Antibody data
- Antibody Data
- Antigen structure
- References [18]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [4]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 17-2239-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD223 (LAG-3) Monoclonal Antibody (3DS223H), APC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: This 3DS223H monoclonal antibody recognizes human CD223 also known as Lymphocyte Activation Gene 3 (LAG-3). LAG-3 is a 70-kDa surface glycoprotein belonging to the Ig superfamily with homology to CD4. LAG-3 binds to MHC class II with higher affinity than CD4 and is thought to be involved in the negative regulation of T cell activation and homeostatic proliferation. Surface expression of LAG-3 has been reported on activated T cells (including regulatory T cells) and NK cells. CD8+ T cells usually express LAG-3 at significantly higher levels than CD4+ T cells. Coexpression of LAG-3 and CD49b has been proposed to identify human and mouse Type 1 regulatory T cells (Tr1 cells). This 3DS223H antibody will recognize a formaldehyde-fixed epitope. Applications Reported: This 3DS223H antibody has been reported for use in flow cytometric analysis. Applications Tested: This 3DS223H antibody has been pre-titrated and tested by flow cytometric analysis of stimulated normal human peripheral blood cells. This can be used at 5 µL (0.06 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 633-647 nm; Emission: 660 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 3DS223H
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Bispecific T-Cell Engaging Antibodies Against MUC16 Demonstrate Efficacy Against Ovarian Cancer in Monotherapy and in Combination With PD-1 and VEGF Inhibition.
Increased Intrahepatic Expression of Immune Checkpoint Molecules in Autoimmune Liver Disease.
Systematically optimized BCMA/CS1 bispecific CAR-T cells robustly control heterogeneous multiple myeloma.
MAVS Genetic Variation Is Associated with Decreased HIV-1 Replication In Vitro and Reduced CD4(+) T Cell Infection in HIV-1-Infected Individuals.
Symmetric Arginine Dimethylation Is Selectively Required for mRNA Splicing and the Initiation of Type I and Type III Interferon Signaling.
PD-1 Blockade Reinvigorates Bone Marrow CD8(+) T Cells from Patients with Multiple Myeloma in the Presence of TGFβ Inhibitors.
Differential expression pattern of co-inhibitory molecules on CD4(+) T cells in uncomplicated versus complicated malaria.
Primary T Cells from Cutaneous T-cell Lymphoma Skin Explants Display an Exhausted Immune Checkpoint Profile.
Low-dose interleukin-2 promotes STAT-5 phosphorylation, T(reg) survival and CTLA-4-dependent function in autoimmune liver diseases.
OMIP-037: 16-color panel to measure inhibitory receptor signatures from multiple human immune cell subsets.
Regulatory T Cell Modulation by CBP/EP300 Bromodomain Inhibition.
Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection.
4-1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors.
Progression of Lung Cancer Is Associated with Increased Dysfunction of T Cells Defined by Coexpression of Multiple Inhibitory Receptors.
Coexpression of CD49b and LAG-3 identifies human and mouse T regulatory type 1 cells.
LAG-3 expression defines a subset of CD4(+)CD25(high)Foxp3(+) regulatory T cells that are expanded at tumor sites.
Identification of a human CD8+ regulatory T cell subset that mediates suppression through the chemokine CC chemokine ligand 4.
The negative regulatory function of the lymphocyte-activation gene-3 co-receptor (CD223) on human T cells.
Yeku OO, Rao TD, Laster I, Kononenko A, Purdon TJ, Wang P, Cui Z, Liu H, Brentjens RJ, Spriggs D
Frontiers in immunology 2021;12:663379
Frontiers in immunology 2021;12:663379
Increased Intrahepatic Expression of Immune Checkpoint Molecules in Autoimmune Liver Disease.
Macek Jilkova Z, Hilleret MN, Gerster T, Sturm N, Mercey-Ressejac M, Zarski JP, Leroy V, Marche PN, Costentin C, Decaens T
Cells 2021 Oct 6;10(10)
Cells 2021 Oct 6;10(10)
Systematically optimized BCMA/CS1 bispecific CAR-T cells robustly control heterogeneous multiple myeloma.
Zah E, Nam E, Bhuvan V, Tran U, Ji BY, Gosliner SB, Wang X, Brown CE, Chen YY
Nature communications 2020 May 8;11(1):2283
Nature communications 2020 May 8;11(1):2283
MAVS Genetic Variation Is Associated with Decreased HIV-1 Replication In Vitro and Reduced CD4(+) T Cell Infection in HIV-1-Infected Individuals.
Stunnenberg M, van Pul L, Sprokholt JK, van Dort KA, Gringhuis SI, Geijtenbeek TBH, Kootstra NA
Viruses 2020 Jul 16;12(7)
Viruses 2020 Jul 16;12(7)
Symmetric Arginine Dimethylation Is Selectively Required for mRNA Splicing and the Initiation of Type I and Type III Interferon Signaling.
Metz PJ, Ching KA, Xie T, Delgado Cuenca P, Niessen S, Tatlock JH, Jensen-Pergakes K, Murray BW
Cell reports 2020 Feb 11;30(6):1935-1950.e8
Cell reports 2020 Feb 11;30(6):1935-1950.e8
PD-1 Blockade Reinvigorates Bone Marrow CD8(+) T Cells from Patients with Multiple Myeloma in the Presence of TGFβ Inhibitors.
Kwon M, Kim CG, Lee H, Cho H, Kim Y, Lee EC, Choi SJ, Park J, Seo IH, Bogen B, Song IC, Jo DY, Kim JS, Park SH, Choi I, Choi YS, Shin EC
Clinical cancer research : an official journal of the American Association for Cancer Research 2020 Apr 1;26(7):1644-1655
Clinical cancer research : an official journal of the American Association for Cancer Research 2020 Apr 1;26(7):1644-1655
Differential expression pattern of co-inhibitory molecules on CD4(+) T cells in uncomplicated versus complicated malaria.
Abel A, Steeg C, Aminkiah F, Addai-Mensah O, Addo M, Gagliani N, Casar C, Yar DD, Owusu-Dabo E, Jacobs T, Mackroth MS
Scientific reports 2018 Mar 19;8(1):4789
Scientific reports 2018 Mar 19;8(1):4789
Primary T Cells from Cutaneous T-cell Lymphoma Skin Explants Display an Exhausted Immune Checkpoint Profile.
Querfeld C, Leung S, Myskowski PL, Curran SA, Goldman DA, Heller G, Wu X, Kil SH, Sharma S, Finn KJ, Horwitz S, Moskowitz A, Mehrara B, Rosen ST, Halpern AC, Young JW
Cancer immunology research 2018 Aug;6(8):900-909
Cancer immunology research 2018 Aug;6(8):900-909
Low-dose interleukin-2 promotes STAT-5 phosphorylation, T(reg) survival and CTLA-4-dependent function in autoimmune liver diseases.
Jeffery HC, Jeffery LE, Lutz P, Corrigan M, Webb GJ, Hirschfield GM, Adams DH, Oo YH
Clinical and experimental immunology 2017 Jun;188(3):394-411
Clinical and experimental immunology 2017 Jun;188(3):394-411
OMIP-037: 16-color panel to measure inhibitory receptor signatures from multiple human immune cell subsets.
Belkina AC, Snyder-Cappione JE
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2017 Feb;91(2):175-179
Cytometry. Part A : the journal of the International Society for Analytical Cytology 2017 Feb;91(2):175-179
Regulatory T Cell Modulation by CBP/EP300 Bromodomain Inhibition.
Ghosh S, Taylor A, Chin M, Huang HR, Conery AR, Mertz JA, Salmeron A, Dakle PJ, Mele D, Cote A, Jayaram H, Setser JW, Poy F, Hatzivassiliou G, DeAlmeida-Nagata D, Sandy P, Hatton C, Romero FA, Chiang E, Reimer T, Crawford T, Pardo E, Watson VG, Tsui V, Cochran AG, Zawadzke L, Harmange JC, Audia JE, Bryant BM, Cummings RT, Magnuson SR, Grogan JL, Bellon SF, Albrecht BK, Sims RJ 3rd, Lora JM
The Journal of biological chemistry 2016 Jun 17;291(25):13014-27
The Journal of biological chemistry 2016 Jun 17;291(25):13014-27
Follicular regulatory T cells impair follicular T helper cells in HIV and SIV infection.
Miles B, Miller SM, Folkvord JM, Kimball A, Chamanian M, Meditz AL, Arends T, McCarter MD, Levy DN, Rakasz EG, Skinner PJ, Connick E
Nature communications 2015 Oct 20;6:8608
Nature communications 2015 Oct 20;6:8608
4-1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors.
Long AH, Haso WM, Shern JF, Wanhainen KM, Murgai M, Ingaramo M, Smith JP, Walker AJ, Kohler ME, Venkateshwara VR, Kaplan RN, Patterson GH, Fry TJ, Orentas RJ, Mackall CL
Nature medicine 2015 Jun;21(6):581-90
Nature medicine 2015 Jun;21(6):581-90
Progression of Lung Cancer Is Associated with Increased Dysfunction of T Cells Defined by Coexpression of Multiple Inhibitory Receptors.
Thommen DS, Schreiner J, Müller P, Herzig P, Roller A, Belousov A, Umana P, Pisa P, Klein C, Bacac M, Fischer OS, Moersig W, Savic Prince S, Levitsky V, Karanikas V, Lardinois D, Zippelius A
Cancer immunology research 2015 Dec;3(12):1344-55
Cancer immunology research 2015 Dec;3(12):1344-55
Coexpression of CD49b and LAG-3 identifies human and mouse T regulatory type 1 cells.
Gagliani N, Magnani CF, Huber S, Gianolini ME, Pala M, Licona-Limon P, Guo B, Herbert DR, Bulfone A, Trentini F, Di Serio C, Bacchetta R, Andreani M, Brockmann L, Gregori S, Flavell RA, Roncarolo MG
Nature medicine 2013 Jun;19(6):739-46
Nature medicine 2013 Jun;19(6):739-46
LAG-3 expression defines a subset of CD4(+)CD25(high)Foxp3(+) regulatory T cells that are expanded at tumor sites.
Camisaschi C, Casati C, Rini F, Perego M, De Filippo A, Triebel F, Parmiani G, Belli F, Rivoltini L, Castelli C
Journal of immunology (Baltimore, Md. : 1950) 2010 Jun 1;184(11):6545-51
Journal of immunology (Baltimore, Md. : 1950) 2010 Jun 1;184(11):6545-51
Identification of a human CD8+ regulatory T cell subset that mediates suppression through the chemokine CC chemokine ligand 4.
Joosten SA, van Meijgaarden KE, Savage ND, de Boer T, Triebel F, van der Wal A, de Heer E, Klein MR, Geluk A, Ottenhoff TH
Proceedings of the National Academy of Sciences of the United States of America 2007 May 8;104(19):8029-34
Proceedings of the National Academy of Sciences of the United States of America 2007 May 8;104(19):8029-34
The negative regulatory function of the lymphocyte-activation gene-3 co-receptor (CD223) on human T cells.
Maçon-Lemaître L, Triebel F
Immunology 2005 Jun;115(2):170-8
Immunology 2005 Jun;115(2):170-8
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
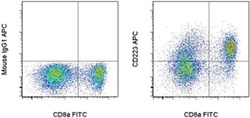
- Experimental details
- Normal human peripheral blood cells were stimulated for 3 days with Human IL-2 Recombinant Protein (Product # 14-8029-81), Anti-Human CD3, and Anti-Human CD28 Functional Grade Purifieds (Product # 16-0037-81 and Product # 16-0289-81). These cells were then surface stained with Anti-Human CD8a FITC (Product # 11-0087-42) and Mouse IgG1 K Isotype Control APC (Product # 17-4714-81) (left) or Anti-Human CD223 (LAG-3) APC (right). Viable cells, as determined by Fixable Viability Dye eFluor® 450 (Product # 65-0863-14), in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
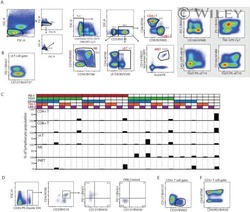
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 6 T FR exhibit an enhanced regulatory phenotype in ex vivo HIV infection. Tonsil cells were mock-, X4-, or R5-spinoculated and cultured under experimental conditions as indicated. T FR were then analysed for expression of regulatory receptors and cytokine production by intracellular cytokine staining. ( a ) Percentage of total (surface and intracellular) T FR CTLA-4 expression ( n =15). ( b ) Percentage of surface T FR LAG-3 expression ( n =8). ( c ) Production of IL-10 by T FR ( n =7). ( d ) Production of TGF-beta-1 (measured as LAP) by T FR ( n =5). The horizontal bars of each graph indicate the median value and are listed where appropriate for clarity. Statistical analyses were performed by Friedman ( a , b ) or Mann-Whitney tests ( c , d ) and significance is denoted by asterisks where * P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
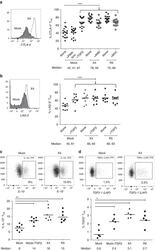
- Experimental details
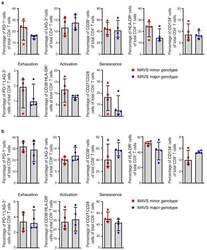
- Figure 3 MAVS genetic variation does not affect T cell exhaustion, activation, and senescence. ( a ) Percentages of PD-1 + , LAG-3 + , CD38 + , HLA-DR + , CD134 + , exhausted (PD-1 + LAG-3 + ), activated (CD38 + HLA-DR + ), and senescent (CD27 - CD28 - ) cells within CD4 + and ( b ) CD8 + T cells of untreated HIV-1-infected individuals with a MAVS minor or MAVS major genotype 2.5-3.5 years p.SC were analyzed using flow cytometry. Each square or dot represents a different study participant (median +- IQR). No significant differences between HIV-1-infected individuals with a MAVS minor or MAVS major genotype were observed.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 MAVS minor genotype is associated with a decreased percentage of naive CD4 + T cells. ( a ) Percentages and ( b ) cell counts of naive (T naive ; CD45RA + CD27 + CCR7 + ), terminally differentiated effector memory (TEMRA; CD45RA + CCR7 - CD27 - ), central memory (CM; CD45RA - CCR7 + CD27 + ), transitional memory (TM; CD45RA - CCR7 - CD27 + ), and effector memory (EM; CD45RA - CCR7 - CD27 - ) cells within CD4 + and CD8 + T cells were analyzed using flow cytometry. ( c ) Percentages of exhausted (PD-1 + LAG-3 + ) and activated (CD38 + HLA-DR + ) CD4 + T cells and ( d ) CD8 + T cells within T naive , TEMRA, CM, TM, and EM populations were analyzed using flow cytometry. Each square or dot represents a different study participant (median +- IQR). All significant differences are indicated: * p < 0.05, unpaired Mann-Whitney test.
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry