Antibody data
- Antibody Data
- Antigen structure
- References [11]
- Comments [0]
- Validations
- Immunocytochemistry [7]
- Other assay [6]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA1-977 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- PSMB5 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- PA1-977 detects proteasome 20S X from bovine, mouse and human samples. PA1-977 has been successfully used in Western blot and ICC/IF procedures. By Western blot, this antibody detects an ~23 kDa protein representing proteasome 20S X. The PA1-977 immunizing peptide corresponds to amino acid residues 243-259 from mouse proteasome 20S X. This sequence is 94% conserved in human and rat. PA1-977 immunizing peptide (Cat. # PEP-172) is available for use in neutralization and control procedures.
- Reactivity
- Human, Mouse, Bovine
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 μg
- Concentration
- 1 mg/mL
- Storage
- -20°C, Avoid Freeze/Thaw Cycles
Submitted references Evaluation of Immunoproteasome-Specific Proteolytic Activity Using Fluorogenic Peptide Substrates.
A protein-protein interaction map reveals that the Coxiella burnetii effector CirB inhibits host proteasome activity.
H727 cells are inherently resistant to the proteasome inhibitor carfilzomib, yet require proteasome activity for cell survival and growth.
Skeletal muscle denervation investigations: selecting an experimental control wisely.
Novel Potent Capsid Assembly Modulators Regulate Multiple Steps of the Hepatitis B Virus Life Cycle.
Induction of the Immunoproteasome Subunit Lmp7 Links Proteostasis and Immunity in α-Synuclein Aggregation Disorders.
Denervation-Induced Activation of the Ubiquitin-Proteasome System Reduces Skeletal Muscle Quantity Not Quality.
A selective inhibitor of the immunoproteasome subunit LMP2 induces apoptosis in PC-3 cells and suppresses tumour growth in nude mice.
Persistent hijacking of brain proteasomes in HIV-associated dementia.
Reduced ubiquitin C-terminal hydrolase-1 expression levels in dementia with Lewy bodies.
Resveratrol promotes clearance of Alzheimer's disease amyloid-beta peptides.
Kim S, Park SH, Choi WH, Lee MJ
Immune network 2022 Jun;22(3):e28
Immune network 2022 Jun;22(3):e28
A protein-protein interaction map reveals that the Coxiella burnetii effector CirB inhibits host proteasome activity.
Fu M, Liu Y, Wang G, Wang P, Zhang J, Chen C, Zhao M, Zhang S, Jiao J, Ouyang X, Yu Y, Wen B, He C, Wang J, Zhou D, Xiong X
PLoS pathogens 2022 Jul;18(7):e1010660
PLoS pathogens 2022 Jul;18(7):e1010660
H727 cells are inherently resistant to the proteasome inhibitor carfilzomib, yet require proteasome activity for cell survival and growth.
Lee MJ, Miller Z, Park JE, Bhattarai D, Lee W, Kim KB
Scientific reports 2019 Mar 11;9(1):4089
Scientific reports 2019 Mar 11;9(1):4089
Skeletal muscle denervation investigations: selecting an experimental control wisely.
Liu H, Thompson LV
American journal of physiology. Cell physiology 2019 Mar 1;316(3):C456-C461
American journal of physiology. Cell physiology 2019 Mar 1;316(3):C456-C461
Novel Potent Capsid Assembly Modulators Regulate Multiple Steps of the Hepatitis B Virus Life Cycle.
Lahlali T, Berke JM, Vergauwen K, Foca A, Vandyck K, Pauwels F, Zoulim F, Durantel D
Antimicrobial agents and chemotherapy 2018 Oct;62(10)
Antimicrobial agents and chemotherapy 2018 Oct;62(10)
Induction of the Immunoproteasome Subunit Lmp7 Links Proteostasis and Immunity in α-Synuclein Aggregation Disorders.
Ugras S, Daniels MJ, Fazelinia H, Gould NS, Yocum AK, Luk KC, Luna E, Ding H, McKennan C, Seeholzer S, Martinez D, Evans P, Brown D, Duda JE, Ischiropoulos H
EBioMedicine 2018 May;31:307-319
EBioMedicine 2018 May;31:307-319
Denervation-Induced Activation of the Ubiquitin-Proteasome System Reduces Skeletal Muscle Quantity Not Quality.
Baumann CW, Liu HM, Thompson LV
PloS one 2016;11(8):e0160839
PloS one 2016;11(8):e0160839
A selective inhibitor of the immunoproteasome subunit LMP2 induces apoptosis in PC-3 cells and suppresses tumour growth in nude mice.
Wehenkel M, Ban JO, Ho YK, Carmony KC, Hong JT, Kim KB
British journal of cancer 2012 Jun 26;107(1):53-62
British journal of cancer 2012 Jun 26;107(1):53-62
Persistent hijacking of brain proteasomes in HIV-associated dementia.
Nguyen TP, Soukup VM, Gelman BB
The American journal of pathology 2010 Feb;176(2):893-902
The American journal of pathology 2010 Feb;176(2):893-902
Reduced ubiquitin C-terminal hydrolase-1 expression levels in dementia with Lewy bodies.
Barrachina M, Castaño E, Dalfó E, Maes T, Buesa C, Ferrer I
Neurobiology of disease 2006 May;22(2):265-73
Neurobiology of disease 2006 May;22(2):265-73
Resveratrol promotes clearance of Alzheimer's disease amyloid-beta peptides.
Marambaud P, Zhao H, Davies P
The Journal of biological chemistry 2005 Nov 11;280(45):37377-82
The Journal of biological chemistry 2005 Nov 11;280(45):37377-82
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
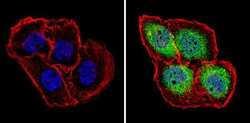
- Experimental details
- Immunofluorescent analysis of Proteasome 20S X (green) showing staining in the cytoplasm and nucleus of A431 cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a Proteasome 20S X polyclonal antibody (Product # PA1-977) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
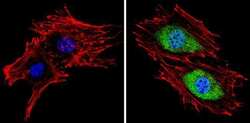
- Experimental details
- Immunofluorescent analysis of Proteasome 20S X (green) showing staining in the cytoplasm and nucleus of BAEC cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a Proteasome 20S X polyclonal antibody (Product # PA1-977) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
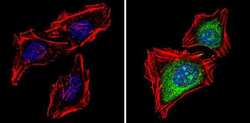
- Experimental details
- Immunofluorescent analysis of Proteasome 20S X (green) showing staining in the cytoplasm and nucleus of Hela cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a Proteasome 20S X polyclonal antibody (Product # PA1-977) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
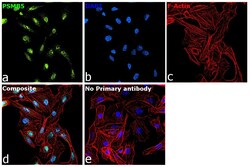
- Experimental details
- Immunofluorescence analysis of PSMB5 was performed using 70% confluent log phase DU 145 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with PSMB5 Polyclonal Antibody (Product # PA1-977) at 1:100 dilution in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32790), (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b:Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing nuclear localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
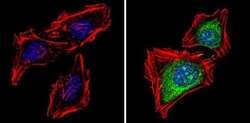
- Experimental details
- Immunofluorescent analysis of Proteasome 20S X (green) showing staining in the cytoplasm and nucleus of Hela cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a Proteasome 20S X polyclonal antibody (Product # PA1-977) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
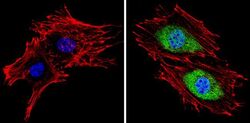
- Experimental details
- Immunofluorescent analysis of Proteasome 20S X (green) showing staining in the cytoplasm and nucleus of BAEC cells (right) compared to a negative control without primary antibody (left). Formalin-fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 5-10 minutes and blocked with 3% BSA-PBS for 30 minutes at room temperature. Cells were probed with a Proteasome 20S X polyclonal antibody (Product # PA1-977) in 3% BSA-PBS at a dilution of 1:100 and incubated overnight at 4 ºC in a humidified chamber. Cells were washed with PBST and incubated with a DyLight-conjugated secondary antibody in PBS at room temperature in the dark. F-actin (red) was stained with a fluorescent red phalloidin and nuclei (blue) were stained with Hoechst or DAPI. Images were taken at a magnification of 60x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
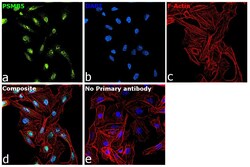
- Experimental details
- Immunofluorescence analysis of PSMB5 was performed using 70% confluent log phase DU 145 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 2% BSA for 45 minutes at room temperature. The cells were labeled with PSMB5 Polyclonal Antibody (Product # PA1-977) at 1:100 dilution in 0.1% BSA, incubated at 4 degree celsius overnight and then labeled with Donkey anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor Plus 488 (Product # A32790), (1:2000 dilution), for 45 minutes at room temperature (Panel a: Green). Nuclei (Panel b:Blue) were stained with ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962). F-actin (Panel c: Red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing nuclear localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
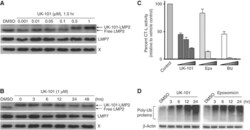
- Experimental details
- Figure 3 UK-101 selectively inhibits LMP2 and its catalytic activity via a stable covalent modification. ( A ) Following a 90 min incubation of cells with UK-101, the concentration dependence of UK-101 binding to LMP2 in PC-3 cells was examined via western blotting. The increase in apparent molecular weight of LMP2 indicates its covalent modification by UK-101. No mobility changes in the X subunit of the constitutive proteasome or the LMP7 subunit of the immunoproteasome were observed. ( B ) The stability of the covalent modification of LMP2 with UK-101 was examined following removal of excess-free UK-101 from the system. PC-3 cells were treated with drug or vehicle for 90 min, then washed and given fresh media. Samples were collected at the indicated times and subjected to western blotting. The binding of UK-101 to LMP2 is maintained for at least 48 h. ( C ) Inhibition of the chymotrypsin-like (CT-L) activity of cellular proteasomes. A proteasome activity assay was performed with PC-3 cells treated with increasing concentrations of proteasome inhibitors (UK-101: 1, 10, 50 mu M; epoxomicin or bortezomib: 5, 50, 500 nM) for 2 h. The CT-L activity of total cellular proteasomes was measured using the CT-L luminogenic substrate Suc-Leu-Leu-Val-Tyr-aminoluciferin. Values were obtained in triplicate, and error bars represent standard deviations. ( D ) Cells were treated with inhibitors, lysed, and probed for ubiquitin. Both UK-101 (25 mu M) and epoxomicin (1 mu M) caused an accumul
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
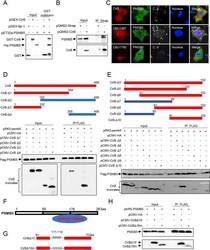
- Experimental details
- 10.1371/journal.ppat.1010660.g005 Fig 5 CirB interacts with the host protein PSMB5. (A) pGEX-CirB, pGEX-GST or pET32a-PSMB5 was separately expressed in E . coli BL21. The purified GST-CirB or GST was co-incubated with GST Sepharose beads, then HIS-PSMB5 was subjected to the GST-conjugated beads. After eluting, samples were loaded for Western blotting. (B) HEK-293T cells were transfected with pQM02-CirB or the corresponding control plasmid. Then, the cells were lysed and proteins were immunoprecipitated with an anti-Strep antibody, and the intracellular PSMB5 was immunoblotted with an anti-PSMB5 antibody. (C) HeLa cells were transfected with mCherry-tagged CirB, CBU1780 or CBU1387 before C . burnetii infection at an MOI of 100, respectively. Two days later, cells were fixed, and endogenous PSMB5 was probed with an anti-PSMB5 and a corresponding goat anti-rabbit Alexa Fluor 488 antibody (green). C . burnetii was stained with anti- C . burnetii serum, followed by a Cy5 conjugated goat anti-mouse IgG H&L antibody (white). The nucleus was stained with DAPI (blue). Images were captured using a confocal microscope (magnification, x600, bar = 20 mum). (D-E) FLAG-PSMB5 and HA-tagged CirB truncation mutants were co-transfected into 293T cells, and the cell lysates were immunoprecipitated with an anti-FLAG antibody and immunoblotted with an anti-HA or anti-FLAG antibody. Schematic diagram of the truncation mutants of CirB is shown in the upper panel. Sections marked in red represent the
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2. Proteasome properties in gastrocnemius muscles from internal controls (7d Ctrl, 14d Ctrl, and Sham Ctrl) and external control (Ex Ctrl). Data points represent fold change of Ex Ctrl from individual mice. D-F : representative Western blots for each corresponding proteasome subunit. Images of representative bands in each panel were from the same blot; dashed lines indicate rearrangement of the noncontinuous lanes. Values are means +- SE; n = 5, 7, 8, and 8 for alpha 7 and n = 5, 5, 6, and 6 for the other proteasome properties in Ex Ctrl, 7d Ctrl, 14d Ctrl, and Sham Ctrl, respectively * P < 0.05 vs. Ex Ctrl.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
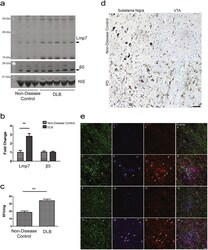
- Experimental details
- Fig. 4 The Immunoproteasome in Human Disease Driven by alpha-Synuclein Aggregation. ( a ) Representative western blot analysis in human DLB and non-disease control brains for Lmp7 and the proteasome protein beta5. ( b ) Densitometric analysis of western blots normalized to the levels of NSE protein indicate a significant increase (**p < 0.01, t -test) in Lmp7 levels in DLB brains (n = 5) compared with controls, (n = 3). ( c ) Concomitant with the increase in Lmp7 protein levels the chymotrypsin-like activity, a proxy for immunoproteasome activity, is increased by 189% in DLB brain compared with control brain (**p < 0.01, n = 3, t-test). ( d ) Immunohistochemical staining revealed increased immunoreactivity for Lmp7 in PD substantia nigra and VTA. ( d 1 ) Representative staining for Lmp7b in control substantia nigra and ( d 2 ) VTA. Increased Lmp7 staining in both neurons (asterisks) and glia (arrows) in PD substantia nigra ( d 3 ) and VTA ( d 4 ). Nuclei were counterstained with hematoxylin (blue). Scale bar = 50 mum. ( e ) Co-localization of Lmp7 with glial markers. Representative fluorescent staining for the astrocyte marker, GFAP; microglial marker, IBA1; and Lmp7. Overlay of GFAP, IBA1, LMP7 and Hoechst nuclear stain is displayed in the right column. Staining in non-disease control substantia nigra ( e 1-4 ) and in VTA ( e 5-8 ). Increased staining for Lmp7 in the PD substantia nigra ( e 9-12 ) and in the VTA ( e 12-16 ). Lmp7 co-localized with both astrocytes and microg
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
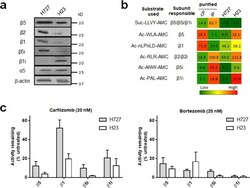
- Experimental details
- Figure 3 ( a ) Immunoblots showing the expression of cP and iP catalytic subunits in H727 and H23 cells. Immunoblots for additional cell lines displaying differential expression of proteasome catalytic subunits are presented in Supplementary Fig. S2 . ( b ) Heat map displaying differential proteasome activity profiles in H727 and H23 cell lines. Purified human 20S cP and iP were used as controls for individual subunits: 20S cP for beta5 and beta1 and 20S iP for beta5i and beta1i. The numbers represent hydrolysis rates of respective substrates (RFU/min, mean values derived from three technical replicates) and were converted to color format and clustered by using the program R ( http://www.R-project.org ). ( c ) Remaining catalytic activities of individual proteasome subunits in H727 and H23 cells 4 h after treatment with 20 nM of carfilzomib (left panel) or 20 nM bortezomib (right panel). Data are presented as mean +- SD derived from three technical replicates.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
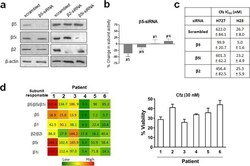
- Experimental details
- Figure 5 ( a ) Immunoblots of proteasome catalytic subunits in H727 cells transfected with siRNA targeting beta5, beta2 or beta5i. ( b ) The catalytic activity of beta5 subunit was decreased in H727 cells transfected with siRNA targeting beta5 compared with H727 cells transfected with scrambled siRNA. ( c ) Effects of siRNA knockdown of beta5, beta5i, or beta2 on Cfz sensitivity (IC 50 values) in H23 and H727 cells. Data are shown as mean +- SD derived from a single non-linear regression based on n = 3 replicates per compound per concentration. ( d ) Heat map showing proteasome catalytic subunit activity profiles of 6 PI-naive patient MM samples purchased from Conversant Biologics and AllCells (left). The numbers represent hydrolysis rates of respective substrates (RFU/min, mean values derived from three technical replicates) and were converted to color format and clustered by using the program R ( http://www.R-project.org ) (left panel). Carfilzomib (Cfz) cell viability of the same 6 patient MM cells was measured via CellTiter-Glo Luminescent Cell Viability Assay (right panel).
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry