Antibody data
- Antibody Data
- Antigen structure
- References [14]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 12-2239-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD223 (LAG-3) Monoclonal Antibody (3DS223H), PE, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: This 3DS223H monoclonal antibody recognizes human CD223 also known as Lymphocyte Activation Gene 3 (LAG-3). LAG-3 is a 70-kDa surface glycoprotein belonging to the Ig superfamily with homology to CD4. LAG-3 binds to MHC class II with higher affinity than CD4 and is thought to be involved in the negative regulation of T cell activation and homeostatic proliferation. Surface expression of LAG-3 has been reported on activated T cells (including regulatory T cells) and NK cells. CD8+ T cells usually express LAG-3 at significantly higher levels than CD4+ T cells. Coexpression of LAG-3 and CD49b has been proposed to identify human and mouse Type 1 regulatory T cells (Tr1 cells). This 3DS223H antibody will recognize a formaldehyde-fixed epitope. Applications Reported: This 3DS223H antibody has been reported for use in flow cytometric analysis. Applications Tested: This 3DS223H antibody has been pre-titrated and tested by flow cytometric analysis of stimulated normal human peripheral blood cells. This can be used at 5 µL (0.06 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488-561 nm; Emission: 578 nm; Laser: Blue Laser, Green Laser, Yellow-Green Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Yellow dye
- Isotype
- IgG
- Antibody clone number
- 3DS223H
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Neoadjuvant chemotherapy plus nivolumab with or without ipilimumab in operable non-small cell lung cancer: the phase 2 platform NEOSTAR trial.
GPC2-CAR T cells tuned for low antigen density mediate potent activity against neuroblastoma without toxicity.
CAR T cells targeting tumor-associated exons of glypican 2 regress neuroblastoma in mice.
Humans with inherited T cell CD28 deficiency are susceptible to skin papillomaviruses but are otherwise healthy.
Neoadjuvant Chemotherapy Increases Cytotoxic T Cell, Tissue Resident Memory T Cell, and B Cell Infiltration in Resectable NSCLC.
Immunogenic Chemotherapy Enhances Recruitment of CAR-T Cells to Lung Tumors and Improves Antitumor Efficacy when Combined with Checkpoint Blockade.
Combined p14ARF and Interferon-β Gene Transfer to the Human Melanoma Cell Line SK-MEL-147 Promotes Oncolysis and Immune Activation.
Circulating Exosomes Inhibit B Cell Proliferation and Activity.
MAVS Genetic Variation Is Associated with Decreased HIV-1 Replication In Vitro and Reduced CD4(+) T Cell Infection in HIV-1-Infected Individuals.
CAR T Cells Targeting B7-H3, a Pan-Cancer Antigen, Demonstrate Potent Preclinical Activity Against Pediatric Solid Tumors and Brain Tumors.
YY1 Upregulates Checkpoint Receptors and Downregulates Type I Cytokines in Exhausted, Chronically Stimulated Human T Cells.
PD-1 Blockade Promotes Emerging Checkpoint Inhibitors in Enhancing T Cell Responses to Allogeneic Dendritic Cells.
OMIP-037: 16-color panel to measure inhibitory receptor signatures from multiple human immune cell subsets.
Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection.
Cascone T, Leung CH, Weissferdt A, Pataer A, Carter BW, Godoy MCB, Feldman H, William WN Jr, Xi Y, Basu S, Sun JJ, Yadav SS, Rojas Alvarez FR, Lee Y, Mishra AK, Chen L, Pradhan M, Guo H, Sinjab A, Zhou N, Negrao MV, Le X, Gay CM, Tsao AS, Byers LA, Altan M, Glisson BS, Fossella FV, Elamin YY, Blumenschein G Jr, Zhang J, Skoulidis F, Wu J, Mehran RJ, Rice DC, Walsh GL, Hofstetter WL, Rajaram R, Antonoff MB, Fujimoto J, Solis LM, Parra ER, Haymaker C, Wistuba II, Swisher SG, Vaporciyan AA, Lin HY, Wang J, Gibbons DL, Jack Lee J, Ajami NJ, Wargo JA, Allison JP, Sharma P, Kadara H, Heymach JV, Sepesi B
Nature medicine 2023 Mar;29(3):593-604
Nature medicine 2023 Mar;29(3):593-604
GPC2-CAR T cells tuned for low antigen density mediate potent activity against neuroblastoma without toxicity.
Heitzeneder S, Bosse KR, Zhu Z, Zhelev D, Majzner RG, Radosevich MT, Dhingra S, Sotillo E, Buongervino S, Pascual-Pasto G, Garrigan E, Xu P, Huang J, Salzer B, Delaidelli A, Raman S, Cui H, Martinez B, Bornheimer SJ, Sahaf B, Alag A, Fetahu IS, Hasselblatt M, Parker KR, Anbunathan H, Hwang J, Huang M, Sakamoto K, Lacayo NJ, Klysz DD, Theruvath J, Vilches-Moure JG, Satpathy AT, Chang HY, Lehner M, Taschner-Mandl S, Julien JP, Sorensen PH, Dimitrov DS, Maris JM, Mackall CL
Cancer cell 2022 Jan 10;40(1):53-69.e9
Cancer cell 2022 Jan 10;40(1):53-69.e9
CAR T cells targeting tumor-associated exons of glypican 2 regress neuroblastoma in mice.
Li N, Torres MB, Spetz MR, Wang R, Peng L, Tian M, Dower CM, Nguyen R, Sun M, Tai CH, de Val N, Cachau R, Wu X, Hewitt SM, Kaplan RN, Khan J, St Croix B, Thiele CJ, Ho M
Cell reports. Medicine 2021 Jun 15;2(6):100297
Cell reports. Medicine 2021 Jun 15;2(6):100297
Humans with inherited T cell CD28 deficiency are susceptible to skin papillomaviruses but are otherwise healthy.
Béziat V, Rapaport F, Hu J, Titeux M, Bonnet des Claustres M, Bourgey M, Griffin H, Bandet É, Ma CS, Sherkat R, Rokni-Zadeh H, Louis DM, Changi-Ashtiani M, Delmonte OM, Fukushima T, Habib T, Guennoun A, Khan T, Bender N, Rahman M, About F, Yang R, Rao G, Rouzaud C, Li J, Shearer D, Balogh K, Al Ali F, Ata M, Dabiri S, Momenilandi M, Nammour J, Alyanakian MA, Leruez-Ville M, Guenat D, Materna M, Marcot L, Vladikine N, Soret C, Vahidnezhad H, Youssefian L, Saeidian AH, Uitto J, Catherinot É, Navabi SS, Zarhrate M, Woodley DT, Jeljeli M, Abraham T, Belkaya S, Lorenzo L, Rosain J, Bayat M, Lanternier F, Lortholary O, Zakavi F, Gros P, Orth G, Abel L, Prétet JL, Fraitag S, Jouanguy E, Davis MM, Tangye SG, Notarangelo LD, Marr N, Waterboer T, Langlais D, Doorbar J, Hovnanian A, Christensen N, Bossuyt X, Shahrooei M, Casanova JL
Cell 2021 Jul 8;184(14):3812-3828.e30
Cell 2021 Jul 8;184(14):3812-3828.e30
Neoadjuvant Chemotherapy Increases Cytotoxic T Cell, Tissue Resident Memory T Cell, and B Cell Infiltration in Resectable NSCLC.
Gaudreau PO, Negrao MV, Mitchell KG, Reuben A, Corsini EM, Li J, Karpinets TV, Wang Q, Diao L, Wang J, Federico L, Parra-Cuentas ER, Khairullah R, Behrens C, Correa AM, Gomez D, Little L, Gumbs C, Kadara HN, Fujimoto J, McGrail DJ, Vaporciyan AA, Swisher SG, Walsh G, Antonoff MB, Weissferdt A, Tran H, Roarty E, Haymaker C, Bernatchez C, Zhang J, Futreal PA, Wistuba II, Cascone T, Heymach JV, Sepesi B, Zhang J, Gibbons DL
Journal of thoracic oncology : official publication of the International Association for the Study of Lung Cancer 2021 Jan;16(1):127-139
Journal of thoracic oncology : official publication of the International Association for the Study of Lung Cancer 2021 Jan;16(1):127-139
Immunogenic Chemotherapy Enhances Recruitment of CAR-T Cells to Lung Tumors and Improves Antitumor Efficacy when Combined with Checkpoint Blockade.
Srivastava S, Furlan SN, Jaeger-Ruckstuhl CA, Sarvothama M, Berger C, Smythe KS, Garrison SM, Specht JM, Lee SM, Amezquita RA, Voillet V, Muhunthan V, Yechan-Gunja S, Pillai SPS, Rader C, Houghton AM, Pierce RH, Gottardo R, Maloney DG, Riddell SR
Cancer cell 2021 Feb 8;39(2):193-208.e10
Cancer cell 2021 Feb 8;39(2):193-208.e10
Combined p14ARF and Interferon-β Gene Transfer to the Human Melanoma Cell Line SK-MEL-147 Promotes Oncolysis and Immune Activation.
Cerqueira OLD, Clavijo-Salomon MA, Cardoso EC, Citrangulo Tortelli Junior T, Mendonça SA, Barbuto JAM, Strauss BE
Frontiers in immunology 2020;11:576658
Frontiers in immunology 2020;11:576658
Circulating Exosomes Inhibit B Cell Proliferation and Activity.
Schroeder JC, Puntigam L, Hofmann L, Jeske SS, Beccard IJ, Doescher J, Laban S, Hoffmann TK, Brunner C, Theodoraki MN, Schuler PJ
Cancers 2020 Jul 29;12(8)
Cancers 2020 Jul 29;12(8)
MAVS Genetic Variation Is Associated with Decreased HIV-1 Replication In Vitro and Reduced CD4(+) T Cell Infection in HIV-1-Infected Individuals.
Stunnenberg M, van Pul L, Sprokholt JK, van Dort KA, Gringhuis SI, Geijtenbeek TBH, Kootstra NA
Viruses 2020 Jul 16;12(7)
Viruses 2020 Jul 16;12(7)
CAR T Cells Targeting B7-H3, a Pan-Cancer Antigen, Demonstrate Potent Preclinical Activity Against Pediatric Solid Tumors and Brain Tumors.
Majzner RG, Theruvath JL, Nellan A, Heitzeneder S, Cui Y, Mount CW, Rietberg SP, Linde MH, Xu P, Rota C, Sotillo E, Labanieh L, Lee DW, Orentas RJ, Dimitrov DS, Zhu Z, Croix BS, Delaidelli A, Sekunova A, Bonvini E, Mitra SS, Quezado MM, Majeti R, Monje M, Sorensen PHB, Maris JM, Mackall CL
Clinical cancer research : an official journal of the American Association for Cancer Research 2019 Apr 15;25(8):2560-2574
Clinical cancer research : an official journal of the American Association for Cancer Research 2019 Apr 15;25(8):2560-2574
YY1 Upregulates Checkpoint Receptors and Downregulates Type I Cytokines in Exhausted, Chronically Stimulated Human T Cells.
Balkhi MY, Wittmann G, Xiong F, Junghans RP
iScience 2018 Apr 27;2:105-122
iScience 2018 Apr 27;2:105-122
PD-1 Blockade Promotes Emerging Checkpoint Inhibitors in Enhancing T Cell Responses to Allogeneic Dendritic Cells.
Stecher C, Battin C, Leitner J, Zettl M, Grabmeier-Pfistershammer K, Höller C, Zlabinger GJ, Steinberger P
Frontiers in immunology 2017;8:572
Frontiers in immunology 2017;8:572
OMIP-037: 16-color panel to measure inhibitory receptor signatures from multiple human immune cell subsets.
Belkina AC, Snyder-Cappione JE
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2017 Feb;91(2):175-179
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2017 Feb;91(2):175-179
Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection.
Miles B, Miller SM, Folkvord JM, Kimball A, Chamanian M, Meditz AL, Arends T, McCarter MD, Levy DN, Rakasz EG, Skinner PJ, Connick E
Nature communications 2015 Oct 20;6:8608
Nature communications 2015 Oct 20;6:8608
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
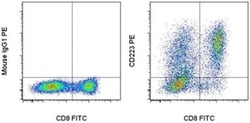
- Experimental details
- Normal human peripheral blood cells were stimulated for 3 days with Human IL-2 Recombinant Protein (Product # 14-8029-81), Anti-Human CD3, and Anti-Human CD28 Functional Grade Purifieds (Product # 16-0037-81 and Product # 16-0289-81). These cells were then surface stained with Anti-Human CD8a FITC (Product # 11-0086-42) and Mouse IgG1 K Isotype Control PE (Product # 12-4714-81) (left) or Anti-Human CD223 (LAG-3) PE (right). Cells in the lymphocyte gate were used for analysis.
- Conjugate
- Yellow dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Yellow dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
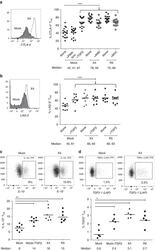
- Experimental details
- Figure 6 T FR exhibit an enhanced regulatory phenotype in ex vivo HIV infection. Tonsil cells were mock-, X4-, or R5-spinoculated and cultured under experimental conditions as indicated. T FR were then analysed for expression of regulatory receptors and cytokine production by intracellular cytokine staining. ( a ) Percentage of total (surface and intracellular) T FR CTLA-4 expression ( n =15). ( b ) Percentage of surface T FR LAG-3 expression ( n =8). ( c ) Production of IL-10 by T FR ( n =7). ( d ) Production of TGF-beta-1 (measured as LAP) by T FR ( n =5). The horizontal bars of each graph indicate the median value and are listed where appropriate for clarity. Statistical analyses were performed by Friedman ( a , b ) or Mann-Whitney tests ( c , d ) and significance is denoted by asterisks where * P
- Conjugate
- Yellow dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry