hernansanz-Agustín P, Choya-Foces C, Carregal-Romero S, Ramos E, Oliva T, Villa-Piña T, Moreno L, Izquierdo-Álvarez A, Cabrera-García JD, Cortés A, Lechuga-Vieco AV, Jadiya P, Navarro E, Parada E, Palomino-Antolín A, Tello D, Acín-Pérez R, Rodríguez-Aguilera JC, Navas P, Cogolludo Á, López-Montero I, Martínez-Del-Pozo Á, Egea J, López MG, Elrod JW, Ruíz-Cabello J, Bogdanova A, Enríquez JA, Martínez-Ruiz A
Nature 2020 Oct;586(7828):287-291
Nature 2020 Oct;586(7828):287-291
A Broad Response to Intracellular Long-Chain Polyphosphate in Human Cells.
Bondy-Chorney E, Abramchuk I, Nasser R, Holinier C, Denoncourt A, Baijal K, McCarthy L, Khacho M, Lavallée-Adam M, Downey M
Cell reports 2020 Oct 27;33(4):108318
Cell reports 2020 Oct 27;33(4):108318
Leigh Syndrome Due to NDUFV1 Mutations Initially Presenting as LBSL.
Borna NN, Kishita Y, Sakai N, Hamada Y, Kamagata K, Kohda M, Ohtake A, Murayama K, Okazaki Y
Genes 2020 Nov 9;11(11)
Genes 2020 Nov 9;11(11)
Autophagy inhibition prevents glucocorticoid-increased adiposity via suppressing BAT whitening.
Deng J, Guo Y, Yuan F, Chen S, Yin H, Jiang X, Jiao F, Wang F, Ji H, Hu G, Ying H, Chen Y, Zhai Q, Xiao F, Guo F
Autophagy 2020 Mar;16(3):451-465
Autophagy 2020 Mar;16(3):451-465
CHCHD10-regulated OPA1-mitofilin complex mediates TDP-43-induced mitochondrial phenotypes associated with frontotemporal dementia.
Liu T, Woo JA, Bukhari MZ, LePochat P, Chacko A, Selenica MB, Yan Y, Kotsiviras P, Buosi SC, Zhao X, Kang DE
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2020 Jun;34(6):8493-8509
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2020 Jun;34(6):8493-8509
GLP-1 Receptor Signaling in Astrocytes Regulates Fatty Acid Oxidation, Mitochondrial Integrity, and Function.
Timper K, Del Río-Martín A, Cremer AL, Bremser S, Alber J, Giavalisco P, Varela L, Heilinger C, Nolte H, Trifunovic A, Horvath TL, Kloppenburg P, Backes H, Brüning JC
Cell metabolism 2020 Jun 2;31(6):1189-1205.e13
Cell metabolism 2020 Jun 2;31(6):1189-1205.e13
A novel approach to measure mitochondrial respiration in frozen biological samples.
Acin-Perez R, Benador IY, Petcherski A, Veliova M, Benavides GA, Lagarrigue S, Caudal A, Vergnes L, Murphy AN, Karamanlidis G, Tian R, Reue K, Wanagat J, Sacks H, Amati F, Darley-Usmar VM, Liesa M, Divakaruni AS, Stiles L, Shirihai OS
The EMBO journal 2020 Jul 1;39(13):e104073
The EMBO journal 2020 Jul 1;39(13):e104073
Use of in-gel peroxidase assay for cytochrome c to visualize mitochondrial complexes III and IV.
Hara T, Shibata Y, Amagai R, Okado-Matsumoto A
Biology open 2020 Jan 8;9(1)
Biology open 2020 Jan 8;9(1)
Fine-tuning of the respiratory complexes stability and supercomplexes assembly in cells defective of complex III.
Tropeano CV, Aleo SJ, Zanna C, Roberti M, Scandiffio L, Loguercio Polosa P, Fiori J, Porru E, Roda A, Carelli V, Steimle S, Daldal F, Rugolo M, Ghelli A
Biochimica et biophysica acta. Bioenergetics 2020 Feb 1;1861(2):148133
Biochimica et biophysica acta. Bioenergetics 2020 Feb 1;1861(2):148133
ERdj8 governs the size of autophagosomes during the formation process.
Yamamoto YH, Kasai A, Omori H, Takino T, Sugihara M, Umemoto T, Hamasaki M, Hatta T, Natsume T, Morimoto RI, Arai R, Waguri S, Sato M, Sato K, Bar-Nun S, Yoshimori T, Noda T, Nagata K
The Journal of cell biology 2020 Aug 3;219(8)
The Journal of cell biology 2020 Aug 3;219(8)
A Scaffold-Free 3-D Co-Culture Mimics the Major Features of the Reverse Warburg Effect In Vitro.
Keller F, Bruch R, Schneider R, Meier-Hubberten J, Hafner M, Rudolf R
Cells 2020 Aug 13;9(8)
Cells 2020 Aug 13;9(8)
A novel compound heterozygous variant of ECHS1 identified in a Japanese patient with Leigh syndrome.
Uchino S, Iida A, Sato A, Ishikawa K, Mimaki M, Nishino I, Goto YI
Human genome variation 2019;6:19
Human genome variation 2019;6:19
Chloramphenicol Mitigates Oxidative Stress by Inhibiting Translation of Mitochondrial Complex I in Dopaminergic Neurons of Toxin-Induced Parkinson's Disease Model.
Han J, Kim SJ, Ryu MJ, Jang Y, Lee MJ, Ju X, Lee YL, Cui J, Shong M, Heo JY, Kweon GR
Oxidative medicine and cellular longevity 2019;2019:4174803
Oxidative medicine and cellular longevity 2019;2019:4174803
Molecular Characterization of New FBXL4 Mutations in Patients With mtDNA Depletion Syndrome.
Emperador S, Garrido-Pérez N, Amezcua-Gil J, Gaudó P, Andrés-Sanz JA, Yubero D, Fernández-Marmiesse A, O'Callaghan MM, Ortigoza-Escobar JD, Iriondo M, Ruiz-Pesini E, García-Cazorla A, Gil-Campos M, Artuch R, Montoya J, Bayona-Bafaluy MP
Frontiers in genetics 2019;10:1300
Frontiers in genetics 2019;10:1300
Brain pyrimidine nucleotide synthesis and Alzheimer disease.
Pesini A, Iglesias E, Bayona-Bafaluy MP, Garrido-Pérez N, Meade P, Gaudó P, Jiménez-Salvador I, Andrés-Benito P, Montoya J, Ferrer I, Pesini P, Ruiz-Pesini E
Aging 2019 Sep 27;11(19):8433-8462
Aging 2019 Sep 27;11(19):8433-8462
Mitochondrial supercomplex assembly promotes breast and endometrial tumorigenesis by metabolic alterations and enhanced hypoxia tolerance.
Ikeda K, Horie-Inoue K, Suzuki T, Hobo R, Nakasato N, Takeda S, Inoue S
Nature communications 2019 Sep 11;10(1):4108
Nature communications 2019 Sep 11;10(1):4108
Fascin Controls Metastatic Colonization and Mitochondrial Oxidative Phosphorylation by Remodeling Mitochondrial Actin Filaments.
Lin S, Huang C, Gunda V, Sun J, Chellappan SP, Li Z, Izumi V, Fang B, Koomen J, Singh PK, Hao J, Yang S
Cell reports 2019 Sep 10;28(11):2824-2836.e8
Cell reports 2019 Sep 10;28(11):2824-2836.e8
Effects of prolonged type 2 diabetes on mitochondrial function in cerebral blood vessels.
Merdzo I, Rutkai I, Sure VNLR, Katakam PVG, Busija DW
American journal of physiology. Heart and circulatory physiology 2019 Nov 1;317(5):H1086-H1092
American journal of physiology. Heart and circulatory physiology 2019 Nov 1;317(5):H1086-H1092
CerS6-Derived Sphingolipids Interact with Mff and Promote Mitochondrial Fragmentation in Obesity.
Hammerschmidt P, Ostkotte D, Nolte H, Gerl MJ, Jais A, Brunner HL, Sprenger HG, Awazawa M, Nicholls HT, Turpin-Nolan SM, Langer T, Krüger M, Brügger B, Brüning JC
Cell 2019 May 30;177(6):1536-1552.e23
Cell 2019 May 30;177(6):1536-1552.e23
Mutations in the mitochondrial complex I assembly factor NDUFAF6 cause isolated bilateral striatal necrosis and progressive dystonia in childhood.
Baide-Mairena H, Gaudó P, Marti-Sánchez L, Emperador S, Sánchez-Montanez A, Alonso-Luengo O, Correa M, Grau AM, Ortigoza-Escobar JD, Artuch R, Vázquez E, Del Toro M, Garrido-Pérez N, Ruiz-Pesini E, Montoya J, Bayona-Bafaluy MP, Pérez-Dueñas B
Molecular genetics and metabolism 2019 Mar;126(3):250-258
Molecular genetics and metabolism 2019 Mar;126(3):250-258
CAPRI enables comparison of evolutionarily conserved RNA interacting regions.
Panhale A, Richter FM, Ramírez F, Shvedunova M, Manke T, Mittler G, Akhtar A
Nature communications 2019 Jun 18;10(1):2682
Nature communications 2019 Jun 18;10(1):2682
Loss of the mitochondrial i-AAA protease YME1L leads to ocular dysfunction and spinal axonopathy.
Sprenger HG, Wani G, Hesseling A, König T, Patron M, MacVicar T, Ahola S, Wai T, Barth E, Rugarli EI, Bergami M, Langer T
EMBO molecular medicine 2019 Jan;11(1)
EMBO molecular medicine 2019 Jan;11(1)
Renal cold storage followed by transplantation impairs proteasome function and mitochondrial protein homeostasis.
Lo S, MacMillan-Crow LA, Parajuli N
American journal of physiology. Renal physiology 2019 Jan 1;316(1):F42-F53
American journal of physiology. Renal physiology 2019 Jan 1;316(1):F42-F53
IL-15 improves skeletal muscle oxidative metabolism and glucose uptake in association with increased respiratory chain supercomplex formation and AMPK pathway activation.
Nadeau L, Patten DA, Caron A, Garneau L, Pinault-Masson E, Foretz M, Haddad P, Anderson BG, Quinn LS, Jardine K, McBurney MW, Pistilli EE, Harper ME, Aguer C
Biochimica et biophysica acta. General subjects 2019 Feb;1863(2):395-407
Biochimica et biophysica acta. General subjects 2019 Feb;1863(2):395-407
ROMO1 is a constituent of the human presequence translocase required for YME1L protease import.
Richter F, Dennerlein S, Nikolov M, Jans DC, Naumenko N, Aich A, MacVicar T, Linden A, Jakobs S, Urlaub H, Langer T, Rehling P
The Journal of cell biology 2019 Feb 4;218(2):598-614
The Journal of cell biology 2019 Feb 4;218(2):598-614
The failing heart utilizes 3-hydroxybutyrate as a metabolic stress defense.
Horton JL, Davidson MT, Kurishima C, Vega RB, Powers JC, Matsuura TR, Petucci C, Lewandowski ED, Crawford PA, Muoio DM, Recchia FA, Kelly DP
JCI insight 2019 Feb 21;4(4)
JCI insight 2019 Feb 21;4(4)
COX6A2 variants cause a muscle-specific cytochrome c oxidase deficiency.
Inoue M, Uchino S, Iida A, Noguchi S, Hayashi S, Takahashi T, Fujii K, Komaki H, Takeshita E, Nonaka I, Okada Y, Yoshizawa T, Van Lommel L, Schuit F, Goto YI, Mimaki M, Nishino I
Annals of neurology 2019 Aug;86(2):193-202
Annals of neurology 2019 Aug;86(2):193-202
Exposure of Monocytic Cells to Lipopolysaccharide Induces Coordinated Endotoxin Tolerance, Mitochondrial Biogenesis, Mitophagy, and Antioxidant Defenses.
Widdrington JD, Gomez-Duran A, Pyle A, Ruchaud-Sparagano MH, Scott J, Baudouin SV, Rostron AJ, Lovat PE, Chinnery PF, Simpson AJ
Frontiers in immunology 2018;9:2217
Frontiers in immunology 2018;9:2217
Mild Impairment of Mitochondrial OXPHOS Promotes Fatty Acid Utilization in POMC Neurons and Improves Glucose Homeostasis in Obesity.
Timper K, Paeger L, Sánchez-Lasheras C, Varela L, Jais A, Nolte H, Vogt MC, Hausen AC, Heilinger C, Evers N, Pospisilik JA, Penninger JM, Taylor EB, Horvath TL, Kloppenburg P, Brüning JC
Cell reports 2018 Oct 9;25(2):383-397.e10
Cell reports 2018 Oct 9;25(2):383-397.e10
APP promotes osteoblast survival and bone formation by regulating mitochondrial function and preventing oxidative stress.
Pan JX, Tang F, Xiong F, Xiong L, Zeng P, Wang B, Zhao K, Guo H, Shun C, Xia WF, Mei L, Xiong WC
Cell death & disease 2018 Oct 22;9(11):1077
Cell death & disease 2018 Oct 22;9(11):1077
CLPP deficiency protects against metabolic syndrome but hinders adaptive thermogenesis.
Becker C, Kukat A, Szczepanowska K, Hermans S, Senft K, Brandscheid CP, Maiti P, Trifunovic A
EMBO reports 2018 May;19(5)
EMBO reports 2018 May;19(5)
PIMT/NCOA6IP Deletion in the Mouse Heart Causes Delayed Cardiomyopathy Attributable to Perturbation in Energy Metabolism.
Jia Y, Liu N, Viswakarma N, Sun R, Schipma MJ, Shang M, Thorp EB, Kanwar YS, Thimmapaya B, Reddy JK
International journal of molecular sciences 2018 May 16;19(5)
International journal of molecular sciences 2018 May 16;19(5)
Mitochondrial Alterations (Inhibition of Mitochondrial Protein Expression, Oxidative Metabolism, and Ultrastructure) Induced by Linezolid and Tedizolid at Clinically Relevant Concentrations in Cultured Human HL-60 Promyelocytes and THP-1 Monocytes.
Milosevic TV, Payen VL, Sonveaux P, Muccioli GG, Tulkens PM, Van Bambeke F
Antimicrobial agents and chemotherapy 2018 Mar;62(3)
Antimicrobial agents and chemotherapy 2018 Mar;62(3)
Mutations in COQ8B (ADCK4) found in patients with steroid-resistant nephrotic syndrome alter COQ8B function.
Vazquez Fonseca L, Doimo M, Calderan C, Desbats MA, Acosta MJ, Cerqua C, Cassina M, Ashraf S, Hildebrandt F, Sartori G, Navas P, Trevisson E, Salviati L
Human mutation 2018 Mar;39(3):406-414
Human mutation 2018 Mar;39(3):406-414
Base-excision repair deficiency alone or combined with increased oxidative stress does not increase mtDNA point mutations in mice.
Kauppila JHK, Bonekamp NA, Mourier A, Isokallio MA, Just A, Kauppila TES, Stewart JB, Larsson NG
Nucleic acids research 2018 Jul 27;46(13):6642-6669
Nucleic acids research 2018 Jul 27;46(13):6642-6669
Therapeutic synergy between tigecycline and venetoclax in a preclinical model of MYC/BCL2 double-hit B cell lymphoma.
Ravà M, D'Andrea A, Nicoli P, Gritti I, Donati G, Doni M, Giorgio M, Olivero D, Amati B
Science translational medicine 2018 Jan 31;10(426)
Science translational medicine 2018 Jan 31;10(426)
PARL partitions the lipid transfer protein STARD7 between the cytosol and mitochondria.
Saita S, Tatsuta T, Lampe PA, König T, Ohba Y, Langer T
The EMBO journal 2018 Feb 15;37(4)
The EMBO journal 2018 Feb 15;37(4)
Impaired Mitochondrial Respiration in Large Cerebral Arteries of Rats with Type 2 Diabetes.
Merdzo I, Rutkai I, Sure VN, McNulty CA, Katakam PV, Busija DW
Journal of vascular research 2017;54(1):1-12
Journal of vascular research 2017;54(1):1-12
Dual loss of succinate dehydrogenase (SDH) and complex I activity is necessary to recapitulate the metabolic phenotype of SDH mutant tumors.
Lorendeau D, Rinaldi G, Boon R, Spincemaille P, Metzger K, Jäger C, Christen S, Dong X, Kuenen S, Voordeckers K, Verstreken P, Cassiman D, Vermeersch P, Verfaillie C, Hiller K, Fendt SM
Metabolic engineering 2017 Sep;43(Pt B):187-197
Metabolic engineering 2017 Sep;43(Pt B):187-197
Biallelic C1QBP Mutations Cause Severe Neonatal-, Childhood-, or Later-Onset Cardiomyopathy Associated with Combined Respiratory-Chain Deficiencies.
Feichtinger RG, Oláhová M, Kishita Y, Garone C, Kremer LS, Yagi M, Uchiumi T, Jourdain AA, Thompson K, D'Souza AR, Kopajtich R, Alston CL, Koch J, Sperl W, Mastantuono E, Strom TM, Wortmann SB, Meitinger T, Pierre G, Chinnery PF, Chrzanowska-Lightowlers ZM, Lightowlers RN, DiMauro S, Calvo SE, Mootha VK, Moggio M, Sciacco M, Comi GP, Ronchi D, Murayama K, Ohtake A, Rebelo-Guiomar P, Kohda M, Kang D, Mayr JA, Taylor RW, Okazaki Y, Minczuk M, Prokisch H
American journal of human genetics 2017 Oct 5;101(4):525-538
American journal of human genetics 2017 Oct 5;101(4):525-538
Transcriptomic and proteomic landscape of mitochondrial dysfunction reveals secondary coenzyme Q deficiency in mammals.
Kühl I, Miranda M, Atanassov I, Kuznetsova I, Hinze Y, Mourier A, Filipovska A, Larsson NG
eLife 2017 Nov 14;6
eLife 2017 Nov 14;6
CLUH regulates mitochondrial metabolism by controlling translation and decay of target mRNAs.
Schatton D, Pla-Martin D, Marx MC, Hansen H, Mourier A, Nemazanyy I, Pessia A, Zentis P, Corona T, Kondylis V, Barth E, Schauss AC, Velagapudi V, Rugarli EI
The Journal of cell biology 2017 Mar 6;216(3):675-693
The Journal of cell biology 2017 Mar 6;216(3):675-693
Age-Associated Loss of OPA1 in Muscle Impacts Muscle Mass, Metabolic Homeostasis, Systemic Inflammation, and Epithelial Senescence.
Tezze C, Romanello V, Desbats MA, Fadini GP, Albiero M, Favaro G, Ciciliot S, Soriano ME, Morbidoni V, Cerqua C, Loefler S, Kern H, Franceschi C, Salvioli S, Conte M, Blaauw B, Zampieri S, Salviati L, Scorrano L, Sandri M
Cell metabolism 2017 Jun 6;25(6):1374-1389.e6
Cell metabolism 2017 Jun 6;25(6):1374-1389.e6
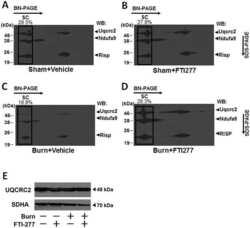
Burn-induced muscle metabolic derangements and mitochondrial dysfunction are associated with activation of HIF-1α and mTORC1: Role of protein farnesylation.
Nakazawa H, Ikeda K, Shinozaki S, Kobayashi M, Ikegami Y, Fu M, Nakamura T, Yasuhara S, Yu YM, Martyn JAJ, Tompkins RG, Shimokado K, Yorozu T, Ito H, Inoue S, Kaneki M
Scientific reports 2017 Jul 26;7(1):6618
Scientific reports 2017 Jul 26;7(1):6618
Post-translational modifications of Annexin A2 are linked to its association with perinuclear nonpolysomal mRNP complexes.
Aukrust I, Rosenberg LA, Ankerud MM, Bertelsen V, Hollås H, Saraste J, Grindheim AK, Vedeler A
FEBS open bio 2017 Feb;7(2):160-173
FEBS open bio 2017 Feb;7(2):160-173
Sixty years old is the breakpoint of human frontal cortex aging.
Cabré R, Naudí A, Dominguez-Gonzalez M, Ayala V, Jové M, Mota-Martorell N, Piñol-Ripoll G, Gil-Villar MP, Rué M, Portero-Otín M, Ferrer I, Pamplona R
Free radical biology & medicine 2017 Feb;103:14-22
Free radical biology & medicine 2017 Feb;103:14-22
Mitochondrial respiratory-chain adaptations in macrophages contribute to antibacterial host defense.
Garaude J, Acín-Pérez R, Martínez-Cano S, Enamorado M, Ugolini M, Nistal-Villán E, Hervás-Stubbs S, Pelegrín P, Sander LE, Enríquez JA, Sancho D
Nature immunology 2016 Sep;17(9):1037-1045
Nature immunology 2016 Sep;17(9):1037-1045
Mutant desmin substantially perturbs mitochondrial morphology, function and maintenance in skeletal muscle tissue.
Winter L, Wittig I, Peeva V, Eggers B, Heidler J, Chevessier F, Kley RA, Barkovits K, Strecker V, Berwanger C, Herrmann H, Marcus K, Kornblum C, Kunz WS, Schröder R, Clemen CS
Acta neuropathologica 2016 Sep;132(3):453-73
Acta neuropathologica 2016 Sep;132(3):453-73
Twinkle overexpression prevents cardiac rupture after myocardial infarction by alleviating impaired mitochondrial biogenesis.
Inoue T, Ikeda M, Ide T, Fujino T, Matsuo Y, Arai S, Saku K, Sunagawa K
American journal of physiology. Heart and circulatory physiology 2016 Sep 1;311(3):H509-19
American journal of physiology. Heart and circulatory physiology 2016 Sep 1;311(3):H509-19
Mitochondrial Protein Synthesis Adapts to Influx of Nuclear-Encoded Protein.
Richter-Dennerlein R, Oeljeklaus S, Lorenzi I, Ronsör C, Bareth B, Schendzielorz AB, Wang C, Warscheid B, Rehling P, Dennerlein S
Cell 2016 Oct 6;167(2):471-483.e10
Cell 2016 Oct 6;167(2):471-483.e10
The mitochondrial translation machinery as a therapeutic target in Myc-driven lymphomas.
D'Andrea A, Gritti I, Nicoli P, Giorgio M, Doni M, Conti A, Bianchi V, Casoli L, Sabò A, Mironov A, Beznoussenko GV, Amati B
Oncotarget 2016 Nov 8;7(45):72415-72430
Oncotarget 2016 Nov 8;7(45):72415-72430
Control of mitochondrial function and cell growth by the atypical cadherin Fat1.
Cao LL, Riascos-Bernal DF, Chinnasamy P, Dunaway CM, Hou R, Pujato MA, O'Rourke BP, Miskolci V, Guo L, Hodgson L, Fiser A, Sibinga NE
Nature 2016 Nov 24;539(7630):575-578
Nature 2016 Nov 24;539(7630):575-578
Amyloid β-peptides interfere with mitochondrial preprotein import competence by a coaggregation process.
Cenini G, Rüb C, Bruderek M, Voos W
Molecular biology of the cell 2016 Nov 1;27(21):3257-3272
Molecular biology of the cell 2016 Nov 1;27(21):3257-3272
Reduction in mitochondrial iron alleviates cardiac damage during injury.
Chang HC, Wu R, Shang M, Sato T, Chen C, Shapiro JS, Liu T, Thakur A, Sawicki KT, Prasad SV, Ardehali H
EMBO molecular medicine 2016 Mar 1;8(3):247-67
EMBO molecular medicine 2016 Mar 1;8(3):247-67
Loss of CLPP alleviates mitochondrial cardiomyopathy without affecting the mammalian UPRmt.
Seiferling D, Szczepanowska K, Becker C, Senft K, Hermans S, Maiti P, König T, Kukat A, Trifunovic A
EMBO reports 2016 Jul;17(7):953-64
EMBO reports 2016 Jul;17(7):953-64
Loss of OMA1 delays neurodegeneration by preventing stress-induced OPA1 processing in mitochondria.
Korwitz A, Merkwirth C, Richter-Dennerlein R, Tröder SE, Sprenger HG, Quirós PM, López-Otín C, Rugarli EI, Langer T
The Journal of cell biology 2016 Jan 18;212(2):157-66
The Journal of cell biology 2016 Jan 18;212(2):157-66
Mitochondrial protein hyperacetylation in the failing heart.
Horton JL, Martin OJ, Lai L, Riley NM, Richards AL, Vega RB, Leone TC, Pagliarini DJ, Muoio DM, Bedi KC Jr, Margulies KB, Coon JJ, Kelly DP
JCI insight 2016 Feb;2(1)
JCI insight 2016 Feb;2(1)
A ketogenic diet accelerates neurodegeneration in mice with induced mitochondrial DNA toxicity in the forebrain.
Lauritzen KH, Hasan-Olive MM, Regnell CE, Kleppa L, Scheibye-Knudsen M, Gjedde A, Klungland A, Bohr VA, Storm-Mathisen J, Bergersen LH
Neurobiology of aging 2016 Dec;48:34-47
Neurobiology of aging 2016 Dec;48:34-47
Downstream effects of plectin mutations in epidermolysis bullosa simplex with muscular dystrophy.
Winter L, Türk M, Harter PN, Mittelbronn M, Kornblum C, Norwood F, Jungbluth H, Thiel CT, Schlötzer-Schrehardt U, Schröder R
Acta neuropathologica communications 2016 Apr 27;4(1):44
Acta neuropathologica communications 2016 Apr 27;4(1):44
The mitochondrial function of the cerebral vasculature in insulin-resistant Zucker obese rats.
Merdzo I, Rutkai I, Tokes T, Sure VN, Katakam PV, Busija DW
American journal of physiology. Heart and circulatory physiology 2016 Apr 1;310(7):H830-8
American journal of physiology. Heart and circulatory physiology 2016 Apr 1;310(7):H830-8
AMP-activated protein kinase controls exercise training- and AICAR-induced increases in SIRT3 and MnSOD.
Brandauer J, Andersen MA, Kellezi H, Risis S, Frøsig C, Vienberg SG, Treebak JT
Frontiers in physiology 2015;6:85
Frontiers in physiology 2015;6:85
Overexpression of TFAM or twinkle increases mtDNA copy number and facilitates cardioprotection associated with limited mitochondrial oxidative stress.
Ikeda M, Ide T, Fujino T, Arai S, Saku K, Kakino T, Tyynismaa H, Yamasaki T, Yamada K, Kang D, Suomalainen A, Sunagawa K
PloS one 2015;10(3):e0119687
PloS one 2015;10(3):e0119687
Structure and function of the N-terminal domain of the human mitochondrial calcium uniporter.
Lee Y, Min CK, Kim TG, Song HK, Lim Y, Kim D, Shin K, Kang M, Kang JY, Youn HS, Lee JG, An JY, Park KR, Lim JJ, Kim JH, Kim JH, Park ZY, Kim YS, Wang J, Kim DH, Eom SH
EMBO reports 2015 Oct;16(10):1318-33
EMBO reports 2015 Oct;16(10):1318-33
Dynamics of enhanced mitochondrial respiration in female compared with male rat cerebral arteries.
Rutkai I, Dutta S, Katakam PV, Busija DW
American journal of physiology. Heart and circulatory physiology 2015 Nov;309(9):H1490-500
American journal of physiology. Heart and circulatory physiology 2015 Nov;309(9):H1490-500
Cytochrome c oxidase deficiency accelerates mitochondrial apoptosis by activating ceramide synthase 6.
Schüll S, Günther SD, Brodesser S, Seeger JM, Tosetti B, Wiegmann K, Pongratz C, Diaz F, Witt A, Andree M, Brinkmann K, Krönke M, Wiesner RJ, Kashkar H
Cell death & disease 2015 Mar 12;6(3):e1691
Cell death & disease 2015 Mar 12;6(3):e1691
A keratin scaffold regulates epidermal barrier formation, mitochondrial lipid composition, and activity.
Kumar V, Bouameur JE, Bär J, Rice RH, Hornig-Do HT, Roop DR, Schwarz N, Brodesser S, Thiering S, Leube RE, Wiesner RJ, Vijayaraj P, Brazel CB, Heller S, Binder H, Löffler-Wirth H, Seibel P, Magin TM
The Journal of cell biology 2015 Dec 7;211(5):1057-75
The Journal of cell biology 2015 Dec 7;211(5):1057-75
Imbalanced OPA1 processing and mitochondrial fragmentation cause heart failure in mice.
Wai T, García-Prieto J, Baker MJ, Merkwirth C, Benit P, Rustin P, Rupérez FJ, Barbas C, Ibañez B, Langer T
Science (New York, N.Y.) 2015 Dec 4;350(6265):aad0116
Science (New York, N.Y.) 2015 Dec 4;350(6265):aad0116
CNC-bZIP protein Nrf1-dependent regulation of glucose-stimulated insulin secretion.
Zheng H, Fu J, Xue P, Zhao R, Dong J, Liu D, Yamamoto M, Tong Q, Teng W, Qu W, Zhang Q, Andersen ME, Pi J
Antioxidants & redox signaling 2015 Apr 1;22(10):819-31
Antioxidants & redox signaling 2015 Apr 1;22(10):819-31
Mitochondrial dynamics associated with oxygen-glucose deprivation in rat primary neuronal cultures.
Wappler EA, Institoris A, Dutta S, Katakam PV, Busija DW
PloS one 2013;8(5):e63206
PloS one 2013;8(5):e63206
Identification of mitochondrial dysfunction in Hutchinson-Gilford progeria syndrome through use of stable isotope labeling with amino acids in cell culture.
Rivera-Torres J, Acín-Perez R, Cabezas-Sánchez P, Osorio FG, Gonzalez-Gómez C, Megias D, Cámara C, López-Otín C, Enríquez JA, Luque-García JL, Andrés V
Journal of proteomics 2013 Oct 8;91:466-77
Journal of proteomics 2013 Oct 8;91:466-77
Irgm1 (LRG-47), a regulator of cell-autonomous immunity, does not localize to mycobacterial or listerial phagosomes in IFN-γ-induced mouse cells.
Springer HM, Schramm M, Taylor GA, Howard JC
Journal of immunology (Baltimore, Md. : 1950) 2013 Aug 15;191(4):1765-74
Journal of immunology (Baltimore, Md. : 1950) 2013 Aug 15;191(4):1765-74
IKKα and alternative NF-κB regulate PGC-1β to promote oxidative muscle metabolism.
Bakkar N, Ladner K, Canan BD, Liyanarachchi S, Bal NC, Pant M, Periasamy M, Li Q, Janssen PM, Guttridge DC
The Journal of cell biology 2012 Feb 20;196(4):497-511
The Journal of cell biology 2012 Feb 20;196(4):497-511
Identification of human fumarylacetoacetate hydrolase domain-containing protein 1 (FAHD1) as a novel mitochondrial acylpyruvase.
Pircher H, Straganz GD, Ehehalt D, Morrow G, Tanguay RM, Jansen-Dürr P
The Journal of biological chemistry 2011 Oct 21;286(42):36500-8
The Journal of biological chemistry 2011 Oct 21;286(42):36500-8
Mitochondrial DNA toxicity compromises mitochondrial dynamics and induces hippocampal antioxidant defenses.
Lauritzen KH, Cheng C, Wiksen H, Bergersen LH, Klungland A
DNA repair 2011 Jun 10;10(6):639-53
DNA repair 2011 Jun 10;10(6):639-53
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
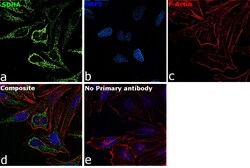
- Experimental details
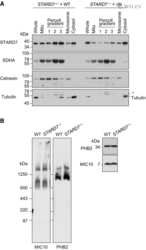
- Immunofluorescence analysis of SDHA was performed using 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with SDHA Monoclonal Antibody (2E3GC12FB2AE2) (Product # 459200) at 1:200 dilution in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32723), (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b:Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300 dilution). Panel d represents the merged image showing cytoplasmic (mitochondria-like) localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
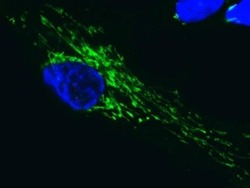
- Experimental details
- Immunofluorescent analysis of SDHA in human embryonic lung-derived fibroblasts using an SDHA Monoclonal antibody (Product # 459200) at a concentration of 0.2 µg/mL. Cultured human embryonic lung-derived fibroblasts (strain MRC5) were fixed, permeabilized and then labeled with an SDHA Monoclonal antibody (Product # 459200) at a concentration of 0.2 µg/mL, followed by an AlexaFluor® 488-conjugated-goat-anti-mouse IgG2a isotype specific secondary antibody at a concentration of 2 µg/mL.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
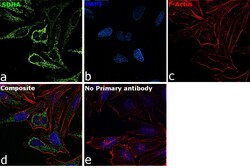
- Experimental details
- Immunofluorescence analysis of SDHA was performed using 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with SDHA Monoclonal Antibody (2E3GC12FB2AE2) (Product # 459200) at 1:200 dilution in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32723), (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b:Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300 dilution). Panel d represents the merged image showing cytoplasmic (mitochondria-like) localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
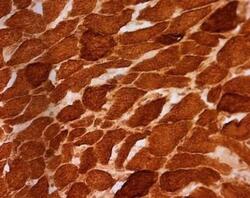
- Experimental details
- Immunohistochemical analysis of SDHA in skeletal muscle using an SDHA Monoclonal antibody (Product # 459200). Fixed frozen skeletal muscle tissue sections are from a patient with a single large deletion of the mtDNA. All muscle fibers exhibit complex II immunoreactivity, consistent with the nuclear DNA-encoded expression pattern of this and all other subunits of complex II.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
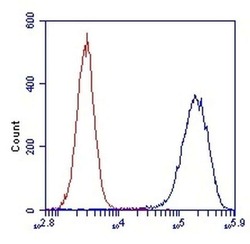
- Experimental details
- Flow cytometric analysis of SDHA in HL-60 cells using an SDHA monoclonal antibody (Product # 459200) at 1µg/mL is depicted by the blue line. The red line indicates an isotype control antibody.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
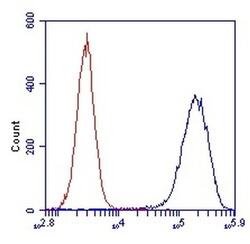
- Experimental details
- Flow cytometric analysis of SDHA in HL-60 cells using an SDHA monoclonal antibody (Product # 459200) at 1µg/mL is depicted by the blue line. The red line indicates an isotype control antibody.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
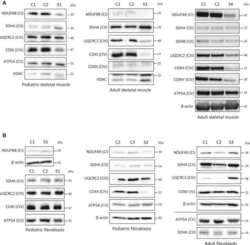
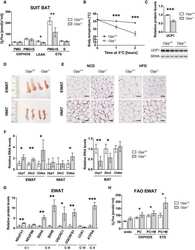
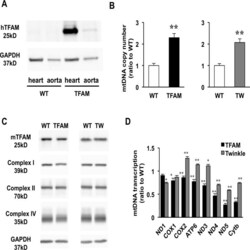
- Fig 1 Characterization of TFAM and Twinkle (TW) mice. (A) Expression of human TFAM (hTFAM) in left ventricle (LV) and aorta in TFAM and wild-type (WT) control mice. (B) mtDNA copy number in myocardium from TFAM and TW mice by real-time PCR (n = 4). (C) Expression of endogenous murine TFAM (mTFAM) and mitochondrial complex proteins in LV of TFAM and TW mice. (D) Transcription of mtDNA-encoded genes in TFAM and TW mice (n = 6). Data are expressed as mean +- SEM. * P < 0.05 vs. WT, ** P < 0.01 vs. WT, analyzed by Student's t- test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
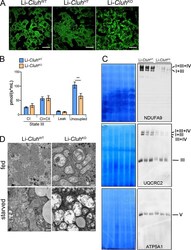
- Figure 5. Liver-specific Cluh deletion affects mitochondrial distribution and structure, assembled respiratory supercomplexes, and respiratory capacity. (A) Representative confocal images of livers of 8-wk-old mice of the indicated genotypes. To analyze mitochondrial morphology, mice were crossed with a stop-mito-YFP reporter line activated by Cre recombination. n = 4. Bars, 5 um. (B) Oxygen consumption of mitochondria isolated from livers of 8-wk-old mice. State III respiration was measured in the presence of pyruvate, malate, glutamate, and ADP (complex I [CI]), followed by addition of succinate (complex I + complex II [CI + CII]). The proton leak was measured after addition of oligomycin, whereas maximal respiration was assessed by CCCP titration. n = 5. Graph shows means +- SEM. **, P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
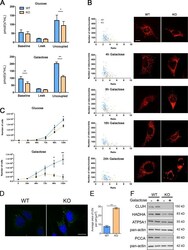
- Figure 7. Cluh -deficient MEFs mimic liver phenotypes. (A) Oxygen consumption of intact MEFs cultured in glucose or galactose medium. The proton leak was measured after the addition of oligomycin, whereas maximal respiration was assessed by CCCP titration. n >= 4. (B) Mitochondrial morphology in MEFs transfected with mito-mCherry. Graphs show the mean aspect ratio (area/perimeter) on the x axis and the number of mitochondria on the y axis for individual cells from three independent experiments. Right panels show representative images of mitochondrial morphology at the indicated time points. Bar, 12 um. (C) Growth curves of MEFs cultured in glucose or galactose medium during five consecutive days. n = 3. (D) Representative images of LD staining in MEFs grown in glucose medium. Nuclei were stained with DAPI (blue), and LDs were stained with BODIPY 493/503 (green). Bar, 20 um. (E) Quantification of LD staining shown in D. 50 cells were analyzed per genotype per experiment. Graph shows the mean area of LDs per cell. n = 3. (A, C, and E) Error bars are means +- SEM. *, P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
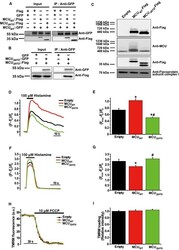
- Experimental details
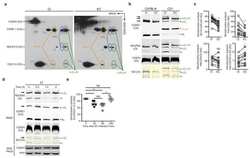
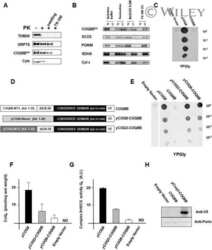
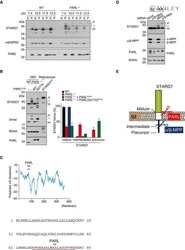
- Figure 3 MCU Delta NTD overexpression has a dominant-negative effect on mitochondrial Ca 2+ uptake A Co-immunoprecipitation of MCU WT -Flag or MCU Delta NTD -Flag with MCU WT - GFP . HeLa cells were transiently co-transfected with MCU WT - GFP and MCU WT -Flag/ MCU Delta NTD -Flag. MCU Delta NTD migrated farther than MCU WT , indicating an apparent molecular weight difference of 9 kD a in SDS - PAGE (see also Fig EV4 A). MCU WT - GFP with MCU WT -Flag or MCU Delta NTD -Flag was precipitated from the cell lysates with an anti- GFP antibody. The precipitates were separated on SDS - PAGE and immunoblotted with the antibodies indicated. B Co-immunoprecipitation of MCU Delta NTD -Flag with MCU Delta NTD - GFP . HeLa cells were transiently co-transfected with MCU Delta NTD -Flag and MCU Delta NTD - GFP . MCU Delta NTD - GFP with MCU Delta NTD -Flag was precipitated from cell lysates with an anti- GFP antibody. The precipitates were separated on SDS - PAGE and immunoblotted with the indicated antibodies. C HeLa cells were transiently transfected with MCU WT -Flag or MCU Delta NTD -Flag. After isolation and solubilization of crude mitochondria, the lysates were subjected to BN-PAGE and immunoblotted with anti-Flag and anti- MCU antibodies to detect ectopic MCU WT -Flag, MCU Delta NTD -Flag and endogenous MCU . MCU WT and MCU Delta NTD were detected at apparent molecular weights of 480 and 440 kD a, respectively. The shift shown in MCU Delta NTD complex correlated with the difference
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 Focal depletion of mitochondria in skeletal muscles of a human R350P desminopathy patient and R349P desmin knock-in mice. a Cytochrome C oxidase (COX, brown ) and succinate dehydrogenase (SDH, blue ) double-stains of transverse and longitudinal cryosections from a German patient harboring the heterozygous R350P (c.1049G>C) desmin missense mutation [ 3 ]. Note the multiple rubbed-out areas ( asterisks ) devoid of COX and SDH enzyme activities demonstrating the absence of mitochondria. The right image represents a false color representation of two superimposed serial cryosections stained for COX ( magenta ) and desmin ( black ). Sarcoplasmic desmin-positive protein aggregates clearly display a distribution which is independent from the mitochondrial lesion pathology. b , c COX ( b ) and SDH ( c ) stains of transverse and longitudinal cryosections from 3-month-old R349P (c.1045_1047delAGG>insCCC) desmin knock-in mice. Fibers of homozygote (HOM) animals display large and small areas of diminished enzyme stains ( asterisks ). Furthermore, muscle fibers in homozygote animals show an abnormal, thread-like distribution of mitochondria. Heterozygous (HET) mice showed no overt pathology as compared to wild-type (WT) littermates
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
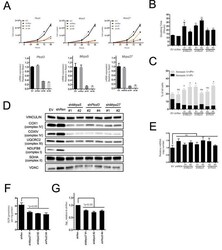
- Experimental details
- Figure 2 The mitochondrial translation machinery regulates global mitochondrial activity and Emu -myc cell growth A. Emu- myc lymphoma cells were transduced with shRNAs against Ptcd3 , Mrps5 , Mrps27 , Renilla luciferase (Ren) or a control empty vector (EV). The number of viable cells at each time point (top) was determined by trypan blue staining. For each single shRNA the efficiency of knockdown was evaluated by real-time qPCR three days post-transduction (bottom). Transcript abundance is expressed as mean +- s.d. of triplicate measurements, expressed relative to the EV control and normalized to Rplp0 . B. Doubling time was calculated from the growth curves in A., using the formula described in experimental procedures. C. Cell death was evaluated by Annexin V and propidium iodide (PI) staining at the 48hrs time-point. The graph shows the percentage of Annexin V+/PI- (black) and Annexin V+/PI+ cells (grey) corresponding to early and late apoptotic cells, respectively. D. Western blot analysis of components of the Electron Transport Chain (ETC) Complexes I-IV following 48hrs of knock-down. As loading controls, we used antibodies against cytoplasmic Vinculin and the mitochondrial Voltage-dependent anion channel (VDAC). E. Real-time qPCR quantification of mtDNA, normalized to nuclear DNA. Results are shown as mean +- s.d. from triplicate measurements. F. Oxygen consumption rate (OCR) was determined as described in experimental procedures at 48hrs. Data were normalized to total
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
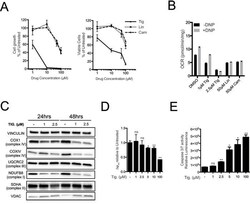
- Experimental details
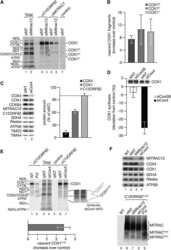
- Figure 3 Tigecycline impairs the growth of Emu -myc lymphoma cells and inhibits translation of mitochondrial proteins A. Dose-response curve of Emu- myc lymphoma cells treated with the indicated doses of Tigecycline (Tig), Linezolid (Lin) or Cloramphenicol (Cam) for 48hrs. Data are shown as cell numbers (left panel) or viability (right panel), as percentage of untreated cells (mock-treated with DMSO). Cell number and viability was determined by trypan blue staining. B. Oxygen consumption rate (OCR) measured as defined in Figure 2F on Emu- myc lymphoma cells treated with the indicated antibiotics or carrier (DMSO). To measure spare respiratory capacity, cells were treated with the ETC uncoupler 2,4-Dinitrophenol (DNP). C. Western blot analysis of components of the ETC complexes I-IV. Cells were treated with the indicated concentration of Tigecycline for 24 or 48hrs. The loading controls are Vinculin and VDAC, as in Figure 2D . D. Mitochondrial membrane potential was determined by staining with the cationic cyanine dye DilC1(5). E. Cleaved caspase 3/7 activity was determined with the caspase 3/7 glo assay luminescent kit. Measurements in D and E were taken after 6 hrs of treatment. Means, s.d. and statistical significance are as defined in Figure 2 . See also Supplementary Figures S3 and S4 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
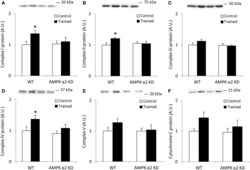
- Experimental details
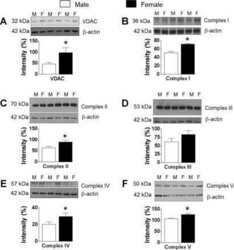
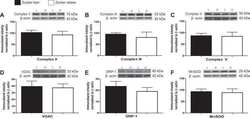
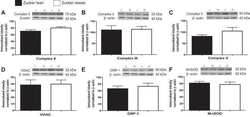
- Figure 1 Exercise training increase mitochondrial oxidative phosphorylation complexes in an AMPK alpha2-dependent manner . Protein levels of oxidative phosphorylation complexes, (A) Complex I, (B) Complex II, (C) Complex III, (D) Complex IV, (E) Complex V (oligomycin-sensitivity conferring protein subunit, OSCP), and (F) Cytochrome C were evaluated in quadriceps muscle of sedentary (control) or exercise trained female WT and AMPK alpha2 KD mice ( n = 13-15). Values are mean +- SEM. * indicates vs. WT control ( p < 0.05) analyzed by unpaired t -tests.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
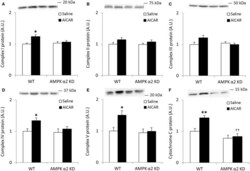
- Experimental details
- Figure 4 Repeated treatment with AICAR increases abundance of mitochondrial electron transport chain proteins in an AMPK alpha2-dependent manner . WT and AMPK alpha2 KD male mice were given daily subcutaneous injections with AICAR (500 mg/kg body weight) or saline for 4 weeks. (A-E) Protein abundance of oxidative phosphorylation Complexes I-V and (F) Cytochrome C was measured in quadriceps muscle ( n = 7-8). A significant interaction effect (treatment x genotype; p < 0.05) was observed for Cytochrome C. Values are mean +- SEM. * indicates vs. WT saline ( p < 0.05) analyzed by t -tests, ** indicates vs. WT saline ( p < 0.01), and ++ indicates genotype effect within AICAR treated animals ( p < 0.01).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
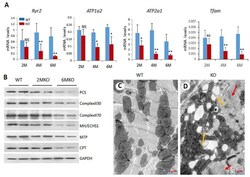
- Experimental details
- Figure 3 csPIMT -/- hearts show significant mitochondrial damage. ( A ) Quantification of mRNA levels of Atp1a2, Atp2a1, Ryr2 and Tfam genes. Each group was analyzed using 5 different mice (assayed individually) and the values are expressed as the mean +- SD. * p < 0.05, ** p < 001, NS: not significant; ( B ) Western blot showing protein levels for PCS (palmitoyl-CoA synthetase; 62 Kda), complex II30 and 70, MH/ECHS1 (mitochondrial enoyl-CoA hydratase; 31 Kda), MTP (mitochondrial trifunctional protein; 100 Kda) and CPT1alpha (carnitine palmitoyltransferase; 88 Kda)). The protein extracts were prepared from 5 hearts pooled together. The protein expression of each gene was normalized to GAPDH. Percent decrease as compared WT controls were as follows: PCS, 67%; complex II30 and 70, 28% and 38%; MTP, 26%, and CPT, 42%; ( C , D ) display the electron micrographs of 6-month csPIMT fl/fl and csPIMT -/- mouse hearts. Red arrows in D indicate abnormal sarcomeres and H zone absent. Yellow arrows point to lipid droplets and damage in mitochondria.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 1. IKKalpha regulates mitochondrial biogenesis and fiber type specification in vivo. (A) Hind limbs from four independent litters of IKKalpha +/+ and IKKalpha -/- embryos (total n = 8) were isolated at E20. Cytochrome c and COX Va were measured by real-time quantitative RT-PCR. (B) Additional tissues from E20 limbs from A were homogenized and probed by Western blotting. (C) Total DNA was extracted from E20 limbs, and real-time RT-PCR analysis was performed to quantitate mitochondrial and nuclear (GAPDH) genome copy number. Results are means of five mice per genotype and calculated as fold mitochondrial over genomic DNA. P = 0.004. (D) AAV-GFP and AAV-IKKalpha viruses were injected into the TA. Mice were sacrificed after 3-4 mo, and protein lysates were prepared and analyzed by Western blotting. Shown is a representative blot from five independent experiments. (E and F) After similar viral injections as described in D, mitochondria were fractionated, and protein concentrations or DNA content was determined and normalized to total protein/DNA content (E; n = 5 per group; P < 0.005) or TA muscles were analyzed by EM (F). Bar, 1 um. Parallel muscles were stained for SDH. Bar, 200 um. Arrowheads point to individual mitochondria. (G) TA muscles were isolated from AAV-GFP- and AAV-IKKalpha-injected mice ( n = 3 per group, repeated five times), and myosin isoforms were measured by real-time RT-PCR. The data shown are from a single representative experiment out of three replica
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
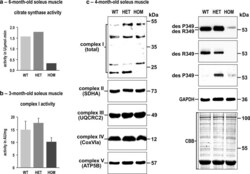
- Experimental details
- Fig. 4 Citrate synthase and complex I activities and expression levels of single subunit proteins of respiratory complexes I-V in R349P desmin knock-in mice. a Determination of the citrate synthase activity by a spectrophotometric assay using identical amounts of total protein extracts. Note the marked activity reduction in homozygous mice. For this approach, soleus muscles obtained from five mice of each genotype (WT, HET, HOM) were pooled and subjected to four-time repeated measurements. Column chart shows mean values of these technical replicates. b Determination of complex I activity by a colorimetric assay normalized to muscle weight. Though this analysis, performed in duplicate on non-pooled samples derived from three animals per genotype, showed a reduction of complex I activity in homozygous mice, the data failed to reach statistical significance. An additional normalization of the obtained complex I values to the citrate synthase values resulted in similar activity levels for the three genotypes (data not shown). Column chart shows mean values and standard errors. c Immunoblotting using antibodies directed against specific complex I-V proteins revealed increased, decreased, and missing signals of complex I subunit proteins in heterozygous and homozygous mice, whereas the signal intensities of complex II-V subunit proteins showed no obvious differences. Desmin immunoblotting confirmed a markedly decreased level of mutant desmin in homozygous mice. In heterozygous mice
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
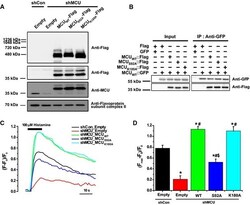
- Figure 5 Substitution of Ser92 to Ala in MCU NTD reduced mitochondrial Ca 2+ uptake activity A Expression of MCU WT -Flag, MCU S 92A -Flag and MCU K 180A -Flag in stable MCU - KD HeLa cells. The solubilized mitochondrial fraction from each sample was subjected to BN - PAGE and immunoblotted with anti-Flag antibody. The lysates of each sample were subjected to SDS - PAGE and detected with the indicated antibodies. Flavoprotein subunit of complex I is used as a loading control of each mitochondrial fraction. B Interaction of MCU mutants with MCU WT was not altered. After co-expression of MCU WT and MCU mutants, co-immunoprecipitation was performed. The precipitates were separated by SDS - PAGE and immunoblotted with the indicated antibodies. C, D Mitochondrial Ca 2+ uptakes evoked by 100 muM histamine were quantified by measuring the peak amplitudes of the traces. F 0 is initial fluorescence intensity. F and F max indicate fluorescence intensity at each time point and maximal fluorescence intensity after the stimulation, respectively. (F max -F 0 )/F 0 indicates the maximal Ca 2+ concentration evoked by the stimulation (mean +- SEM , n = 13-15, * P < 0.05 versus control sh RNA -expressing cells. # P < 0.05 versus stable MCU - KD cells. SS P < 0.05 versus MCU WT -rescued stable MCU - KD cells). Unpaired two-sided Student's t -test was used to calculate statistical significance.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
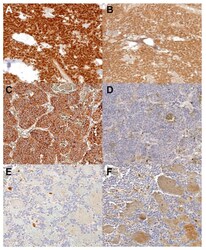
- Figure 3 SDHA and SDHB immunoreactivity. ( A , C , E )--SDHA immunohistochemistry. ( B , D , F )--SDHB immunoreactivity. ( A ) and ( B ) represent a case with both SDHA and SDHB immunoreactivity; ( C ) and ( D ) have SDHA but lack SDHB immunoreactivity; ( E ) and ( F ) represent a case lacking both SDHA and SDHB immunoreactivity. SDHA--Succinate dehydrogenase A, SDHB--Succinate dehydrogenase B, Original objective 40x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
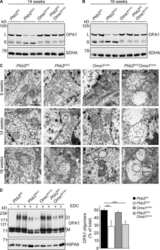
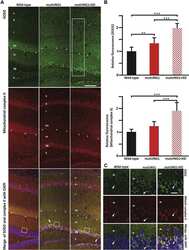
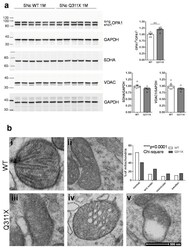
- Figure 3 Expression of the human parkin variant parkinQ311X induces early mitochondrial dysfunction in SNc DA neurons. ( a ) Representative Western blots showing the levels of mitochondrial proteins (OPA1, SDHA, VDAC) in lysates prepared from the substantia nigra of WT or parkinQ311X mice at 1 month of age. Samples were run in triplicate, with each lane loaded with lysate from an individual mouse. The histograms on the right show the mean +-SEM calculated from densitometer quantification (** p < 0.01 unpaired Student's t -test). ( b ) Representative TEM images showing mitochondria in SNc DA neurons of WT (control, i,ii ) and parkinQ311X ( iii,iv,v ) mice. The mitochondria in control specimens show the typical rounded/tubular morphology ( i,ii ). The mitochondria in parkinQ311X mice display marked ultrastructure disruption ( iii,iv,v ) and a lack of the typical crystal organization found in the control tissue: 26% of the mitochondria in parkinQ311X DA neurons displayed an almost complete loss of outer membrane shape and form ( iii ). Other mitochondria in the parkinQ311X DA neurons retained outer membrane integrity but demonstrated ultrastructural damage: dilation of intracrystal spaces, loss of matrix density, and deposits of electron-dense material within the matrix (vesicular mitochondria) ( iv ). Swollen, clear mitochondria showed herniation of the swollen matrix covered by the inner membrane through the ruptured regions of the outer membrane ( v ). The histograms on the r
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
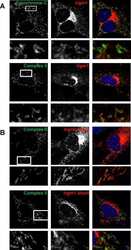
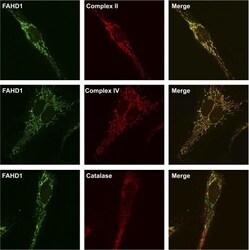
- FIGURE 8. FAHD1 immunofluorescence. Confocal images of immunofluorescence staining of HUVEC for FAHD1 ( green , left column ), three different localization markers ( red , center column ), and the resulting merge ( right column ) are shown. Complex II and complex IV were used as mitochondrial markers, and catalase was used as peroxisomal marker.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
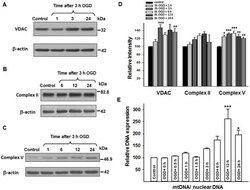
- Figure 5 Changes in VDAC, complex II and V proteins, and mtDNA expression following 3 h OGD. Representative western blots for VDAC, Complex II, and Complex V with their corresponding beta-actin blots (A, B, C), together with relative intensity values for these proteins (D). VDAC and Complex V proteins showed increased expression, whereas Complex II protein expression did not change significantly following OGD. (E) Shows the changes in mtDNA expression following 3 h of OGD. MtDNA/nuclear DNA ratio significantly increased 12 h following 3 h OGD. *p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
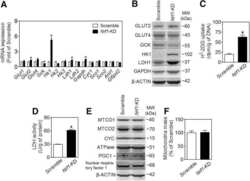
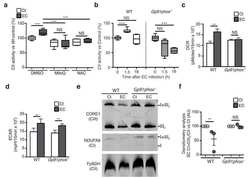
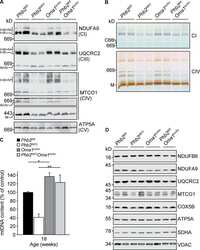
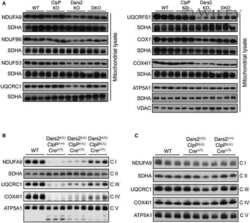
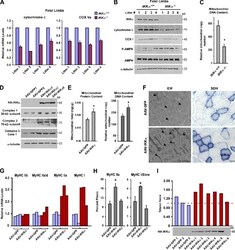
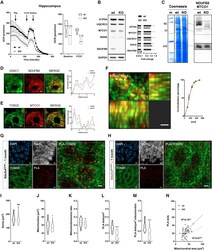
- Figure 2 CI*CIV-SC content diminishes in the brain of Ndusf4 KO mice (A) OCR measurement in ex vivo hippocampal brain sections from control (WT) and Ndufs4 knockout mice (KO) (WT, n = 3; Ndufs4 KO, n = 3, two-way RM ANOVA). In the violin plot, numbers represent p values. (B) Immunoblot analysis of samples from WT and KO mice using antibodies against ETC subunits (CI, NDUFB8; CII, SDHA; CIII, UQCRC2; CIV, MTCO1; CV, ATP5A; WT, n = 2; Ndufs4 KO, n = 2). GAPDH was used as loading control. Densitometry is relative to WT samples and reported as mean +- SEM (nd, not determined). (C) BN-PAGE and composite image of immunoblots obtained using antibodies against NDUFB8 (CI) and MTCO1 (CIV, cropped bands). Samples were from WT and KO brain homogenates (WT, n = 3; Ndufs4 KO, n = 3). (D and E) Coimmunostaining and fluorescence profiles of (D) VDAC1 and NDUFB8 and (E) TOM20 and MTCO1 in control mouse brain sections (two-way ANOVA, *p < 0.05). Scale bar, 5 mum. (F) Colocalization between mitochondria (TOM20) and CI*CIV-SCs (PLA). Scale bar, 1 mum. (G and H) Representative pictures of hippocampal CA1 pyramidal neurons from (G) WT and (H) KO mice stained with DAPI (nucleus), NeuN (neurons), TOM20 (mitochondria), and PLA (CI*CIV-SCs). Scale bar, 5 mum. (I-N) Quantification of (I) area of the soma, (J) mitochondrial area, (K) mitochondria/soma ratio, (L) PLA dots normalized to the soma, (M) PLA quantification of CI*CIV-SCs normalized to the mitochondrial area, and (N) linear correlation of PLA
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
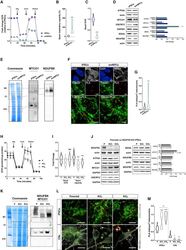
- Figure 4 CI*CIV-SC remodeling occurs during iPSC differentiation (A) OCR measurement of iPSCs and iPSC-derived smNPCs using a conventional Seahorse protocol (n = 3). (B) Mitochondrial spare respiratory capacity in human iPSCs and smNPCs. Percentage is relative to their respective basal OCRs (Student's t test, ****p < 0.0001). (C) Basal OCR in iPSCs and smNPCs (Student's t test, ****p < 0.0001). (D) Immunoblots using antibodies against OXPHOS system components in iPSCs and smNPCs. Densitometry is relative to iPSCs and reported as mean +- SEM (n = 3, Student's t test, ***p < 0.001, *p < 0.05). (E) BN-PAGE and corresponding immunoblots using antibodies against MTCO1 and NDUFB8. (F) Representative pictures of iPSCs and smNPCs stained with DAPI (nucleus, blue), TOM20 (mitochondria, green), and PLA (CI*CIV-SCs, red). Oct3 and nestin staining (both in gray) was used as markers of pluripotency and differentiation, respectively. Scale bar, 5 mum. (G) Quantification of PLA dots normalized to mitochondrial area in iPSCs and smNPCs (n = 3; iPSCs = 20 cells; smNPCs = 17 cells; Student's t test, ***p < 0.001). (H) OCR measurement of WT (P) and two independent clones of NDUFS4 KO iPSCs (KO 1 and KO 2 ). (I) Basal OCR and mitochondrial spare capacity of WT and NDUFS4 KO iPSCs (fold change, relative to WT; two-way RM ANOVA, ***p < 0.001, *p < 0.05; ns, not significant). (J) Immunoblot analysis of homogenates from parental and NDUFS4 KO iPSCs using antibodies against ETC subunits (CI, NDUFS4,
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
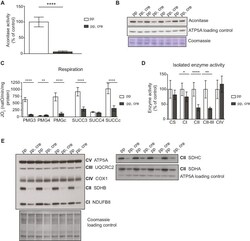
- Figure 5. [4Fe-4S] cluster proteins are severely affected in heart Sod2 knockout mice indicating strong increase in superoxide levels. ( A ) Aconitase activity from purified mitochondria from control (pp) and Sod2 loxP x Ckmm cre mice (pp, cre). White bar indicates activity in control samples ( n = 6, 9-10 week old) and gray bar in Sod2 loxP x Ckmm cre samples ( n = 6, 9-12 week old). Activity is normalized to control. ( B ) Western blot analysis of ACO 2 (aconitase) protein levels from purified mitochondria of control (pp) and Sod2 loxP x Ckmm cre mice (pp, cre) (9-10 week old). ATP5A and Coomassie-stained membrane were used as loading controls. ( C ) Oxygen consumption rate of isolated heart mitochondria from control (pp, white bars, n = 9, 9-11 week old) and Sod2 loxP x Ckmm cre mice (pp, cre, gray bars, n = 9, 9-12 week old). Isolated mitochondria were incubated with complex I (PMG) or complex II (SUCC) substrates. Each set of substrates was successively combined with ADP (to assess the phosphorylating respiration, PMG3, SUCC3), oligomycin (to assess the non-phosphorylating respiration PMG4, SUCC4) and CCCP (to assess uncoupled respiration PMGc, SUCCc). ( D ) Activity of the respiratory chain complexes I (CI), II (CII), IV (CIV) and the activity from complex II to III (CII-III) of heart mitochondria from control (pp, write bars, n = 3, 11-week old) and Sod2 loxP x Ckmm cre mice (pp, cre, gray bars n = 3, 11-12 week old). Citrate synthase
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
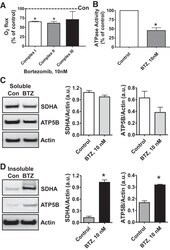
- Experimental details
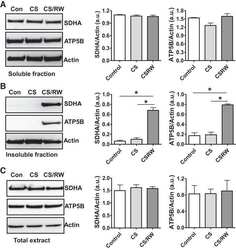
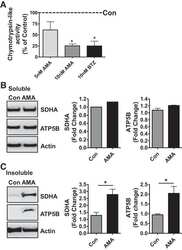
- Fig. 6. Proteasome inhibition decreases mitochondrial respiration and impairs mitochondrial respiratory complex protein homeostasis in NRK cells. A : digitonin-permeabilized NRK cells (5 x 10 6 ) were treated with vehicle control (DMSO) or the ChT-L proteasome inhibitor bortezomib (BTZ; 10 nM) for 24 h. High-resolution respirometry was used to determine oxygen flux through complexes I, II, and III. Values are expressed as % of control of 4 independent experiments. Differences between groups were compared with an unpaired Student's t- test. *Means are significantly different ( P < 0.05) compared with control. B : ATPase activity of ATP synthase (complex V) was assayed in NRK cells treated with DMSO (vehicle control) or BTZ for 24 h. Renal cell extracts (10 mug) with or without oligomycin (20 muM) were incubated with the enzyme mix in the presence of ATP and NADPH (MitoCheck Complex V Activity Assay kit, Cayman). NADH oxidation rate was monitored at 340 nm for up to 30 min with a BioTek Gen5 microplate reader. Bar graph shows oligomycin-inhibitable ATPase activity expressed as % of control ( n = 3). Differences between groups were compared with a Student's t -test. *Means are significantly different ( P < 0.001). C and D : Western blots of SDHA and ATP5B in soluble ( C ) and insoluble fractions ( D ) of NRK cell extracts after treatment with vehicle control (Con; DMSO) or BTZ (10 nM) for 24 h. Representative blots of 4 independent experiments are shown
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
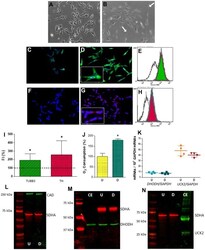
- Experimental details
- Figure 2 Expression of selected genes from pyrimidine nucleotide synthesis pathways in human neuroblastoma SH-SY5Y cells. ( A , B ) Representative optic microscopy images of ( A ) undifferentiated and ( B ) neuron-differentiated SH-SY5Y cells. White arrows point to neurites. ( C , D ) Representative immunofluorescence microscopy images of anti-TUBB3 stained ( C ) undifferentiated and ( D ) neuron-differentiated SH-SY5Y cells. Inset: enlarged figure detail. ( E ) Representative image of a flow cytometry histogram of anti-TUBB3 stained undifferentiated (white) and neuron-differentiated (green) cells. ( F , G ) Representative immunofluorescence microscopy images of anti-TH stained ( F ) undifferentiated and ( G ) neuron-differentiated SH-SY5Y cells. Inset: enlarged figure detail. ( H ) Representative image of a flow cytometry histogram of anti-TH stained undifferentiated (white) and neuron-differentiated (red) cells. ( I ) Graph showing the change of fluorescence intensity (FI) in TUBB3 and TH levels after neuronal differentiation. Dashed line (100 %) represents TUBB3 or TH mean values of undifferentiated cells. Bars indicate mean values and standard deviations in differentiated cells. N = 11. *: p < 0.05 (versus undifferentiated cells). ( J ) Oxygen consumption of (U) undifferentiated and ( D ) neuron-differentiated cells. N = 3. *: p < 0.05 (versus undifferentiated cells). ( K ) DHODH and UCK2 mRNA levels in (U) undifferentiated and ( D ) neuron-differentiated cells. Points re
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
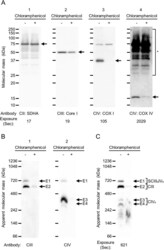
- Experimental details
- Fig. 4. Depletion of mitochondrially encoded components of CIII and CIV in N2a cells by inhibitor of mitochondrial translation . Mouse neuroblastoma N2a cells were treated for 120 h with chloramphenicol, which is an inhibitor of mitochondrial translation. (A) The effects of chloramphenicol treatment for the protein synthesis of components were examined. 5 mug/lane of whole-membrane protein of N2a cells with (+) or without (-) chloramphenicol treatment was separated by SDS-PAGE, and the components of complexes II, III and IV were detected by western blotting using anti-SDHA (A-1), anti-Core I (A-2) and anti-COX I antibodies (A-3). Then, anti-COX IV was reprobed onto the anti-SDHA-probed membrane without the stripping process, and complex IV was detected (A-4). Each arrow indicates SDHA (A-1), Core I (A-2), COX I (A-3) and COX IV (A-4). Asterisk indicates unstripping SDHA signals on the PVDF membrane (A-4). (B,C) 15 mug/lane of whole-membrane protein of N2a cells with (+) or without (-) chloramphenicol treatment were separated by hrCN-PAGE in a 3-14% gradient gel. E1-E4 denote signals detected by western blotting (B) and ECL solution (C). CIII (anti-Core I, B-1) and CIV (anti-COX I, B-2) were detected by western blotting with exposure times of 58 and 17 s, respectively. E1-E4 are shown as signals detected at the same position with the molecular mass. Peroxidase activity was assayed by the ECL solution after pre-incubation of 1% of SDS solution (C). CII, complex II; CIII, comple
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
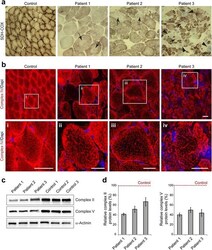
- Experimental details
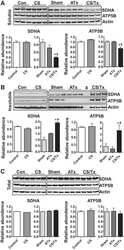
- Fig. 4 Mitochondrial alterations in EBS-MD muscle. a Skeletal muscle specimens from a healthy control and EBS-MD patients were histologically double-stained for SDH and COX. Note the presence of rubbed-out areas ( arrows ), and the presence of COX-negative fibers in patient 3 ( arrowheads ). Scale bar: 50 mum. b Confocal imaging of mitochondrial respiratory complex IV-stained skeletal muscle specimens from a healthy control and EBS-MD patients. Panels i-iv are magnifications of the boxed areas in panel b. Note the reduced staining intensity in all EBS-MD samples. Scale bars: 50 mum ( b ), 25 mum (panels i-iv). c Immunoblotting of cell lysates prepared from EBS-MD patients and three healthy controls using antibodies for mitochondrial respiratory complex II or V. alpha-Actinin was used as loading control. d Signal intensities of respiratory complex II or V protein bands as shown in ( c ) were densitometrically measured and normalized to the total protein content (assessed by alpha-actinin staining). Healthy controls ( dashed line ) are set to 100 %. Mean values +- SEM, 3 replicates
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
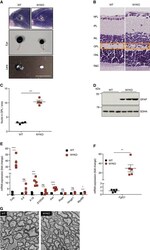
- Experimental details
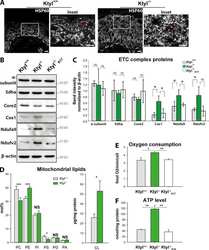
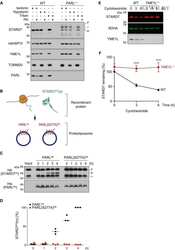
- Figure 1 Loss of YME1L in the nervous system causes microphthalmia, cataracts, and retinal inflammation A Representative images of eyes and lenses from 6- to 7-week-old wild-type (WT) and nervous system-specific YME1L knockout (NYKO) mice. Orange dashed lines mark eye morphology. Scale bars, 5 mm. B Retinal sagittal cross sections from 6- to 7-week-old mice stained with hematoxylin and eosin. NFL = nerve fiber layer, IPL = inner plexiform layer, INL = inner nuclear layer, OPL = outer plexiform layer, ONL = outer nuclear layer, R&C = rods and cones. Scale bars, 30 mum. C Quantification of nuclei in OPL (area = 1,000 mum 2 ) from retina cross sections of 6- to 7-week-old WT ( n = 4) and NYKO ( n = 4) mice. D Immunoblot analysis of retinal lysates from 6- to 7-week-old WT and NYKO mice. GFAP was used as a marker for reactive astrogliosis and SDHA as a loading control. E mRNA levels of proinflammatory cytokines and NF-kappaB target genes from 6- to 7-week-old retinas (WT, n = 5; NYKO, n = 5). Transcript levels were normalized to Hprt mRNA levels. F mRNA levels of Fgf21 from 6- to 7-week-old retinas (WT, n = 5; NYKO, n = 5). Transcript levels were normalized to Hprt mRNA levels. G Transmission electron micrographs of optic nerves from 6- to 7-week-old WT and NYKO mice. Scale bars, 2 mum. Data information: Data were analyzed using unpaired t -test, * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
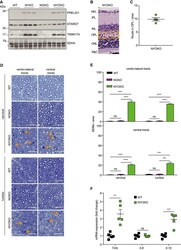
- Experimental details
- Figure EV4 Ablation of Oma1 in NYKO mice does not restore retinal organization and causes axonal degeneration in ventro-lateral and ventral tracts of the spinal cord A Immunoblot analysis of spinal cord lysates from 6- to 7-week-old WT, NYKO, NOKO, and NYOKO mice ( n = 3) using the indicated antibodies. SDHA was used as a loading control. * indicates unspecific antibody binding. B, C Retinal sagittal cross sections from 6- to 7-week-old NYOKO mice ( n = 4) stained with hematoxylin and eosin. NFL = nerve fiber layer, IPL = inner plexiform layer, INL = inner nuclear layer, OPL = outer plexiform layer, ONL = outer nuclear layer, R&C = rods and cones. Scale bar, 30 mum. D Transverse semithin sections of spinal cords of 6- to 7-week-old WT, NOKO, and NYOKO mice. Sections were stained with toluidine blue. Orange arrows point to MDBs indicating degenerating neurons. Scale bars, 25 mum. E MDBs in ventro-lateral and ventral tracts of 6- to 7-week-old WT ( n = 3), NOKO ( n = 3), and NYOKO ( n = 3) mice. F mRNA levels of proinflammatory cytokines from 6- to 7-week-old retinas (WT, n = 5; NYOKO, n = 5). Transcript levels were normalized to Hprt mRNA levels. Data information: Unpaired t -test was used for comparison of two groups, ordinary one-way ANOVA for comparison of three groups. ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
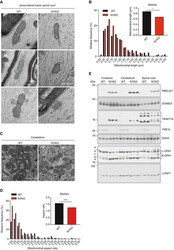
- Experimental details
- Figure 7 Defective proteostasis and accelerated OPA1 processing in brain and spinal cord of NYKO mice A TEM analysis of sagittal spinal cord sections of 6- to 7-week-old WT and NYKO mice. Scale bars, 500 nm. B Frequency distribution of mitochondrial lengths in spinal cords of 6- to 7-week-old WT (182 mitochondria) and NYKO mice (192 mitochondria). Kruskal-Wallis test, * P < 0.05. C TEM analysis of cerebellar sections of 28- to 32-week-old WT and NYKO mice. Scale bars, 1 mum. D Frequency distribution of mitochondrial aspect ratios were calculated from WT ( n = 210) and NYKO ( n = 230) mitochondria. Mann-Whitney test, **** P < 0.0001. Data are presented as median values. E Immunoblot analysis of forebrain, cerebellar and spinal cord lysates from 31- to 32-week-old WT and NYKO mice ( n = 3) using the indicated antibodies. SDHA and LONP1 were used to control for gel loading. * indicates unspecific antibody binding. Source data are available online for this figure.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
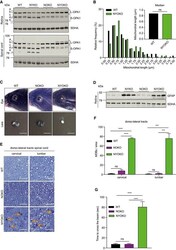
- Experimental details
- Figure 8 Loss of OMA1 does not restore ocular dysfunction and deteriorates axonal degeneration in the spinal cord of NYKO mice A Immunoblot analysis of retinal and spinal cord lysates from 6- to 7-week-old WT, NYKO, NOKO, and NYOKO mice ( n = 3) using the indicated antibodies. SDHA was used as a loading control. * was not reproducibly detectable in all experiments. B Frequency distribution of mitochondrial lengths in spinal cords of 6- to 7-week-old NYOKO mice (176 mitochondria) compared to WT (as in Fig 7 B). Kruskal-Wallis test, ns = not significant. C Representative images of eyes and lenses from 6- to 7-week-old WT and mice lacking OMA1 (NOKO) or both YME1L and OMA1 (NYOKO) in the nervous system. Orange dashed lines mark eye morphology. Scale bars, 2.5 mm. D Immunoblot analysis of retina lysates of 6- to 7-week-old WT, NYKO, NOKO, and NYOKO mice ( n = 3). SDHA was used to control for gel loading. E Transverse semithin sections of spinal cords of 6- to 7-week-old WT, NOKO, and NYOKO mice. Sections were stained with toluidine blue. Orange arrows point to MDBs indicating degenerating neurons. Scale bars, 25 mum. F MDBs in dorso-lateral tracts of 6- to 7-week-old WT ( n = 3), NOKO ( n = 3), and NYOKO ( n = 3) mice. G Walking beam test of 6- to 7-week-old WT ( n = 6), NOKO ( n = 5), and NYOKO ( n = 7) mice. Data information: Data were analyzed using one-way ANOVA. *** P
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry