17-3109-42
antibody from Invitrogen Antibodies
Targeting: HAVCR2
CD366, FLJ14428, Tim-3, TIM3, TIMD3
Antibody data
- Antibody Data
- Antigen structure
- References [13]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 17-3109-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD366 (TIM3) Monoclonal Antibody (F38-2E2), APC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: This F38-2E2 monoclonal antibody reacts with human CD366, also known as T cell immunoglobulin and mucin domain-containing protein 3 (TIM3) or HAVCR2. This cell surface receptor is expressed on activated CD4+ T cell subsets (e.g. Th1, Th17, and Treg), CD8+ T cells, monocytes, dendritic cells, and mast cells. Due to alternative splicing, CD366 exists as membrane-bound and soluble forms. Galectin-9 has been identified as the ligand for CD366. In humans, this receptor negatively regulates CD4+ T cells, influencing the secretion of some Th1- and Th17-related cytokines. CD366 has also been implicated in tolerance, autoimmune disease (e.g. multiple sclerosis), and HIV infection. Applications Reported: This F38-2E2 antibody has been reported for use in flow cytometric analysis. Applications Tested: This F38-2E2 antibody has been pre-titrated and tested by flow cytometric analysis of stimulated normal human peripheral blood cells. This can be used at 5 µL (0.06 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 633-647 nm; Emission: 660 nm; Laser: Red Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- F38-2E2
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Expanding anti-CD38 immunotherapy for lymphoid malignancies.
Bispecific T-Cell Engaging Antibodies Against MUC16 Demonstrate Efficacy Against Ovarian Cancer in Monotherapy and in Combination With PD-1 and VEGF Inhibition.
TIGIT(+) TIM-3(+) NK cells are correlated with NK cell exhaustion and disease progression in patients with hepatitis B virus‑related hepatocellular carcinoma.
SLC1A1 mediated glutamine addiction and contributed to natural killer T-cell lymphoma progression with immunotherapeutic potential.
CRISPR Screening of CAR T Cells and Cancer Stem Cells Reveals Critical Dependencies for Cell-Based Therapies.
Neutrophil expansion defines an immunoinhibitory peripheral and intratumoral inflammatory milieu in resected non-small cell lung cancer: a descriptive analysis of a prospectively immunoprofiled cohort.
Cytomegalovirus Infection Leads to Development of High Frequencies of Cytotoxic Virus-Specific CD4+ T Cells Targeted to Vascular Endothelium.
CEACAM1 mediates B cell aggregation in central nervous system autoimmunity.
Activation and propagation of tumor-infiltrating lymphocytes on clinical-grade designer artificial antigen-presenting cells for adoptive immunotherapy of melanoma.
Cis association of galectin-9 with Tim-3 differentially regulates IL-12/IL-23 expressions in monocytes via TLR signaling.
TIM-3 is expressed on activated human CD4+ T cells and regulates Th1 and Th17 cytokines.
TIM-1 and TIM-3 proteins in immune regulation.
Dysregulated T cell expression of TIM3 in multiple sclerosis.
Wang X, Yu X, Li W, Neeli P, Liu M, Li L, Zhang M, Fang X, Young KH, Li Y
Journal of experimental & clinical cancer research : CR 2022 Jun 28;41(1):210
Journal of experimental & clinical cancer research : CR 2022 Jun 28;41(1):210
Bispecific T-Cell Engaging Antibodies Against MUC16 Demonstrate Efficacy Against Ovarian Cancer in Monotherapy and in Combination With PD-1 and VEGF Inhibition.
Yeku OO, Rao TD, Laster I, Kononenko A, Purdon TJ, Wang P, Cui Z, Liu H, Brentjens RJ, Spriggs D
Frontiers in immunology 2021;12:663379
Frontiers in immunology 2021;12:663379
TIGIT(+) TIM-3(+) NK cells are correlated with NK cell exhaustion and disease progression in patients with hepatitis B virus‑related hepatocellular carcinoma.
Yu L, Liu X, Wang X, Yan F, Wang P, Jiang Y, Du J, Yang Z
Oncoimmunology 2021;10(1):1942673
Oncoimmunology 2021;10(1):1942673
SLC1A1 mediated glutamine addiction and contributed to natural killer T-cell lymphoma progression with immunotherapeutic potential.
Xiong J, Wang N, Zhong HJ, Cui BW, Cheng S, Sun R, Chen JY, Xu PP, Cai G, Wang L, Sun XJ, Huang JY, Zhao WL
EBioMedicine 2021 Oct;72:103614
EBioMedicine 2021 Oct;72:103614
CRISPR Screening of CAR T Cells and Cancer Stem Cells Reveals Critical Dependencies for Cell-Based Therapies.
Wang D, Prager BC, Gimple RC, Aguilar B, Alizadeh D, Tang H, Lv D, Starr R, Brito A, Wu Q, Kim LJY, Qiu Z, Lin P, Lorenzini MH, Badie B, Forman SJ, Xie Q, Brown CE, Rich JN
Cancer discovery 2021 May;11(5):1192-1211
Cancer discovery 2021 May;11(5):1192-1211
Neutrophil expansion defines an immunoinhibitory peripheral and intratumoral inflammatory milieu in resected non-small cell lung cancer: a descriptive analysis of a prospectively immunoprofiled cohort.
Mitchell KG, Diao L, Karpinets T, Negrao MV, Tran HT, Parra ER, Corsini EM, Reuben A, Federico L, Bernatchez C, Dejima H, Francisco-Cruz A, Wang J, Antonoff MB, Vaporciyan AA, Swisher SG, Cascone T, Wistuba II, Heymach JV, Gibbons DL, Zhang J, Haymaker CL, Sepesi B
Journal for immunotherapy of cancer 2020 Apr;8(1)
Journal for immunotherapy of cancer 2020 Apr;8(1)
Cytomegalovirus Infection Leads to Development of High Frequencies of Cytotoxic Virus-Specific CD4+ T Cells Targeted to Vascular Endothelium.
Pachnio A, Ciaurriz M, Begum J, Lal N, Zuo J, Beggs A, Moss P
PLoS pathogens 2016 Sep;12(9):e1005832
PLoS pathogens 2016 Sep;12(9):e1005832
CEACAM1 mediates B cell aggregation in central nervous system autoimmunity.
Rovituso DM, Scheffler L, Wunsch M, Kleinschnitz C, Dörck S, Ulzheimer J, Bayas A, Steinman L, Ergün S, Kuerten S
Scientific reports 2016 Jul 20;6:29847
Scientific reports 2016 Jul 20;6:29847
Activation and propagation of tumor-infiltrating lymphocytes on clinical-grade designer artificial antigen-presenting cells for adoptive immunotherapy of melanoma.
Forget MA, Malu S, Liu H, Toth C, Maiti S, Kale C, Haymaker C, Bernatchez C, Huls H, Wang E, Marincola FM, Hwu P, Cooper LJ, Radvanyi LG
Journal of immunotherapy (Hagerstown, Md. : 1997) 2014 Nov-Dec;37(9):448-60
Journal of immunotherapy (Hagerstown, Md. : 1997) 2014 Nov-Dec;37(9):448-60
Cis association of galectin-9 with Tim-3 differentially regulates IL-12/IL-23 expressions in monocytes via TLR signaling.
Ma CJ, Li GY, Cheng YQ, Wang JM, Ying RS, Shi L, Wu XY, Niki T, Hirashima M, Li CF, Moorman JP, Yao ZQ
PloS one 2013;8(8):e72488
PloS one 2013;8(8):e72488
TIM-3 is expressed on activated human CD4+ T cells and regulates Th1 and Th17 cytokines.
Hastings WD, Anderson DE, Kassam N, Koguchi K, Greenfield EA, Kent SC, Zheng XX, Strom TB, Hafler DA, Kuchroo VK
European journal of immunology 2009 Sep;39(9):2492-501
European journal of immunology 2009 Sep;39(9):2492-501
TIM-1 and TIM-3 proteins in immune regulation.
Su EW, Lin JY, Kane LP
Cytokine 2008 Oct;44(1):9-13
Cytokine 2008 Oct;44(1):9-13
Dysregulated T cell expression of TIM3 in multiple sclerosis.
Koguchi K, Anderson DE, Yang L, O'Connor KC, Kuchroo VK, Hafler DA
The Journal of experimental medicine 2006 Jun 12;203(6):1413-8
The Journal of experimental medicine 2006 Jun 12;203(6):1413-8
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
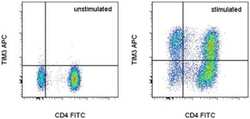
- Experimental details
- Staining of unstimulated (left) or Anti-Human CD3 and Anti-Human CD28 Functional Grade Purified (Product # 16-0037-81 and Product # 16-0289-81)-stimulated (right) normal human peripheral blood cells with Anti-Human CD4 FITC (Product # 11-0048-42) and Anti-Human CD366 (TIM3) APC. Total viable cells, as determined by Fixable Viability Dye eFluor® 780 (Product # 65-0865-14), were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure2. The co-expression of TIGIT and TIM-3 is elevated on NK cells of progression patients with HBV-HCC. a-b Percentages of TIGIT + TIM-3 + NK cells on total NK cells and NK cell subsets(CD56 bright NK cells,CD56 dim NK cells, and CD56 - NK cells) from HBV-HCC, HDs, CHB and HBV-LC patients by flow cytometry analysis. c Correlation analysis of TIGIT and TIM-3 on NK cells from patients with HBV-HCC. d-e Flow-cytometry analyses (d) of TIGIT and TIM-3 were performed on PBMCs collected from HBV-HCC patients. Representative plots (e) display the expression of TIGIT + NK cells, TIM-3 + NK cells and total TIGIT + TIM-3 + NK cells from patients with progression (n = 61) and no progression (n = 72). P values were calculated by using the Kruskal-Wallis nonparametric H test (a-c). P values were obtained by the unpaired t test (d-e). * P < .05, ** P < .01, *** P < .001, **** P < .0001
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
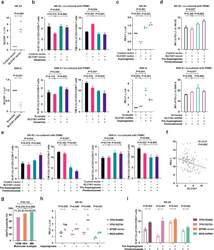
- Experimental details
- Fig. 5 Asparaginase treatment increased NKTCL cell sensitivity to anti-PD-1 antibody. (a) SLC1A1 expression on NK-92 cells transfected with SLC1A1 vector or control vector (upper panel) and SNK-6 cells transfected with SLC1A1 shRNA or scramble (lower panel). (b) Ki-67 and TIM-3 positivity of CD3+/CD8+ T cells in PBMC co-cultured with NK-92 cells (upper panel) or SNK-6 cells (lower panel) transfected with indicated vectors or shRNAs in medium with or without extra glutamine (2mM). (c) PD-L1 mRNA expression in NK-92 cells transfected with SLC1A1 vector or control vector (upper panel) and SNK-6 cells transfected with SLC1A1 shRNA or scramble (lower panel) upon asparaginase (10 IU/mL) treatment. The control vector or scramble values were normalized to 1, respectively. (d and e) Median fluorescence intensity of PD-L1 (d) on NK-92 cells (upper panel) or SNK-6 cells (lower panel), as well as Ki-67 and TIM-3 positivity of CD3+/CD8+ T cells in PBMC co-cultured with NK-92 cells (upper panel) or SNK-6 cells (lower panel) upon indicated treatment. (f) Gene expression correlation of tumor SLC1A1 with PD-L1 in NKTCL patients (n=128). (g) Tumor EAAT3 expression according to the TSIM, HEA, and MB subtypes in NKTCL patients (n=100). (h) PD-L1 mRNA expression of NK-92 cells transfected with TP53 R248Q, TP53 R273H, EP300 vector, or MGA shRNA upon indicated treatment. (i) Ki-67 positivity of CD3+/CD8+ T cells in PBMC co-cultured with NK-92 cells transfected with TP53 R248Q, TP53 R273H, EP300 vec
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry