Antibody data
- Antibody Data
- Antigen structure
- References [84]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [1]
- Flow cytometry [1]
- Other assay [52]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 32-2500 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- alpha Tubulin Monoclonal Antibody (B-5-1-2)
- Antibody type
- Monoclonal
- Antigen
- Other
- Reactivity
- Human, Mouse, Rat, Canine
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- B-5-1-2
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Modest increase of KIF11 expression exposes fragilities in the mitotic spindle, causing chromosomal instability.
The receptor kinase SRF3 coordinates iron-level and flagellin dependent defense and growth responses in plants.
Modulation of protein phosphatase 1 gamma 2 during cell division of cervical cancer HeLa cells.
Peroxisomal L-bifunctional protein (EHHADH) deficiency causes male-specific kidney hypertrophy and proximal tubular injury in mice.
Volumetric interferometric lattice light-sheet imaging.
Expansion Microscopy Reveals Plasmodium falciparum Blood-Stage Parasites Undergo Anaphase with A Chromatin Bridge in the Absence of Mini-Chromosome Maintenance Complex Binding Protein.
An immune-competent, replication-permissive Syrian Hamster glioma model for evaluating Delta-24-RGD oncolytic adenovirus.
Murine deficiency of peroxisomal L-bifunctional protein (EHHADH) causes medium-chain 3-hydroxydicarboxylic aciduria and perturbs hepatic cholesterol homeostasis.
Biallelic variants in MAATS1 encoding CFAP91, a calmodulin-associated and spoke-associated complex protein, cause severe astheno-teratozoospermia and male infertility.
Rapid image deconvolution and multiview fusion for optical microscopy.
Extracellular acidity‑induced expression of Kallikrein‑related peptidases 7 and 8 is involved in increased invasiveness of gastric cancer cells.
Slc22a5 haploinsufficiency does not aggravate the phenotype of the long-chain acyl-CoA dehydrogenase KO mouse.
Hippo-YAP1 Is a Prognosis Marker and Potentially Targetable Pathway in Advanced Gallbladder Cancer.
UHPLC-Q/Orbitrap/MS/MS fingerprinting and antitumoral effects of Prosopis strombulifera (LAM.) BENTH. queous extract on allograft colorectal and melanoma cancer models.
An Antiparasitic Compound from the Medicines for Malaria Venture Pathogen Box Promotes Leishmania Tubulin Polymerization.
Antitumor Effects of Anti-Semaphorin 4D Antibody Unravel a Novel Proinvasive Mechanism of Vascular-Targeting Agents.
Involvement of Hsp90 and cyclophilins in intoxication by AIP56, a metalloprotease toxin from Photobacterium damselae subsp. piscicida.
Selective defects in gene expression control genome instability in yeast splicing mutants.
RNA-Binding Protein HuR Regulates Both Mutant and Wild-Type IDH1 in IDH1-Mutated Cancer.
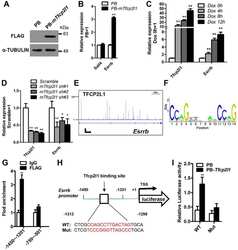
The transcription factor TFCP2L1 induces expression of distinct target genes and promotes self-renewal of mouse and human embryonic stem cells.
Fish Oil Supplementation Reduces Inflammation but Does Not Restore Renal Function and Klotho Expression in an Adenine-Induced CKD Model.
Double genetic disruption of lactate dehydrogenases A and B is required to ablate the "Warburg effect" restricting tumor growth to oxidative metabolism.
Neonatal maternal deprivation impairs localized de novo activity-induced protein translation at the synapse in the rat hippocampus.
Androgens Upregulate Endometrial Epithelial Progesterone Receptor Expression: Potential Implications for Endometriosis.
Counterbalancing anti-adhesive effects of Tenascin-C through fibronectin expression in endothelial cells.
TFCP2L1 represses multiple lineage commitment of mouse embryonic stem cells through MTA1 and LEF1.
Coupling of Insulin Secretion and Display of a Granule-resident Zinc Transporter ZnT8 on the Surface of Pancreatic Beta Cells.
Hili Inhibits HIV Replication in Activated T Cells.
A DII Domain-Based Auxin Reporter Uncovers Low Auxin Signaling during Telophase and Early G1.
Histone H1 and Chromosomal Protein HMGN2 Regulate Prolactin-induced STAT5 Transcription Factor Recruitment and Function in Breast Cancer Cells.
A Chemical-Genetic Approach Reveals the Distinct Roles of GSK3α and GSK3β in Regulating Embryonic Stem Cell Fate.
RECQ-like helicases Sgs1 and BLM regulate R-loop-associated genome instability.
Posttranscriptional Upregulation of IDH1 by HuR Establishes a Powerful Survival Phenotype in Pancreatic Cancer Cells.
Small molecule inhibitor screening identifified HSP90 inhibitor 17-AAG as potential therapeutic agent for gallbladder cancer.
Depletion of Tcf3 and Lef1 maintains mouse embryonic stem cell self-renewal.
Individual Case Analysis of Postmortem Interval Time on Brain Tissue Preservation.
HDAC6 Deacetylates HMGN2 to Regulate Stat5a Activity and Breast Cancer Growth.
Identification of NEK3 Kinase Threonine 165 as a Novel Regulatory Phosphorylation Site That Modulates Focal Adhesion Remodeling Necessary for Breast Cancer Cell Migration.
Impact of HuR inhibition by the small molecule MS-444 on colorectal cancer cell tumorigenesis.
The mRNA-binding protein HuR promotes hypoxia-induced chemoresistance through posttranscriptional regulation of the proto-oncogene PIM1 in pancreatic cancer cells.
PLEKHA7 Recruits PDZD11 to Adherens Junctions to Stabilize Nectins.
A Positive Feed-forward Loop Associating EGR1 and PDGFA Promotes Proliferation and Self-renewal in Glioblastoma Stem Cells.
Housekeeping proteins: How useful are they in skeletal muscle diabetes studies and muscle hypertrophy models?
The Conoid Associated Motor MyoH Is Indispensable for Toxoplasma gondii Entry and Exit from Host Cells.
Wnt/β-catenin and LIF-Stat3 signaling pathways converge on Sp5 to promote mouse embryonic stem cell self-renewal.
Axial superresolution via multiangle TIRF microscopy with sequential imaging and photobleaching.
The Gcn2 Regulator Yih1 Interacts with the Cyclin Dependent Kinase Cdc28 and Promotes Cell Cycle Progression through G2/M in Budding Yeast.
Klf2 and Tfcp2l1, Two Wnt/β-Catenin Targets, Act Synergistically to Induce and Maintain Naive Pluripotency.
Targeting the mRNA-binding protein HuR impairs malignant characteristics of pancreatic ductal adenocarcinoma cells.
SASH1, a new potential link between smoking and atherosclerosis.
Functional Interaction Between Na/K-ATPase and NMDA Receptor in Cerebellar Neurons.
Changes in substance P and NK1 receptor immunohistochemistry following human spinal cord injury.
Long-term disruption of maternal glucose homeostasis induced by prenatal glucocorticoid treatment correlates with miR-29 upregulation.
PLEKHA7 modulates epithelial tight junction barrier function.
Mitochondrial Rab GAPs govern autophagosome biogenesis during mitophagy.
Intracellular trafficking of AIP56, an NF-κB-cleaving toxin from Photobacterium damselae subsp. piscicida.
Progeny from irradiated colorectal cancer cells acquire an EMT-like phenotype and activate Wnt/β-catenin pathway.
Claudin-3 overexpression increases the malignant potential of colorectal cancer cells: roles of ERK1/2 and PI3K-Akt as modulators of EGFR signaling.
Modulation of β-catenin function maintains mouse epiblast stem cell and human embryonic stem cell self-renewal.
Embryonic stem cell self-renewal pathways converge on the transcription factor Tfcp2l1.
H. pylori-encoded CagA disrupts tight junctions and induces invasiveness of AGS gastric carcinoma cells via Cdx2-dependent targeting of Claudin-2.
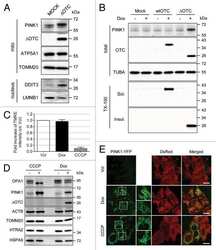
PINK1 is degraded through the N-end rule pathway.
The accumulation of misfolded proteins in the mitochondrial matrix is sensed by PINK1 to induce PARK2/Parkin-mediated mitophagy of polarized mitochondria.
Biomechanics of meniscus cells: regional variation and comparison to articular chondrocytes and ligament cells.
Resolution doubling in live, multicellular organisms via multifocal structured illumination microscopy.
Effects of proinflammatory cytokines on the claudin-19 rich tight junctions of human retinal pigment epithelium.
A gatekeeper residue for NEDD8-activating enzyme inhibition by MLN4924.
How to assess cytotoxicity of (iron oxide-based) nanoparticles: a technical note using cationic magnetoliposomes.
Distinct ATOH1 and Neurog3 requirements define tuft cells as a new secretory cell type in the intestinal epithelium.
Cytotoxic effects of iron oxide nanoparticles and implications for safety in cell labelling.
Cingulin and paracingulin show similar dynamic behaviour, but are recruited independently to junctions.
The nuclear matrix shell proteome of human epidermis.
Cyclophilin B as a co-regulator of prolactin-induced gene expression and function in breast cancer cells.
Differential gene expression in primary human skin keratinocytes and fibroblasts in response to ionizing radiation.
Expression of cyclophilin B is associated with malignant progression and regulation of genes implicated in the pathogenesis of breast cancer.
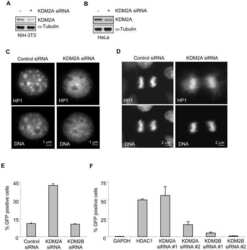
KDM2A represses transcription of centromeric satellite repeats and maintains the heterochromatic state.
Control of chromosome stability by the beta-TrCP-REST-Mad2 axis.
Carbon monoxide produced by heme oxygenase-1 in response to nitrosative stress induces expression of glutamate-cysteine ligase in PC12 cells via activation of phosphatidylinositol 3-kinase and Nrf2 signaling.
ZO-1 is required for protein kinase C gamma-driven disassembly of connexin 43.
Differentiation of mouse embryonic stem cells after RNA interference-mediated silencing of OCT4 and Nanog.
Caspase-7 gene disruption reveals an involvement of the enzyme during the early stages of apoptosis.
Gap junction-microtubule associations in rat alveolar epithelial cells.
A-kinase-anchoring protein AKAP95 is targeted to the nuclear matrix and associates with p68 RNA helicase.
Inhibition of endothelial cell migration, intercellular communication, and vascular tube formation by thromboxane A(2).
Dale KL, Armond JW, Hynds RE, Vladimirou E
Journal of cell science 2022 Sep 1;135(17)
Journal of cell science 2022 Sep 1;135(17)
The receptor kinase SRF3 coordinates iron-level and flagellin dependent defense and growth responses in plants.
Platre MP, Satbhai SB, Brent L, Gleason MF, Cao M, Grison M, Glavier M, Zhang L, Gaillochet C, Goeschl C, Giovannetti M, Enugutti B, Neveu J, von Reth M, Alcázar R, Parker JE, Vert G, Bayer E, Busch W
Nature communications 2022 Aug 1;13(1):4445
Nature communications 2022 Aug 1;13(1):4445
Modulation of protein phosphatase 1 gamma 2 during cell division of cervical cancer HeLa cells.
Agnihotri SK, Kar P, Singh M, Pant G, Mitra K, Bhatt MLB, Sachdev M
Contemporary oncology (Poznan, Poland) 2021;25(2):125-132
Contemporary oncology (Poznan, Poland) 2021;25(2):125-132
Peroxisomal L-bifunctional protein (EHHADH) deficiency causes male-specific kidney hypertrophy and proximal tubular injury in mice.
Ranea-Robles P, Portman K, Bender A, Lee K, He JC, Mulholland DJ, Argmann C, Houten SM
Kidney360 2021 Sep;2(9):1441-1454
Kidney360 2021 Sep;2(9):1441-1454
Volumetric interferometric lattice light-sheet imaging.
Cao B, Coelho S, Li J, Wang G, Pertsinidis A
Nature biotechnology 2021 Nov;39(11):1385-1393
Nature biotechnology 2021 Nov;39(11):1385-1393
Expansion Microscopy Reveals Plasmodium falciparum Blood-Stage Parasites Undergo Anaphase with A Chromatin Bridge in the Absence of Mini-Chromosome Maintenance Complex Binding Protein.
Liffner B, Absalon S
Microorganisms 2021 Nov 6;9(11)
Microorganisms 2021 Nov 6;9(11)
An immune-competent, replication-permissive Syrian Hamster glioma model for evaluating Delta-24-RGD oncolytic adenovirus.
Phillips LM, Li S, Gumin J, Daou M, Ledbetter D, Yang J, Singh S, Parker Kerrigan BC, Hossain A, Yuan Y, Gomez-Manzano C, Fueyo J, Lang FF
Neuro-oncology 2021 Nov 2;23(11):1911-1921
Neuro-oncology 2021 Nov 2;23(11):1911-1921
Murine deficiency of peroxisomal L-bifunctional protein (EHHADH) causes medium-chain 3-hydroxydicarboxylic aciduria and perturbs hepatic cholesterol homeostasis.
Ranea-Robles P, Violante S, Argmann C, Dodatko T, Bhattacharya D, Chen H, Yu C, Friedman SL, Puchowicz M, Houten SM
Cellular and molecular life sciences : CMLS 2021 Jul;78(14):5631-5646
Cellular and molecular life sciences : CMLS 2021 Jul;78(14):5631-5646
Biallelic variants in MAATS1 encoding CFAP91, a calmodulin-associated and spoke-associated complex protein, cause severe astheno-teratozoospermia and male infertility.
Martinez G, Beurois J, Dacheux D, Cazin C, Bidart M, Kherraf ZE, Robinson DR, Satre V, Le Gac G, Ka C, Gourlaouen I, Fichou Y, Petre G, Dulioust E, Zouari R, Thierry-Mieg N, Touré A, Arnoult C, Bonhivers M, Ray P, Coutton C
Journal of medical genetics 2020 Oct;57(10):708-716
Journal of medical genetics 2020 Oct;57(10):708-716
Rapid image deconvolution and multiview fusion for optical microscopy.
Guo M, Li Y, Su Y, Lambert T, Nogare DD, Moyle MW, Duncan LH, Ikegami R, Santella A, Rey-Suarez I, Green D, Beiriger A, Chen J, Vishwasrao H, Ganesan S, Prince V, Waters JC, Annunziata CM, Hafner M, Mohler WA, Chitnis AB, Upadhyaya A, Usdin TB, Bao Z, Colón-Ramos D, La Riviere P, Liu H, Wu Y, Shroff H
Nature biotechnology 2020 Nov;38(11):1337-1346
Nature biotechnology 2020 Nov;38(11):1337-1346
Extracellular acidity‑induced expression of Kallikrein‑related peptidases 7 and 8 is involved in increased invasiveness of gastric cancer cells.
Lim SC, Kee KH, Lee MJ, Hong R, Han SI
Oncology reports 2020 May;43(5):1705-1713
Oncology reports 2020 May;43(5):1705-1713
Slc22a5 haploinsufficiency does not aggravate the phenotype of the long-chain acyl-CoA dehydrogenase KO mouse.
Ranea-Robles P, Yu C, van Vlies N, Vaz FM, Houten SM
Journal of inherited metabolic disease 2020 May;43(3):486-495
Journal of inherited metabolic disease 2020 May;43(3):486-495
Hippo-YAP1 Is a Prognosis Marker and Potentially Targetable Pathway in Advanced Gallbladder Cancer.
García P, Rosa L, Vargas S, Weber H, Espinoza JA, Suárez F, Romero-Calvo I, Elgueta N, Rivera V, Nervi B, Obreque J, Leal P, Viñuela E, Aguayo G, Muñiz S, Sagredo A, Roa JC, Bizama C
Cancers 2020 Mar 25;12(4)
Cancers 2020 Mar 25;12(4)
UHPLC-Q/Orbitrap/MS/MS fingerprinting and antitumoral effects of Prosopis strombulifera (LAM.) BENTH. queous extract on allograft colorectal and melanoma cancer models.
Persia FA, Troncoso ME, Rinaldini E, Simirgiotis M, Tapia A, Bórquez J, Mackern-Oberti JP, Hapon MB, Gamarra-Luques C
Heliyon 2020 Feb;6(2):e03353
Heliyon 2020 Feb;6(2):e03353
An Antiparasitic Compound from the Medicines for Malaria Venture Pathogen Box Promotes Leishmania Tubulin Polymerization.
Ullah I, Gahalawat S, Booshehri LM, Niederstrasser H, Majumdar S, Leija C, Bradford JM, Hu B, Ready JM, Wetzel DM
ACS infectious diseases 2020 Aug 14;6(8):2057-2072
ACS infectious diseases 2020 Aug 14;6(8):2057-2072
Antitumor Effects of Anti-Semaphorin 4D Antibody Unravel a Novel Proinvasive Mechanism of Vascular-Targeting Agents.
Zuazo-Gaztelu I, Pàez-Ribes M, Carrasco P, Martín L, Soler A, Martínez-Lozano M, Pons R, Llena J, Palomero L, Graupera M, Casanovas O
Cancer research 2019 Oct 15;79(20):5328-5341
Cancer research 2019 Oct 15;79(20):5328-5341
Involvement of Hsp90 and cyclophilins in intoxication by AIP56, a metalloprotease toxin from Photobacterium damselae subsp. piscicida.
Rodrigues IS, Pereira LMG, Lisboa J, Pereira C, Oliveira P, Dos Santos NMS, do Vale A
Scientific reports 2019 Jun 21;9(1):9019
Scientific reports 2019 Jun 21;9(1):9019
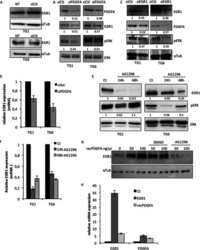
Selective defects in gene expression control genome instability in yeast splicing mutants.
Tam AS, Sihota TS, Milbury KL, Zhang A, Mathew V, Stirling PC
Molecular biology of the cell 2019 Jan 15;30(2):191-200
Molecular biology of the cell 2019 Jan 15;30(2):191-200
RNA-Binding Protein HuR Regulates Both Mutant and Wild-Type IDH1 in IDH1-Mutated Cancer.
Zarei M, Lal S, Vaziri-Gohar A, O'Hayer K, Gunda V, Singh PK, Brody JR, Winter JM
Molecular cancer research : MCR 2019 Feb;17(2):508-520
Molecular cancer research : MCR 2019 Feb;17(2):508-520
The transcription factor TFCP2L1 induces expression of distinct target genes and promotes self-renewal of mouse and human embryonic stem cells.
Wang X, Wang X, Zhang S, Sun H, Li S, Ding H, You Y, Zhang X, Ye SD
The Journal of biological chemistry 2019 Apr 12;294(15):6007-6016
The Journal of biological chemistry 2019 Apr 12;294(15):6007-6016
Fish Oil Supplementation Reduces Inflammation but Does Not Restore Renal Function and Klotho Expression in an Adenine-Induced CKD Model.
Henao Agudelo JS, Baia LC, Ormanji MS, Santos ARP, Machado JR, Saraiva Câmara NO, Navis GJ, de Borst MH, Heilberg IP
Nutrients 2018 Sep 11;10(9)
Nutrients 2018 Sep 11;10(9)
Double genetic disruption of lactate dehydrogenases A and B is required to ablate the "Warburg effect" restricting tumor growth to oxidative metabolism.
Ždralević M, Brand A, Di Ianni L, Dettmer K, Reinders J, Singer K, Peter K, Schnell A, Bruss C, Decking SM, Koehl G, Felipe-Abrio B, Durivault J, Bayer P, Evangelista M, O'Brien T, Oefner PJ, Renner K, Pouysségur J, Kreutz M
The Journal of biological chemistry 2018 Oct 12;293(41):15947-15961
The Journal of biological chemistry 2018 Oct 12;293(41):15947-15961
Neonatal maternal deprivation impairs localized de novo activity-induced protein translation at the synapse in the rat hippocampus.
Ahmad F, Salahuddin M, Alsamman K, Herzallah HK, Al-Otaibi ST
Bioscience reports 2018 Jun 29;38(3)
Bioscience reports 2018 Jun 29;38(3)
Androgens Upregulate Endometrial Epithelial Progesterone Receptor Expression: Potential Implications for Endometriosis.
Babayev SN, Park CW, Keller PW, Carr BR, Word RA, Bukulmez O
Reproductive sciences (Thousand Oaks, Calif.) 2017 Oct;24(10):1454-1461
Reproductive sciences (Thousand Oaks, Calif.) 2017 Oct;24(10):1454-1461
Counterbalancing anti-adhesive effects of Tenascin-C through fibronectin expression in endothelial cells.
Radwanska A, Grall D, Schaub S, Divonne SBF, Ciais D, Rekima S, Rupp T, Sudaka A, Orend G, Van Obberghen-Schilling E
Scientific reports 2017 Oct 6;7(1):12762
Scientific reports 2017 Oct 6;7(1):12762
TFCP2L1 represses multiple lineage commitment of mouse embryonic stem cells through MTA1 and LEF1.
Liu K, Zhang Y, Liu D, Ying QL, Ye S
Journal of cell science 2017 Nov 15;130(22):3809-3817
Journal of cell science 2017 Nov 15;130(22):3809-3817
Coupling of Insulin Secretion and Display of a Granule-resident Zinc Transporter ZnT8 on the Surface of Pancreatic Beta Cells.
Huang Q, Merriman C, Zhang H, Fu D
The Journal of biological chemistry 2017 Mar 10;292(10):4034-4043
The Journal of biological chemistry 2017 Mar 10;292(10):4034-4043
Hili Inhibits HIV Replication in Activated T Cells.
Peterlin BM, Liu P, Wang X, Cary D, Shao W, Leoz M, Hong T, Pan T, Fujinaga K
Journal of virology 2017 Jun 1;91(11)
Journal of virology 2017 Jun 1;91(11)
A DII Domain-Based Auxin Reporter Uncovers Low Auxin Signaling during Telophase and Early G1.
Mir R, Aranda LZ, Biaocchi T, Luo A, Sylvester AW, Rasmussen CG
Plant physiology 2017 Jan;173(1):863-871
Plant physiology 2017 Jan;173(1):863-871
Histone H1 and Chromosomal Protein HMGN2 Regulate Prolactin-induced STAT5 Transcription Factor Recruitment and Function in Breast Cancer Cells.
Schauwecker SM, Kim JJ, Licht JD, Clevenger CV
The Journal of biological chemistry 2017 Feb 10;292(6):2237-2254
The Journal of biological chemistry 2017 Feb 10;292(6):2237-2254
A Chemical-Genetic Approach Reveals the Distinct Roles of GSK3α and GSK3β in Regulating Embryonic Stem Cell Fate.
Chen X, Wang R, Liu X, Wu Y, Zhou T, Yang Y, Perez A, Chen YC, Hu L, Chadarevian JP, Assadieskandar A, Zhang C, Ying QL
Developmental cell 2017 Dec 4;43(5):563-576.e4
Developmental cell 2017 Dec 4;43(5):563-576.e4
RECQ-like helicases Sgs1 and BLM regulate R-loop-associated genome instability.
Chang EY, Novoa CA, Aristizabal MJ, Coulombe Y, Segovia R, Chaturvedi R, Shen Y, Keong C, Tam AS, Jones SJM, Masson JY, Kobor MS, Stirling PC
The Journal of cell biology 2017 Dec 4;216(12):3991-4005
The Journal of cell biology 2017 Dec 4;216(12):3991-4005
Posttranscriptional Upregulation of IDH1 by HuR Establishes a Powerful Survival Phenotype in Pancreatic Cancer Cells.
Zarei M, Lal S, Parker SJ, Nevler A, Vaziri-Gohar A, Dukleska K, Mambelli-Lisboa NC, Moffat C, Blanco FF, Chand SN, Jimbo M, Cozzitorto JA, Jiang W, Yeo CJ, Londin ER, Seifert EL, Metallo CM, Brody JR, Winter JM
Cancer research 2017 Aug 15;77(16):4460-4471
Cancer research 2017 Aug 15;77(16):4460-4471
Small molecule inhibitor screening identifified HSP90 inhibitor 17-AAG as potential therapeutic agent for gallbladder cancer.
Weber H, Valbuena JR, Barbhuiya MA, Stein S, Kunkel H, García P, Bizama C, Riquelme I, Espinoza JA, Kurtz SE, Tyner JW, Calderon JF, Corvalán AH, Grez M, Pandey A, Leal-Rojas P, Roa JC
Oncotarget 2017 Apr 18;8(16):26169-26184
Oncotarget 2017 Apr 18;8(16):26169-26184
Depletion of Tcf3 and Lef1 maintains mouse embryonic stem cell self-renewal.
Ye S, Zhang T, Tong C, Zhou X, He K, Ban Q, Liu D, Ying QL
Biology open 2017 Apr 15;6(4):511-517
Biology open 2017 Apr 15;6(4):511-517
Individual Case Analysis of Postmortem Interval Time on Brain Tissue Preservation.
Blair JA, Wang C, Hernandez D, Siedlak SL, Rodgers MS, Achar RK, Fahmy LM, Torres SL, Petersen RB, Zhu X, Casadesus G, Lee HG
PloS one 2016;11(3):e0151615
PloS one 2016;11(3):e0151615
HDAC6 Deacetylates HMGN2 to Regulate Stat5a Activity and Breast Cancer Growth.
Medler TR, Craig JM, Fiorillo AA, Feeney YB, Harrell JC, Clevenger CV
Molecular cancer research : MCR 2016 Oct;14(10):994-1008
Molecular cancer research : MCR 2016 Oct;14(10):994-1008
Identification of NEK3 Kinase Threonine 165 as a Novel Regulatory Phosphorylation Site That Modulates Focal Adhesion Remodeling Necessary for Breast Cancer Cell Migration.
Harrington KM, Clevenger CV
The Journal of biological chemistry 2016 Oct 7;291(41):21388-21406
The Journal of biological chemistry 2016 Oct 7;291(41):21388-21406
Impact of HuR inhibition by the small molecule MS-444 on colorectal cancer cell tumorigenesis.
Blanco FF, Preet R, Aguado A, Vishwakarma V, Stevens LE, Vyas A, Padhye S, Xu L, Weir SJ, Anant S, Meisner-Kober N, Brody JR, Dixon DA
Oncotarget 2016 Nov 8;7(45):74043-74058
Oncotarget 2016 Nov 8;7(45):74043-74058
The mRNA-binding protein HuR promotes hypoxia-induced chemoresistance through posttranscriptional regulation of the proto-oncogene PIM1 in pancreatic cancer cells.
Blanco FF, Jimbo M, Wulfkuhle J, Gallagher I, Deng J, Enyenihi L, Meisner-Kober N, Londin E, Rigoutsos I, Sawicki JA, Risbud MV, Witkiewicz AK, McCue PA, Jiang W, Rui H, Yeo CJ, Petricoin E, Winter JM, Brody JR
Oncogene 2016 May;35(19):2529-41
Oncogene 2016 May;35(19):2529-41
PLEKHA7 Recruits PDZD11 to Adherens Junctions to Stabilize Nectins.
Guerrera D, Shah J, Vasileva E, Sluysmans S, Méan I, Jond L, Poser I, Mann M, Hyman AA, Citi S
The Journal of biological chemistry 2016 May 20;291(21):11016-29
The Journal of biological chemistry 2016 May 20;291(21):11016-29
A Positive Feed-forward Loop Associating EGR1 and PDGFA Promotes Proliferation and Self-renewal in Glioblastoma Stem Cells.
Sakakini N, Turchi L, Bergon A, Holota H, Rekima S, Lopez F, Paquis P, Almairac F, Fontaine D, Baeza-Kallee N, Van Obberghen-Schilling E, Junier MP, Chneiweiss H, Figarella-Branger D, Burel-Vandenbos F, Imbert J, Virolle T
The Journal of biological chemistry 2016 May 13;291(20):10684-99
The Journal of biological chemistry 2016 May 13;291(20):10684-99
Housekeeping proteins: How useful are they in skeletal muscle diabetes studies and muscle hypertrophy models?
Fortes MA, Marzuca-Nassr GN, Vitzel KF, da Justa Pinheiro CH, Newsholme P, Curi R
Analytical biochemistry 2016 Jul 1;504:38-40
Analytical biochemistry 2016 Jul 1;504:38-40
The Conoid Associated Motor MyoH Is Indispensable for Toxoplasma gondii Entry and Exit from Host Cells.
Graindorge A, Frénal K, Jacot D, Salamun J, Marq JB, Soldati-Favre D
PLoS pathogens 2016 Jan;12(1):e1005388
PLoS pathogens 2016 Jan;12(1):e1005388
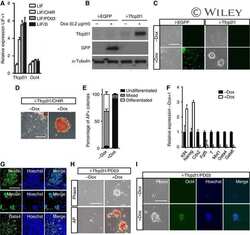
Wnt/β-catenin and LIF-Stat3 signaling pathways converge on Sp5 to promote mouse embryonic stem cell self-renewal.
Ye S, Zhang D, Cheng F, Wilson D, Mackay J, He K, Ban Q, Lv F, Huang S, Liu D, Ying QL
Journal of cell science 2016 Jan 15;129(2):269-76
Journal of cell science 2016 Jan 15;129(2):269-76
Axial superresolution via multiangle TIRF microscopy with sequential imaging and photobleaching.
Fu Y, Winter PW, Rojas R, Wang V, McAuliffe M, Patterson GH
Proceedings of the National Academy of Sciences of the United States of America 2016 Apr 19;113(16):4368-73
Proceedings of the National Academy of Sciences of the United States of America 2016 Apr 19;113(16):4368-73
The Gcn2 Regulator Yih1 Interacts with the Cyclin Dependent Kinase Cdc28 and Promotes Cell Cycle Progression through G2/M in Budding Yeast.
Silva RC, Dautel M, Di Genova BM, Amberg DC, Castilho BA, Sattlegger E
PloS one 2015;10(7):e0131070
PloS one 2015;10(7):e0131070
Klf2 and Tfcp2l1, Two Wnt/β-Catenin Targets, Act Synergistically to Induce and Maintain Naive Pluripotency.
Qiu D, Ye S, Ruiz B, Zhou X, Liu D, Zhang Q, Ying QL
Stem cell reports 2015 Sep 8;5(3):314-22
Stem cell reports 2015 Sep 8;5(3):314-22
Targeting the mRNA-binding protein HuR impairs malignant characteristics of pancreatic ductal adenocarcinoma cells.
Jimbo M, Blanco FF, Huang YH, Telonis AG, Screnci BA, Cosma GL, Alexeev V, Gonye GE, Yeo CJ, Sawicki JA, Winter JM, Brody JR
Oncotarget 2015 Sep 29;6(29):27312-31
Oncotarget 2015 Sep 29;6(29):27312-31
SASH1, a new potential link between smoking and atherosclerosis.
Weidmann H, Touat-Hamici Z, Durand H, Mueller C, Chardonnet S, Pionneau C, Charlotte F, Janssen KP, Verdugo R, Cambien F, Blankenberg S, Tiret L, Zeller T, Ninio E
Atherosclerosis 2015 Oct;242(2):571-9
Atherosclerosis 2015 Oct;242(2):571-9
Functional Interaction Between Na/K-ATPase and NMDA Receptor in Cerebellar Neurons.
Akkuratov EE, Lopacheva OM, Kruusmägi M, Lopachev AV, Shah ZA, Boldyrev AA, Liu L
Molecular neurobiology 2015 Dec;52(3):1726-1734
Molecular neurobiology 2015 Dec;52(3):1726-1734
Changes in substance P and NK1 receptor immunohistochemistry following human spinal cord injury.
Leonard AV, Manavis J, Blumbergs PC, Vink R
Spinal cord 2014 Jan;52(1):17-23
Spinal cord 2014 Jan;52(1):17-23
Long-term disruption of maternal glucose homeostasis induced by prenatal glucocorticoid treatment correlates with miR-29 upregulation.
Gomes PR, Graciano MF, Pantaleão LC, Rennó AL, Rodrigues SC, Velloso LA, Latorraca MQ, Carpinelli AR, Anhê GF, Bordin S
American journal of physiology. Endocrinology and metabolism 2014 Jan 1;306(1):E109-20
American journal of physiology. Endocrinology and metabolism 2014 Jan 1;306(1):E109-20
PLEKHA7 modulates epithelial tight junction barrier function.
Paschoud S, Jond L, Guerrera D, Citi S
Tissue barriers 2014 Jan 1;2(1):e28755
Tissue barriers 2014 Jan 1;2(1):e28755
Mitochondrial Rab GAPs govern autophagosome biogenesis during mitophagy.
Yamano K, Fogel AI, Wang C, van der Bliek AM, Youle RJ
eLife 2014 Feb 25;3:e01612
eLife 2014 Feb 25;3:e01612
Intracellular trafficking of AIP56, an NF-κB-cleaving toxin from Photobacterium damselae subsp. piscicida.
Pereira LM, Pinto RD, Silva DS, Moreira AR, Beitzinger C, Oliveira P, Sampaio P, Benz R, Azevedo JE, dos Santos NM, do Vale A
Infection and immunity 2014 Dec;82(12):5270-85
Infection and immunity 2014 Dec;82(12):5270-85
Progeny from irradiated colorectal cancer cells acquire an EMT-like phenotype and activate Wnt/β-catenin pathway.
Bastos LG, de Marcondes PG, de-Freitas-Junior JC, Leve F, Mencalha AL, de Souza WF, de Araujo WM, Tanaka MN, Abdelhay ES, Morgado-Díaz JA
Journal of cellular biochemistry 2014 Dec;115(12):2175-87
Journal of cellular biochemistry 2014 Dec;115(12):2175-87
Claudin-3 overexpression increases the malignant potential of colorectal cancer cells: roles of ERK1/2 and PI3K-Akt as modulators of EGFR signaling.
de Souza WF, Fortunato-Miranda N, Robbs BK, de Araujo WM, de-Freitas-Junior JC, Bastos LG, Viola JP, Morgado-Díaz JA
PloS one 2013;8(9):e74994
PloS one 2013;8(9):e74994
Modulation of β-catenin function maintains mouse epiblast stem cell and human embryonic stem cell self-renewal.
Kim H, Wu J, Ye S, Tai CI, Zhou X, Yan H, Li P, Pera M, Ying QL
Nature communications 2013;4:2403
Nature communications 2013;4:2403
Embryonic stem cell self-renewal pathways converge on the transcription factor Tfcp2l1.
Ye S, Li P, Tong C, Ying QL
The EMBO journal 2013 Oct 2;32(19):2548-60
The EMBO journal 2013 Oct 2;32(19):2548-60
H. pylori-encoded CagA disrupts tight junctions and induces invasiveness of AGS gastric carcinoma cells via Cdx2-dependent targeting of Claudin-2.
Song X, Chen HX, Wang XY, Deng XY, Xi YX, He Q, Peng TL, Chen J, Chen W, Wong BC, Chen MH
Cellular immunology 2013 Nov-Dec;286(1-2):22-30
Cellular immunology 2013 Nov-Dec;286(1-2):22-30
PINK1 is degraded through the N-end rule pathway.
Yamano K, Youle RJ
Autophagy 2013 Nov 1;9(11):1758-69
Autophagy 2013 Nov 1;9(11):1758-69
The accumulation of misfolded proteins in the mitochondrial matrix is sensed by PINK1 to induce PARK2/Parkin-mediated mitophagy of polarized mitochondria.
Jin SM, Youle RJ
Autophagy 2013 Nov 1;9(11):1750-7
Autophagy 2013 Nov 1;9(11):1750-7
Biomechanics of meniscus cells: regional variation and comparison to articular chondrocytes and ligament cells.
Sanchez-Adams J, Athanasiou KA
Biomechanics and modeling in mechanobiology 2012 Sep;11(7):1047-56
Biomechanics and modeling in mechanobiology 2012 Sep;11(7):1047-56
Resolution doubling in live, multicellular organisms via multifocal structured illumination microscopy.
York AG, Parekh SH, Dalle Nogare D, Fischer RS, Temprine K, Mione M, Chitnis AB, Combs CA, Shroff H
Nature methods 2012 May 13;9(7):749-54
Nature methods 2012 May 13;9(7):749-54
Effects of proinflammatory cytokines on the claudin-19 rich tight junctions of human retinal pigment epithelium.
Peng S, Gan G, Rao VS, Adelman RA, Rizzolo LJ
Investigative ophthalmology & visual science 2012 Jul 27;53(8):5016-28
Investigative ophthalmology & visual science 2012 Jul 27;53(8):5016-28
A gatekeeper residue for NEDD8-activating enzyme inhibition by MLN4924.
Toth JI, Yang L, Dahl R, Petroski MD
Cell reports 2012 Apr 19;1(4):309-16
Cell reports 2012 Apr 19;1(4):309-16
How to assess cytotoxicity of (iron oxide-based) nanoparticles: a technical note using cationic magnetoliposomes.
Soenen SJ, De Cuyper M
Contrast media & molecular imaging 2011 May-Jun;6(3):153-64
Contrast media & molecular imaging 2011 May-Jun;6(3):153-64
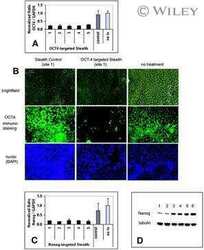
Distinct ATOH1 and Neurog3 requirements define tuft cells as a new secretory cell type in the intestinal epithelium.
Gerbe F, van Es JH, Makrini L, Brulin B, Mellitzer G, Robine S, Romagnolo B, Shroyer NF, Bourgaux JF, Pignodel C, Clevers H, Jay P
The Journal of cell biology 2011 Mar 7;192(5):767-80
The Journal of cell biology 2011 Mar 7;192(5):767-80
Cytotoxic effects of iron oxide nanoparticles and implications for safety in cell labelling.
Soenen SJ, Himmelreich U, Nuytten N, De Cuyper M
Biomaterials 2011 Jan;32(1):195-205
Biomaterials 2011 Jan;32(1):195-205
Cingulin and paracingulin show similar dynamic behaviour, but are recruited independently to junctions.
Paschoud S, Yu D, Pulimeno P, Jond L, Turner JR, Citi S
Molecular membrane biology 2011 Feb;28(2):123-35
Molecular membrane biology 2011 Feb;28(2):123-35
The nuclear matrix shell proteome of human epidermis.
Warters RL, Cassidy PB, Sunseri JA, Parsawar K, Zhuplatov SB, Kramer GF, Leachman SA
Journal of dermatological science 2010 May;58(2):113-22
Journal of dermatological science 2010 May;58(2):113-22
Cyclophilin B as a co-regulator of prolactin-induced gene expression and function in breast cancer cells.
Fang F, Zheng J, Galbaugh TL, Fiorillo AA, Hjort EE, Zeng X, Clevenger CV
Journal of molecular endocrinology 2010 Jun;44(6):319-29
Journal of molecular endocrinology 2010 Jun;44(6):319-29
Differential gene expression in primary human skin keratinocytes and fibroblasts in response to ionizing radiation.
Warters RL, Packard AT, Kramer GF, Gaffney DK, Moos PJ
Radiation research 2009 Jul;172(1):82-95
Radiation research 2009 Jul;172(1):82-95
Expression of cyclophilin B is associated with malignant progression and regulation of genes implicated in the pathogenesis of breast cancer.
Fang F, Flegler AJ, Du P, Lin S, Clevenger CV
The American journal of pathology 2009 Jan;174(1):297-308
The American journal of pathology 2009 Jan;174(1):297-308
KDM2A represses transcription of centromeric satellite repeats and maintains the heterochromatic state.
Frescas D, Guardavaccaro D, Kuchay SM, Kato H, Poleshko A, Basrur V, Elenitoba-Johnson KS, Katz RA, Pagano M
Cell cycle (Georgetown, Tex.) 2008 Nov 15;7(22):3539-47
Cell cycle (Georgetown, Tex.) 2008 Nov 15;7(22):3539-47
Control of chromosome stability by the beta-TrCP-REST-Mad2 axis.
Guardavaccaro D, Frescas D, Dorrello NV, Peschiaroli A, Multani AS, Cardozo T, Lasorella A, Iavarone A, Chang S, Hernando E, Pagano M
Nature 2008 Mar 20;452(7185):365-9
Nature 2008 Mar 20;452(7185):365-9
Carbon monoxide produced by heme oxygenase-1 in response to nitrosative stress induces expression of glutamate-cysteine ligase in PC12 cells via activation of phosphatidylinositol 3-kinase and Nrf2 signaling.
Li MH, Jang JH, Na HK, Cha YN, Surh YJ
The Journal of biological chemistry 2007 Sep 28;282(39):28577-28586
The Journal of biological chemistry 2007 Sep 28;282(39):28577-28586
ZO-1 is required for protein kinase C gamma-driven disassembly of connexin 43.
Akoyev V, Takemoto DJ
Cellular signalling 2007 May;19(5):958-67
Cellular signalling 2007 May;19(5):958-67
Differentiation of mouse embryonic stem cells after RNA interference-mediated silencing of OCT4 and Nanog.
Hough SR, Clements I, Welch PJ, Wiederholt KA
Stem cells (Dayton, Ohio) 2006 Jun;24(6):1467-75
Stem cells (Dayton, Ohio) 2006 Jun;24(6):1467-75
Caspase-7 gene disruption reveals an involvement of the enzyme during the early stages of apoptosis.
Korfali N, Ruchaud S, Loegering D, Bernard D, Dingwall C, Kaufmann SH, Earnshaw WC
The Journal of biological chemistry 2004 Jan 9;279(2):1030-9
The Journal of biological chemistry 2004 Jan 9;279(2):1030-9
Gap junction-microtubule associations in rat alveolar epithelial cells.
Guo Y, Martinez-Williams C, Rannels DE
American journal of physiology. Lung cellular and molecular physiology 2003 Dec;285(6):L1213-21
American journal of physiology. Lung cellular and molecular physiology 2003 Dec;285(6):L1213-21
A-kinase-anchoring protein AKAP95 is targeted to the nuclear matrix and associates with p68 RNA helicase.
Akileswaran L, Taraska JW, Sayer JA, Gettemy JM, Coghlan VM
The Journal of biological chemistry 2001 May 18;276(20):17448-54
The Journal of biological chemistry 2001 May 18;276(20):17448-54
Inhibition of endothelial cell migration, intercellular communication, and vascular tube formation by thromboxane A(2).
Ashton AW, Yokota R, John G, Zhao S, Suadicani SO, Spray DC, Ware JA
The Journal of biological chemistry 1999 Dec 10;274(50):35562-70
The Journal of biological chemistry 1999 Dec 10;274(50):35562-70
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of Alpha-tubulin was performed by loading 20 µg of U2OS (lane1), HEK-293 (lane2), HeLa (lane3), MCF7 (lane4) and Jurkat (lane5) cell lysate using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (LC5800), and iBlot® Dry Blotting System (IB21001). Proteins were transferred to a nitrocellulose membrane and blocked with 5 % skim milk for 1 hour at room temperature. Alpha-Tubulin was detected at ~55 kDa using Alpha-Tubulin Mouse Monoclonal Antibody (Product # 32-2500) at 0.5-1 µg/mL in 2.5 % skim milk at 4°C overnight on a rocking platform. Goat Anti-Mouse IgG - HRP Secondary Antibody (Product # 62-6520) at 1:4000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis was performed on whole cell extracts (30µg lysate) of COS-7 (Lane 1), MDCK (Lane 2), NIH/3T3 (Lane 3), MDA-MB-231 (Lane 4), L6 (Lane 5), RSC96 (Lane 6), tissue extracts of Mouse Lung (Lane 7), Rat Liver (Lane 8) and Mouse Pancreas (Lane 9). The blot was probed with Anti- alpha Tubulin Mouse Monoclonal Antibody (Product # 32-2500, 1µg/mL) and detected by chemiluminescence using Goat anti-Mouse IgG (H+L) Superclonal™ Secondary Antibody, HRP conjugate (Product # A28177, 0.25µg/mL, 1:4000 dilution). A 52 kDa band corresponding to alpha Tubulin was observed across the cell lines and tissues tested. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0322BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane with iBlot® 2 Dry Blotting System (Product # IB21001). The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of Alpha - Tubulin was done on 70% confluent log phase U-2 OS cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with Alpha - Tubulin Mouse Monoclonal Antibody (Product # 32-2500) at 0.5 µg/mL in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Rabbit Anti-Mouse IgG Secondary Antibody (Product # A-11059) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing cytoplasmic localization. Panel e shows no primary antibody control. The images were captured at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Flow cytometry analysis of Alpha-tubulin was done on HeLa cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Tritonª X-100 for 20 minutes, and blocked with 5% BSA for 1 hour at room temperature. Cells were labeled with Alpha-tubulin Mouse Monoclonal Antibody (322500, red histogram) or with mouse isotype control (pink histogram) at 1-3 µg/million cells in 2.5% BSA. After incubation at room temperature for 2-3 hours, the cells were labeled with Alexa Fluor¨ 488 Rabbit Anti-Mouse Secondary Antibody (A11059) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune¨ Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. Molecular characterization of mouse intestinal tuft cells. Immunofluorescent stainings for (A) SOX9 and COX1, (B) SOX9 and COX2, (C) HPGDS and COX1, (D) villin and COX1, (E) alpha-tubulin and COX1, and (F) DCLK1 and COX1. Each panel contains a merged image on the left, and gray level pictures of the indicated individual markers corresponding to the yellow inset area on the right. (G) Whole-mount immunofluorescent staining for DCLK1 and F-actin on a dissociated fragment of intestinal epithelium. Panels on the right show higher magnification of the cropped area of the overlay image. Yellow arrowheads point at tuft cells. Nuclei are stained with Hoechst (blue). Bars, 10 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6. Atoh1 is required for tuft cell differentiation. Immunofluorescent staining for the SOX9 transcription factor (A and C), the COX1 enzyme (A-D), and for the structural- and morphological-related tuft cells markers DCLK1 and alpha-tubulin (B and D) in intestines from control (A and B) and Atoh1 -deficient mice (C and D), 3 wk after tamoxifen injection. Each panel contains the merged image on the left, and separate pictures of the indicated markers corresponding to the yellow inset on the right. Yellow arrowheads point at tuft cells revealed by SOX9 and COX1 or DCLK1, alpha-tubulin, and COX1 expression. Nuclei are stained with Hoechst (blue). Bars, 10 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Effect of EGF on the claudins 1 and 3 expression in Caco-2 and HT-29 cells. Cultured Caco-2 and HT-29 cells were treated with EGF (20 ng/mL) for 6 h ( A ), 24 h and 48 h ( B ). Following EGF treatment, total cell lysates were harvested and analyzed by immunoblotting for the expression of claudins 1 and 3. alpha-tubulin was used as a loading control. The numbers represent the ratio of the optical density of EGF-treated to untreated cells normalized by alpha-tubulin. Claudin ; Cld. alpha -tubulin ; alpha-tub.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1. Generation of stable MDCK tet-off cell clones with inducible expression of PLEKHA7 constructs. ( A ) Schematic diagrams showing the putative minimal regions of PLEKHA7 which can interact with afadin (residues 120-374 in human PLEKHA7), p120ctn (538-696), CGNL1 (620-769), or nezha (680-821), 21 , 23 , 29 and schematic structure of the PLEKHA7 constructs (Full-length FL, N-terminal = N-term., C-terminal = C-term., Central) used to generate stable clones of MDCK tet-off cells. Constructs were tagged N-terminally with YFP, and C-terminally with myc. Putative structural domains are indicated: WW (purple), PH (brown), Pro-rich (P, red), and coiled-coil (CC, blue, CC1 = residues 686-835, and CC2 = residues 1055-1095, based on Phyre2 analysis). Numbers indicate position of PLEKHA7 amino acid residues within constructs. ( B ) Immunoblotting analysis of lysates of MDCK cell lines (two clones for each construct shown) cultured either in the presence (+) or in the absence (-) of doxycycline, using antibodies against the myc tag (to detect exogenous PLEKHA7), E-cadherin (E-cad.), cingulin (CGN) and tubulin (loading control). Numbers on the right indicate the migration of pre-stained markers (kDa). ( C ) Confocal immunofluorescent analysis of induced cultures of stable clones showing the localization of exogenous PLEKHA7 constructs (green, YFP) and alpha-tubulin (red), on the apical plane of focus (ZO-1 labeling is shown for control cells, panel E). Arrows = junctional labeling;
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 2 Deletion of YIH1 leads to the accumulation of cells in the G2/M phases of the cell cycle. (A) Representative images of cells grown asynchronously to log phase in YPD medium, spheroplasted with zymolyase, permeabilized, fixed and stained with DAPI to visualize the nuclear DNA (left panels) and with anti-tubulin antibodies followed by Alexa-488 anti-mouse IgG to visualize spindles (middle panels). (B) Cells prepared as in A were counted and the percentage of cells with medium sized-buds and short spindles and the localization of the nucleus as indicated (left), or with large buds and elongated spindles (right), were determined. (C) Representative images of live exponentially growing yeast cells stained with DAPI. (D) Quantification of the percentage of cells in G1 (unbudded), S (cells with small buds) and G2/M (cells with medium to large buds) determined from (C). Over 450 cells were analyzed per experiment. Data represent the mean +- S.E. as error bars of three independent experiments for B and D. * p < 0.05; (Student's t test).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Characterization of DOX-inducible MIA PaCa-2 cell lines A. qPCR analysis of HuR mRNA expression in Mia.CTRL, Mia.sh290, and Mia.sh700 cells treated with 0 or 2 mug/ml DOX for 5 days ( left ), and Mia.EV and Mia.HuR cells treated with 0 or 2 mug/ml DOX for 2 days ( right ), normalized to 18S rRNA expression. Mia.sh290 and Mia.sh700 are inducible HuR-silencing cell lines, whereas Mia.HuR is an inducible HuR-overexpressing cell line. Mia.CTRL and Mia.EV are the respective control cell lines. B. Western blotting analysis of HuR protein expression in Mia.CTRL, Mia.sh290, and Mia.sh700 cells treated with 0 or 2 mug/ml DOX for 5 days ( left ), and Mia.EV and Mia.HuR cells treated with 0 or 2 mug/ml DOX for 2 days ( right ), normalized to alpha-tubulin protein expression. ns = non-significant; *** = p < 0.001; **** = p < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1. Overexpressing Sp5 partially mimics the effect of CHIR in medium with 2i. (A) Flow chart showing the method used to identify candidate genes downstream of CHIR and Stat3. (B) qRT-PCR analysis of Socs3 and Sp5 expression induced by CHIR, PD03 and 2i (left), or LIF (right) for 12 h. Data represent mean+-s.d. of three biological replicates. ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3. Sp5 is a direct target of Stat3. (A) qRT-PCR analysis of Socs3 and Sp5 expression levels in 46C mESCs deprived of LIF overnight and stimulated with LIF for 1 h in the presence or absence of JAK inhibitor I (JAKi) or cycloheximide (CHX, 50 mg/ml). Data represent mean+-s.d. of three biological replicates. ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4. Forced expression of Sp5 promotes EpiSC reprogramming to naive pluripotency. (A) Phase contrast and alkaline phosphatase (AP) staining images of 46C mESCs and 46C mESC-derived EpiSCs. Scale bar: 100 mum. (B) qRT-PCR analysis of expression patterns of Sp5 , Oct4 , Rex1 and Fgf5 in 46C mESCs and EpiSCs. Data represent mean+-s.d. of three biological replicates. ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
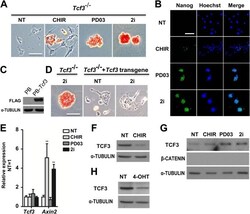
- Experimental details
- Fig. 1. CHIR promotes mESC self-renewal via down-regulation of TCF3 protein in a beta-catenin-dependent manner. (A,B) Alkaline phosphatase (AP) staining and immunofluorescence images of Tcf3 -/- mESCs cultured in N2B27 supplemented with the indicated small molecules for two passages. Hoechst, Hoechst 33342. (C) Western blot analysis of the expression of FLAG in Tcf3 -/- mESCs overexpressing FLAG-tagged Tcf3 transgene. alpha-tubulin is a loading control. PB, PiggyBac. (D) AP staining of Tcf3 -/- mESCs and Tcf3 -/- mESCs overexpressing Tcf3 transgene. Cells were cultured in N2B27 medium with or without 2i for two passages. (E,F) qRT-PCR (E) and western blot (F) analysis of Tcf3 and Axin2 expression in 46C ESCs cultured under the indicated conditions for 12 h. (G,H) Western blot analysis of TCF3 expression in Ctnnb1 (beta-catenin) -/- ESCs (G) and Ctnnb1 -/- ESCs overexpressing Ctnnb1 -ERT2 (H). Cells were deprived of 2i/LIF overnight and then treated with the indicated compounds for 12 h in N2B27 medium. NT, no treatment; 4-OHT, 4-hydroxytamoxifen; ERT2, a mutant estrogen ligand-binding domain. Scale bars: 100 mum. Data represent mean+-s.d. of three biological replicates. ** P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 FN and TNC expression in different endothelial cell models. ( a ) Western analysis (cropped blots) of FN (top) and TNC (bottom) expression in indicated cells. Culture medium (25 ul), left; total cell extracts of equal protein concentration (40 ug), right. Equivalent amounts of FN-depleted serum-containing (10%) culture medium were deposited as control. alpha-tubulin was used as loading control for total cell extracts. Full-length blots are shown in Fig. S5 . ( b ) Detection of cFN isoform transcripts (containing EDB+/EDA+, EDB+/EDA-, EDB-/EDA+, EDB-/EDA-, as schematically presented on the top) by RT-PCR in human endothelial cells and TIFs. 18S rRNA was used as internal control/housekeeping gene. Cropped blots are shown in ( a ) and ( b ). ( c ) QPCR analysis of FN, FN-EDB, FN-EDA and TNC mRNA expression in endothelial cells, normalized with GAPDH and presented as log DeltaCt (+-S.D., n = 5). ( d ) Double immunofluorescence staining (wide-field fluorescence) of total FN (red) and, in green, FN-EDA (left); FN-EDB (middle) or TNC (right) in confluent endothelial cells and TIFs. Cell density was evaluated after Hoechst 33342 staining (30-50 cells in each field). Bar = 100.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Effect of TNC on FN expression and assembly. ( a ) Representative western analysis of FN expression in total cell extracts of HUVECs cultured for 48 h on TNC-coated, BSA-pretreated or non-coated dishes. Equal amounts of protein (40 ug) were loaded in each lane and alpha-tubulin was used as loading control. Fold change over NC (normalized to alpha-tubulin) is shown below cropped blot. Full-length blots are shown in Fig. S7 . ( b ) QPCR analysis of FN mRNA expression levels in different endothelial cells (left, HUVEC; HMEC, middle and HMVEC-d, right) cultured on indicated substrates in comparison to a non-coated (NC) surface ( c ). Time course (12-72 h) expression of FN (left), Axin2 (middle) and DKK1 mRNA (right) in HUVECs on TNC coating, non-coated or BSA-pretreated dish was monitored by QPCR. Fold change over NC (equal to 1, not shown on the graphs) was calculated using the DeltaDeltaCt method (+-S.D., n = 3). ( d ) Immunofluorescence staining of cortactin (white or green), filamentous actin (red) and nuclei (blue) of cells plated for 48 h on non-coated or TNC-coated coverslips. The zoomed area is indicated in white dotted square (right). Bar = 50 um, and 10 um in zoomed area. ( e ) Representative western blot analysis of cofilin phosphorylation at Ser 3 in total cell extracts of HUVECs cultured for 48 h on TNC-coated or non-coated dishes. Equal amounts of protein (40 ug) were loaded in each lane and ERK1/2 was used as loading control. Fold change over NC (normalize
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 Induced HuR silencing suppresses PDA xenograft growth A. Representative images of subcutaneous Mia.sh290 tumors on the flanks of nude female mice, at the termination of the experiment (day 39). B. Tumor growth curves of Mia.sh290 xenografts. Mice were fed normal diet or 200 mg/kg DOX diet starting on day 0 (date of xenograft injection). Each data point represents the mean +- SEM ( n = 10 for -DOX group, and n = 8 for +DOX group). C. Plot of all tumor volumes on the final day of the Mia.sh290 xenograft experiment (day 39). Horizontal bars represent the median tumor volumes. D. Left , qPCR analysis of HuR mRNA expression in all Mia.sh290 xenografts harvested on day 39. 18S rRNA was used as normalization control. Data were plotted relative to the mean HuR mRNA expression in the -DOX group. Horizontal bars represent the medians. Right , plot of HuR mRNA expression vs. tumor volume for all Mia.sh290 tumors. Data were plotted relative to the mean of the -DOX group. E. Left , representative western blot of HuR protein expression in Mia.sh290 tumors harvested on day 39. Alpha-tubulin was used as normalization control. Right , quantification of the western blot. Each bar represents the mean +- SEM ( n = 3 per group). * = p < 0.05; ** = p < 0.01; **** = p < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 MS-444 inhibits HuR cytoplasmic localization A. HCT116 cells treated with 10muM MS-444 for 6 hr were subjected to HuR immunofluorescence analysis (shown in green). DAPI (shown in blue) was used to visualize nuclei. B. HCT116 cells were treated with 10muM MS-444 for the indicated times. Cytoplasmic and whole cell lysates were probed for HuR, along with cytoplasmic alpha-tubulin and nuclear Lamin A/C markers. C. RIE-iRas cells were untreated or with 5 mM IPTG for 24 hr to induce oncogenic Ras expression, followed by 10 muM MS-444 for 8 hr. HuR was detected by immunofluorescence (green) along with DAPI was used to visualize nuclei (merged images shown). D. RIE-iRas were grown in the presence or absence of 5 mM IPTG for 24 hr and then treated with indicated amounts of MS-444 for 48 hr. Relative cell survival was performed by MTT assay and is represented as average of 4 independent experiments +- SEM. E. YAMC and YAMC-Ras cells were grown under non-permissive conditions at 37degC and treated with 10 muM MS-444 for 8 hours. HuR localization was assayed by immunofluorescence. F. MTT assay of YAMC and YAMC-Ras treated with MS-444 for 48 hr under non-permissive conditions. Bars = 10 mum.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Klotho and alpha smooth muscle actin (alpha-SMA) expression by Western blot: ( a ) alpha-SMA and Klotho expression in renal tissue, normalized by alpha-tubulin in the CTL, AD, Post AD-CTL, and Post AD-Fish oil groups. Relative quantification of alpha-SMA ( b , c ) and Klotho ( d , e ) in the CTL, AD, Post AD-CTL and Post AD-Fish oil groups (** p < 0.01).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
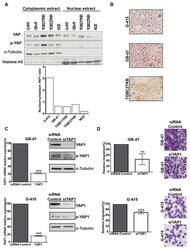
- Experimental details
- Figure 3 Knockdown of YAP1 by siRNA affects the migration capacities in GB-d1 and G-415 gallbladder cancer cell lines. ( A ) Cytoplasmic and nuclear protein expression of YAP1 and phospho-YAP1 in GBC cell lines by Western blot and estimation of the YAP1 nuclear/cytoplasmic ratio. Normalized expression of cytoplasmic phospho-YAP1 and nuclear YAP1 was quantified by densitometry. ( B ) Immunocytochemistry of YAP1 in GBC cell lines (Magnification: 20x). GB-d1 and G-415 cancer cells were transfected with 25nM of siRNA against YAP1 or siRNA non-target control; ( C ) the knockdown of YAP1 was validated by assessing mRNA and protein levels in GB-d1 G-415 cell lines at 24 h. YAP1 was quantified by qRT-PCR using QARS and TFCP2 as internal controls, while alpha-tubulin was used as an internal control for loading protein. ( D ) Migration analysis of GB-d1 and G-415 gallbladder cancer cells treated for 24 h with siRNA non-target control and siRNA YAP1, n = 3. ** p < 0.01 and *** p < 0.001 by one-sample t- test. Magnification: 20x.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 Differential expression of protein phosphatases 1 gamma 2 (PP1gamma2) in whole cell (WC), nuclear (NC) and cytosolic (CYT) fractions of cervical cancer HeLa cell. A - western blot showing PP1gamma2 protein expression in the WC, NC and CYT fractions of HeLa cells, B - densitometry analysis of the western blot confirmed the differential expression of PP1gamma2. beta-actin and Histone-3 were used as a loading control for the cytoplasmic and nuclear fractions, respectively. These results are presented as the mean +- SEM. of 3 independent experiments, ** p < 0.01; as compared to WC, C - sub-cellular localization of PP1gamma2 in HeLa cells showed predominant expression of the PP1gamma2 ( r ) in the nuclear region of the cell, while discrete patches were also observed in the cytoplasm. Blue (b) represents nuclear stain DAPI and green (g) staining shows tubulin expression. Zoom represents 4x magnification of the original. All images were captured using an oil immersion 63x objective lens
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 4 The ATS1 domains of T . gondii myosin H are necessary for its conoid localization. (A) MyoH-3Ty solubility was evaluated by fractionation after extraction in PBS, PBS/NaCl, PBS/Na2CO3 or PBS/Triton X-100. Their distribution in different fractions was assessed by western blot using anti-Ty antibodies. The profilin (PRF) was used as soluble control. (B) Deoxycholate (DOC) extraction shows that MycMyoH (upper panels) and DD-MyoH-NT-DeltaATS1 (middle panels) remain bound to the conoid (arrowhead). Actin polymerization using jasplakinolide at 1 muM (JAS) showed MycMyoH strongly associated to the conoid (arrowhead) and not to the actin-containing apical extensions (lower panels). Scale bar 2 mum. (C) Schematic representation of the TgMyoH and FKBP destabilization domain (DD) constructs. MyoH contains eight IQ motifs and three ATS1/RCC1 domains. NT: Neck and Tail, T: Tail. (D) Western blot analysis using anti-Myc antibodies shows stabilization of DD-MyoH-NT (100 kDa), DD-MyoH-T (64 kDa), and DD-MyoH-NT-DeltaATS1 (76 kDa) after 24 h of Shield-1 (Shld-1) treatment. Catalase (CAT) serves as loading control. (E) Only DD-MyoH-NT is present at the conoid. (F) Plaque assays performed with DD-MyoH-NT, DD-MyoH-T, and DD-MyoH-NT-DeltaATS1 were fixed after 7 days. A strong impairment in the lytic cycle was observed for DD-MyoH-NT and DD-MyoH-NT-DeltaATS1 in the presence of Shld-1.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
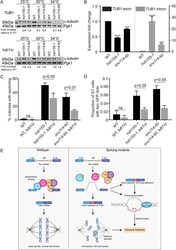
- Experimental details
- FIGURE 5: Tubulin stability contributes to genome maintenance in splicing mutants. (A) Western blot of relative alpha-tubulin protein levels. Top, TUB1 ; bottom, intronless TUB1 . (B) Quantification of TUB1 mRNA transcript levels from exon region (left) and TUB1 intron region (right) by RT-qPCR. Asterisks show p values of DeltaDeltaCt: *** p = 0.0002; **** p < 0.0001. (C) CTF phenotypes in TUB1 or tub1 Delta i strains. (D) Endogenous Chr III stability in hsh155-1 and snu114-60 with intronless TUB1 (gray bars). (C, D) Fisher's exact test. (E) Model of defective splicing-induced genome instability in yeast. (B, C, D) Mean values with SEM are shown, n = 3 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
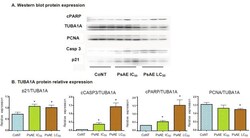
- Experimental details
- Figure 6 Western blot analysis of cultured B16-F0 cells. A. After 24 h, protein expression of control, IC 50 and LC 50 PsAE treated cells (0, 2.5 and 9 mug/mL, respectively) are presented. B. Relative expression of p21, caspase 3, PCNA and cPARP with respect to TUBA1A are expressed in terms of mean +- SEM of three assays by triplicated. Results were compared by one-way ANOVA followed by Fisher LSD test. *: indicate significant differences (p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 Western blot analysis of melanoma tumors. A. After 22 days of tumor induction, control and PsAE treated (150 mg/animal/day) groups are presented. B. Relative expression of p21 and PCNA over TUBA1A are expressed in terms of mean +- SEM. Results were compared by Student T test. *: indicates p = 0.0296. Full, non-adjusted images of p21 blot is provided in Supplementary Figure 5; TUBA1A (upper line) and PCNA (lower line) are provided in Supplementary Figure 6. Figure 7
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot