Antibody data
- Antibody Data
- Antigen structure
- References [82]
- Comments [0]
- Validations
- Western blot [3]
- Immunocytochemistry [2]
- Immunohistochemistry [1]
- Flow cytometry [1]
- Chromatin Immunoprecipitation [2]
- Other assay [67]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 51-1500 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- SMAD3 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 μg
- Concentration
- 0.25 mg/mL
- Storage
- -20°C
Submitted references Astrovirus-induced epithelial-mesenchymal transition via activated TGF-β increases viral replication.
Epstein-Barr virus nuclear antigen 3C (EBNA3C) interacts with the metabolism sensing C-terminal binding protein (CtBP) repressor to upregulate host genes.
Deletion of Akt1 Promotes Kidney Fibrosis in a Murine Model of Unilateral Ureteral Obstruction.
Differential Role of Smad2 and Smad3 in the Acquisition of an Endovascular Trophoblast-Like Phenotype and Preeclampsia.
Treatment of Hypertensive Heart Disease by Targeting Smad3 Signaling in Mice.
Dual deficiency of angiotensin-converting enzyme-2 and Mas receptor enhances angiotensin II-induced hypertension and hypertensive nephropathy.
Inhibition of TGF-β1 Signaling by IL-15: A Novel Role for IL-15 in the Control of Renal Epithelial-Mesenchymal Transition: IL-15 Counteracts TGF-β1-Induced EMT in Renal Fibrosis.
Relationship between ETS Transcription Factor ETV1 and TGF-β-regulated SMAD Proteins in Prostate Cancer.
TGF-β induces Smad2 Phosphorylation, ARE Induction, and Trophoblast Differentiation.
TGF-β Mediates Renal Fibrosis via the Smad3-Erbb4-IR Long Noncoding RNA Axis.
miR‑127 aggravates myocardial failure by promoting the TGF‑β1/Smad3 signaling.
Granulosa Cell Tumors: Novel Predictors of Recurrence in Early-stage Patients.
Restenosis Inhibition and Re-differentiation of TGFβ/Smad3-activated Smooth Muscle Cells by Resveratrol.
Fibulin-6 regulates pro-fibrotic TGF-β responses in neonatal mouse ventricular cardiac fibroblasts.
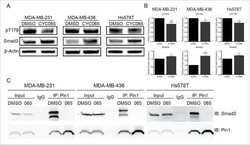
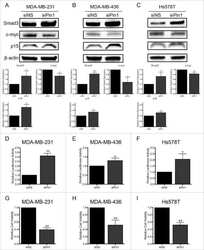
Inhibition of CDK-mediated Smad3 phosphorylation reduces the Pin1-Smad3 interaction and aggressiveness of triple negative breast cancer cells.
Dual tumor suppressing and promoting function of Notch1 signaling in human prostate cancer.
FOXL2, GATA4, and SMAD3 co-operatively modulate gene expression, cell viability and apoptosis in ovarian granulosa cell tumor cells.
TGF-β induction of FGF-2 expression in stromal cells requires integrated smad3 and MAPK pathways.
CEACAM1 long cytoplasmic domain isoform is associated with invasion and recurrence of hepatocellular carcinoma.
The inhibin/activin signalling pathway in human gonadal and adrenal cancers.
Improved detection reveals active β-papillomavirus infection in skin lesions from kidney transplant recipients.
A dynein motor attachment complex regulates TGFß/Smad3 signaling.
Transforming growth factor-β1 (TGF-β1) regulates cell junction restructuring via Smad-mediated repression and clathrin-mediated endocytosis of nectin-like molecule 2 (Necl-2).
Differential effects of fluticasone on extracellular matrix production by airway and parenchymal fibroblasts in severe COPD.
Genetic deletion of cell division autoantigen 1 retards diabetes-associated renal injury.
Alterations of Smad expression and activation in defining 2 subtypes of human head and neck squamous cell carcinoma.
TGF-β-Id1 signaling opposes Twist1 and promotes metastatic colonization via a mesenchymal-to-epithelial transition.
Expression of Wnt and TGF-β pathway components and key adrenal transcription factors in adrenocortical tumors: association to carcinoma aggressiveness.
Structure-function analyses of the human SIX1-EYA2 complex reveal insights into metastasis and BOR syndrome.
Non-Smad transforming growth factor-β signaling regulated by focal adhesion kinase binding the p85 subunit of phosphatidylinositol 3-kinase.
Distinctive mechanism for sustained TGF-β signaling and growth inhibition: MEK1 activation-dependent stabilization of type II TGF-β receptors.
Carcinoembryonic antigen interacts with TGF-{beta} receptor and inhibits TGF-{beta} signaling in colorectal cancers.
Reduction of transforming growth factor-beta type II receptor is caused by the enhanced ubiquitin-dependent degradation in human renal cell carcinoma.
Autocrine transforming growth factor-{beta}1 activation mediated by integrin {alpha}V{beta}3 regulates transcriptional expression of laminin-332 in Madin-Darby canine kidney epithelial cells.
SKI promotes Smad3 linker phosphorylations associated with the tumor-promoting trait of TGFbeta.
Salvianolic acid B prevents epithelial-to-mesenchymal transition through the TGF-beta1 signal transduction pathway in vivo and in vitro.
Bone morphogenetic protein-7 inhibits proximal tubular epithelial cell Smad3 signaling via increased SnoN expression.
SMAD3 and EGR1 physically and functionally interact in promoter-specific fashion.
Transforming growth factor beta promotes complexes between Smad proteins and the CCCTC-binding factor on the H19 imprinting control region chromatin.
Fuzheng Huayu recipe and vitamin E reverse renal interstitial fibrosis through counteracting TGF-beta1-induced epithelial-to-mesenchymal transition.
Plasminogen activator inhibitor-1 is a transcriptional target of the canonical pathway of Wnt/beta-catenin signaling.
Extracellular matrix-induced transforming growth factor-beta receptor signaling dynamics.
Interleukin-1 beta regulates proximal tubular cell transforming growth factor beta-1 signalling.
Erbin and the NF2 tumor suppressor Merlin cooperatively regulate cell-type-specific activation of PAK2 by TGF-beta.
A positive role for Myc in TGFbeta-induced Snail transcription and epithelial-to-mesenchymal transition.
SKI knockdown inhibits human melanoma tumor growth in vivo.
Smad ubiquitination regulatory factor-2 in the fibrotic kidney: regulation, target specificity, and functional implication.
Hepatocyte-specific Smad7 expression attenuates TGF-beta-mediated fibrogenesis and protects against liver damage.
VE-cadherin is a critical endothelial regulator of TGF-beta signalling.
Profibrogenic transforming growth factor-beta/activin receptor-like kinase 5 signaling via connective tissue growth factor expression in hepatocytes.
Localization of TGF-beta signaling intermediates Smad2, 3, 4, and 7 in developing and mature human and mouse kidney.
TGFbeta1 regulation of vimentin gene expression during differentiation of the C2C12 skeletal myogenic cell line requires Smads, AP-1 and Sp1 family members.
Modulation of tumor induction and progression of oncogenic K-ras-positive tumors in the presence of TGF- b1 haploinsufficiency.
Transforming growth factor beta activation of c-Abl is independent of receptor internalization and regulated by phosphatidylinositol 3-kinase and PAK2 in mesenchymal cultures.
Requirement of Smad3 and CREB-1 in mediating transforming growth factor-beta (TGF beta) induction of TGF beta 3 secretion.
BMP-7 opposes TGF-beta1-mediated collagen induction in mouse pulmonary myofibroblasts through Id2.
The mechanism of nuclear export of Smad3 involves exportin 4 and Ran.
Cutting edge: deficiency in the E3 ubiquitin ligase Cbl-b results in a multifunctional defect in T cell TGF-beta sensitivity in vitro and in vivo.
Cutting edge: deficiency in the E3 ubiquitin ligase Cbl-b results in a multifunctional defect in T cell TGF-beta sensitivity in vitro and in vivo.
Activin A mediates growth inhibition and cell cycle arrest through Smads in human breast cancer cells.
Ultraviolet irradiation induces Smad7 via induction of transcription factor AP-1 in human skin fibroblasts.
Repression of bone morphogenetic protein and activin-inducible transcription by Evi-1.
Smad7 abrogates transforming growth factor-beta1-mediated growth inhibition in COLO-357 cells through functional inactivation of the retinoblastoma protein.
Inactivation of TGF-beta signaling in hepatocytes results in an increased proliferative response after partial hepatectomy.
Induction by transforming growth factor-beta1 of epithelial to mesenchymal transition is a rare event in vitro.
Signaling "cross-talk" between TGF-beta1 and ECM signals in chondrocytic cells.
Imatinib mesylate inhibits the profibrogenic activity of TGF-beta and prevents bleomycin-mediated lung fibrosis.
Transforming growth factor beta-mediated transcriptional repression of c-myc is dependent on direct binding of Smad3 to a novel repressive Smad binding element.
Integration of TGF-beta/Smad and Jagged1/Notch signalling in epithelial-to-mesenchymal transition.
Differential trafficking of transforming growth factor-beta receptors and ligand in polarized epithelial cells.
Smad-binding defective mutant of transforming growth factor beta type I receptor enhances tumorigenesis but suppresses metastasis of breast cancer cell lines.
Identification of a gadd45beta 3' enhancer that mediates SMAD3- and SMAD4-dependent transcriptional induction by transforming growth factor beta.
Loss of Smad3 in acute T-cell lymphoblastic leukemia.
Involvement of c-Src kinase in the regulation of TGF-beta1-induced apoptosis.
Intracellular dynamics of Smad-mediated TGFbeta signaling.
Cellular response to hypoxia involves signaling via Smad proteins.
Cell-type-specific activation of PAK2 by transforming growth factor beta independent of Smad2 and Smad3.
Reduction in Smad2/3 signaling enhances tumorigenesis but suppresses metastasis of breast cancer cell lines.
Dynamic regulation of Smad expression during mesenchyme to epithelium transition in the metanephric kidney.
Rapamycin potentiates transforming growth factor beta-induced growth arrest in nontransformed, oncogene-transformed, and human cancer cells.
Rapamycin potentiates transforming growth factor beta-induced growth arrest in nontransformed, oncogene-transformed, and human cancer cells.
Requirement of Ras/MAPK pathway activation by transforming growth factor beta for transforming growth factor beta 1 production in a Smad-dependent pathway.
Hargest V, Bub T, Neale G, Schultz-Cherry S
PLoS pathogens 2022 Apr;18(4):e1009716
PLoS pathogens 2022 Apr;18(4):e1009716
Epstein-Barr virus nuclear antigen 3C (EBNA3C) interacts with the metabolism sensing C-terminal binding protein (CtBP) repressor to upregulate host genes.
Ohashi M, Hayes M, McChesney K, Johannsen E
PLoS pathogens 2021 Mar;17(3):e1009419
PLoS pathogens 2021 Mar;17(3):e1009419
Deletion of Akt1 Promotes Kidney Fibrosis in a Murine Model of Unilateral Ureteral Obstruction.
Kim IY, Lee MY, Park MW, Seong EY, Lee DW, Lee SB, Bae SS, Kim SS, Song SH
BioMed research international 2020;2020:6143542
BioMed research international 2020;2020:6143542
Differential Role of Smad2 and Smad3 in the Acquisition of an Endovascular Trophoblast-Like Phenotype and Preeclampsia.
Brkić J, Dunk C, Shan Y, O'Brien JA, Lye P, Qayyum S, Yang P, Matthews SG, Lye SJ, Peng C
Frontiers in endocrinology 2020;11:436
Frontiers in endocrinology 2020;11:436
Treatment of Hypertensive Heart Disease by Targeting Smad3 Signaling in Mice.
Meng J, Qin Y, Chen J, Wei L, Huang XR, Yu X, Lan HY
Molecular therapy. Methods & clinical development 2020 Sep 11;18:791-802
Molecular therapy. Methods & clinical development 2020 Sep 11;18:791-802
Dual deficiency of angiotensin-converting enzyme-2 and Mas receptor enhances angiotensin II-induced hypertension and hypertensive nephropathy.
Ni J, Yang F, Huang XR, Meng J, Chen J, Bader M, Penninger JM, Fung E, Yu XQ, Lan HY
Journal of cellular and molecular medicine 2020 Nov;24(22):13093-13103
Journal of cellular and molecular medicine 2020 Nov;24(22):13093-13103
Inhibition of TGF-β1 Signaling by IL-15: A Novel Role for IL-15 in the Control of Renal Epithelial-Mesenchymal Transition: IL-15 Counteracts TGF-β1-Induced EMT in Renal Fibrosis.
Devocelle A, Lecru L, François H, Desterke C, Gallerne C, Eid P, Estelle O, Azzarone B, Giron-Michel J
International journal of cell biology 2019;2019:9151394
International journal of cell biology 2019;2019:9151394
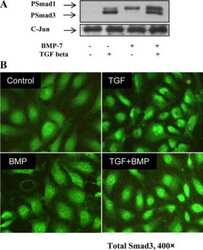
Relationship between ETS Transcription Factor ETV1 and TGF-β-regulated SMAD Proteins in Prostate Cancer.
Oh S, Shin S, Song H, Grande JP, Janknecht R
Scientific reports 2019 Jun 3;9(1):8186
Scientific reports 2019 Jun 3;9(1):8186
TGF-β induces Smad2 Phosphorylation, ARE Induction, and Trophoblast Differentiation.
Albers RE, Selesniemi K, Natale DRC, Brown TL
International journal of stem cells 2018 May 30;11(1):111-120
International journal of stem cells 2018 May 30;11(1):111-120
TGF-β Mediates Renal Fibrosis via the Smad3-Erbb4-IR Long Noncoding RNA Axis.
Feng M, Tang PM, Huang XR, Sun SF, You YK, Xiao J, Lv LL, Xu AP, Lan HY
Molecular therapy : the journal of the American Society of Gene Therapy 2018 Jan 3;26(1):148-161
Molecular therapy : the journal of the American Society of Gene Therapy 2018 Jan 3;26(1):148-161
miR‑127 aggravates myocardial failure by promoting the TGF‑β1/Smad3 signaling.
Xu H, Li F
Molecular medicine reports 2018 Dec;18(6):4839-4846
Molecular medicine reports 2018 Dec;18(6):4839-4846
Granulosa Cell Tumors: Novel Predictors of Recurrence in Early-stage Patients.
Sakr S, Abdulfatah E, Thomas S, Al-Wahab Z, Beydoun R, Morris R, Ali-Fehmi R, Bandyopadhyay S
International journal of gynecological pathology : official journal of the International Society of Gynecological Pathologists 2017 May;36(3):240-252
International journal of gynecological pathology : official journal of the International Society of Gynecological Pathologists 2017 May;36(3):240-252
Restenosis Inhibition and Re-differentiation of TGFβ/Smad3-activated Smooth Muscle Cells by Resveratrol.
Zhu Y, Takayama T, Wang B, Kent A, Zhang M, Binder BY, Urabe G, Shi Y, DiRenzo D, Goel SA, Zhou Y, Little C, Roenneburg DA, Shi XD, Li L, Murphy WL, Kent KC, Ke J, Guo LW
Scientific reports 2017 Feb 6;7:41916
Scientific reports 2017 Feb 6;7:41916
Fibulin-6 regulates pro-fibrotic TGF-β responses in neonatal mouse ventricular cardiac fibroblasts.
Chowdhury A, Hasselbach L, Echtermeyer F, Jyotsana N, Theilmeier G, Herzog C
Scientific reports 2017 Feb 17;7:42725
Scientific reports 2017 Feb 17;7:42725
Inhibition of CDK-mediated Smad3 phosphorylation reduces the Pin1-Smad3 interaction and aggressiveness of triple negative breast cancer cells.
Thomas AL, Lind H, Hong A, Dokic D, Oppat K, Rosenthal E, Guo A, Thomas A, Hamden R, Jeruss JS
Cell cycle (Georgetown, Tex.) 2017 Aug 3;16(15):1453-1464
Cell cycle (Georgetown, Tex.) 2017 Aug 3;16(15):1453-1464
Dual tumor suppressing and promoting function of Notch1 signaling in human prostate cancer.
Lefort K, Ostano P, Mello-Grand M, Calpini V, Scatolini M, Farsetti A, Dotto GP, Chiorino G
Oncotarget 2016 Jul 26;7(30):48011-48026
Oncotarget 2016 Jul 26;7(30):48011-48026
FOXL2, GATA4, and SMAD3 co-operatively modulate gene expression, cell viability and apoptosis in ovarian granulosa cell tumor cells.
Anttonen M, Pihlajoki M, Andersson N, Georges A, L'hôte D, Vattulainen S, Färkkilä A, Unkila-Kallio L, Veitia RA, Heikinheimo M
PloS one 2014;9(1):e85545
PloS one 2014;9(1):e85545
TGF-β induction of FGF-2 expression in stromal cells requires integrated smad3 and MAPK pathways.
Strand DW, Liang YY, Yang F, Barron DA, Ressler SJ, Schauer IG, Feng XH, Rowley DR
American journal of clinical and experimental urology 2014;2(3):239-48
American journal of clinical and experimental urology 2014;2(3):239-48
CEACAM1 long cytoplasmic domain isoform is associated with invasion and recurrence of hepatocellular carcinoma.
Kiriyama S, Yokoyama S, Ueno M, Hayami S, Ieda J, Yamamoto N, Yamaguchi S, Mitani Y, Nakamura Y, Tani M, Mishra L, Shively JE, Yamaue H
Annals of surgical oncology 2014 Dec;21 Suppl 4(0 4):S505-14
Annals of surgical oncology 2014 Dec;21 Suppl 4(0 4):S505-14
The inhibin/activin signalling pathway in human gonadal and adrenal cancers.
Marino FE, Risbridger G, Gold E
Molecular human reproduction 2014 Dec;20(12):1223-37
Molecular human reproduction 2014 Dec;20(12):1223-37
Improved detection reveals active β-papillomavirus infection in skin lesions from kidney transplant recipients.
Borgogna C, Lanfredini S, Peretti A, De Andrea M, Zavattaro E, Colombo E, Quaglia M, Boldorini R, Miglio U, Doorbar J, Bavinck JN, Quint KD, de Koning MN, Landolfo S, Gariglio M
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2014 Aug;27(8):1101-15
Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc 2014 Aug;27(8):1101-15
A dynein motor attachment complex regulates TGFß/Smad3 signaling.
Jin Q, Gao G, Mulder KM
International journal of biological sciences 2013;9(6):531-40
International journal of biological sciences 2013;9(6):531-40
Transforming growth factor-β1 (TGF-β1) regulates cell junction restructuring via Smad-mediated repression and clathrin-mediated endocytosis of nectin-like molecule 2 (Necl-2).
Gao Y, Lui WY
PloS one 2013;8(5):e64316
PloS one 2013;8(5):e64316
Differential effects of fluticasone on extracellular matrix production by airway and parenchymal fibroblasts in severe COPD.
Brandsma CA, Timens W, Jonker MR, Rutgers B, Noordhoek JA, Postma DS
American journal of physiology. Lung cellular and molecular physiology 2013 Oct 15;305(8):L582-9
American journal of physiology. Lung cellular and molecular physiology 2013 Oct 15;305(8):L582-9
Genetic deletion of cell division autoantigen 1 retards diabetes-associated renal injury.
Chai Z, Dai A, Tu Y, Li J, Wu T, Wang Y, Hale LJ, Koentgen F, Thomas MC, Cooper ME
Journal of the American Society of Nephrology : JASN 2013 Nov;24(11):1782-92
Journal of the American Society of Nephrology : JASN 2013 Nov;24(11):1782-92
Alterations of Smad expression and activation in defining 2 subtypes of human head and neck squamous cell carcinoma.
Xie W, Aisner S, Baredes S, Sreepada G, Shah R, Reiss M
Head & neck 2013 Jan;35(1):76-85
Head & neck 2013 Jan;35(1):76-85
TGF-β-Id1 signaling opposes Twist1 and promotes metastatic colonization via a mesenchymal-to-epithelial transition.
Stankic M, Pavlovic S, Chin Y, Brogi E, Padua D, Norton L, Massagué J, Benezra R
Cell reports 2013 Dec 12;5(5):1228-42
Cell reports 2013 Dec 12;5(5):1228-42
Expression of Wnt and TGF-β pathway components and key adrenal transcription factors in adrenocortical tumors: association to carcinoma aggressiveness.
Parviainen H, Schrade A, Kiiveri S, Prunskaite-Hyyryläinen R, Haglund C, Vainio S, Wilson DB, Arola J, Heikinheimo M
Pathology, research and practice 2013 Aug;209(8):503-9
Pathology, research and practice 2013 Aug;209(8):503-9
Structure-function analyses of the human SIX1-EYA2 complex reveal insights into metastasis and BOR syndrome.
Patrick AN, Cabrera JH, Smith AL, Chen XS, Ford HL, Zhao R
Nature structural & molecular biology 2013 Apr;20(4):447-53
Nature structural & molecular biology 2013 Apr;20(4):447-53
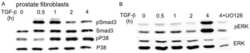
Non-Smad transforming growth factor-β signaling regulated by focal adhesion kinase binding the p85 subunit of phosphatidylinositol 3-kinase.
Hong M, Wilkes MC, Penheiter SG, Gupta SK, Edens M, Leof EB
The Journal of biological chemistry 2011 May 20;286(20):17841-50
The Journal of biological chemistry 2011 May 20;286(20):17841-50
Distinctive mechanism for sustained TGF-β signaling and growth inhibition: MEK1 activation-dependent stabilization of type II TGF-β receptors.
Chen G, Ghosh P, Longo DL
Molecular cancer research : MCR 2011 Jan;9(1):78-89
Molecular cancer research : MCR 2011 Jan;9(1):78-89
Carcinoembryonic antigen interacts with TGF-{beta} receptor and inhibits TGF-{beta} signaling in colorectal cancers.
Li Y, Cao H, Jiao Z, Pakala SB, Sirigiri DN, Li W, Kumar R, Mishra L
Cancer research 2010 Oct 15;70(20):8159-68
Cancer research 2010 Oct 15;70(20):8159-68
Reduction of transforming growth factor-beta type II receptor is caused by the enhanced ubiquitin-dependent degradation in human renal cell carcinoma.
Fukasawa H, Yamamoto T, Fujigaki Y, Misaki T, Ohashi N, Takayama T, Suzuki S, Mugiya S, Oda T, Uchida C, Kitagawa K, Hattori T, Hayashi H, Ozono S, Kitagawa M, Hishida A
International journal of cancer 2010 Oct 1;127(7):1517-25
International journal of cancer 2010 Oct 1;127(7):1517-25
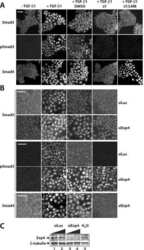
Autocrine transforming growth factor-{beta}1 activation mediated by integrin {alpha}V{beta}3 regulates transcriptional expression of laminin-332 in Madin-Darby canine kidney epithelial cells.
Moyano JV, Greciano PG, Buschmann MM, Koch M, Matlin KS
Molecular biology of the cell 2010 Nov 1;21(21):3654-68
Molecular biology of the cell 2010 Nov 1;21(21):3654-68
SKI promotes Smad3 linker phosphorylations associated with the tumor-promoting trait of TGFbeta.
Lin Q, Chen D, Timchenko NA, Medrano EE
Cell cycle (Georgetown, Tex.) 2010 May;9(9):1684-9
Cell cycle (Georgetown, Tex.) 2010 May;9(9):1684-9
Salvianolic acid B prevents epithelial-to-mesenchymal transition through the TGF-beta1 signal transduction pathway in vivo and in vitro.
Wang QL, Tao YY, Yuan JL, Shen L, Liu CH
BMC cell biology 2010 May 5;11:31
BMC cell biology 2010 May 5;11:31
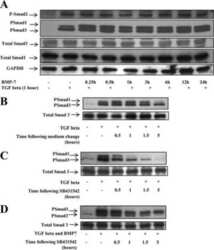
Bone morphogenetic protein-7 inhibits proximal tubular epithelial cell Smad3 signaling via increased SnoN expression.
Luo DD, Phillips A, Fraser D
The American journal of pathology 2010 Mar;176(3):1139-47
The American journal of pathology 2010 Mar;176(3):1139-47
SMAD3 and EGR1 physically and functionally interact in promoter-specific fashion.
Fortin J, Bernard DJ
Cellular signalling 2010 Jun;22(6):936-43
Cellular signalling 2010 Jun;22(6):936-43
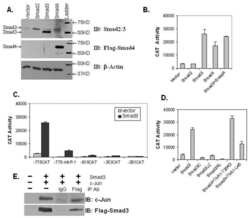
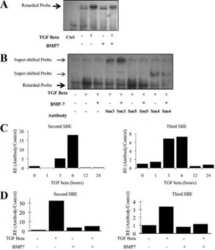
Transforming growth factor beta promotes complexes between Smad proteins and the CCCTC-binding factor on the H19 imprinting control region chromatin.
Bergström R, Savary K, Morén A, Guibert S, Heldin CH, Ohlsson R, Moustakas A
The Journal of biological chemistry 2010 Jun 25;285(26):19727-37
The Journal of biological chemistry 2010 Jun 25;285(26):19727-37
Fuzheng Huayu recipe and vitamin E reverse renal interstitial fibrosis through counteracting TGF-beta1-induced epithelial-to-mesenchymal transition.
Wang QL, Yuan JL, Tao YY, Zhang Y, Liu P, Liu CH
Journal of ethnopharmacology 2010 Feb 17;127(3):631-40
Journal of ethnopharmacology 2010 Feb 17;127(3):631-40
Plasminogen activator inhibitor-1 is a transcriptional target of the canonical pathway of Wnt/beta-catenin signaling.
He W, Tan R, Dai C, Li Y, Wang D, Hao S, Kahn M, Liu Y
The Journal of biological chemistry 2010 Aug 6;285(32):24665-75
The Journal of biological chemistry 2010 Aug 6;285(32):24665-75
Extracellular matrix-induced transforming growth factor-beta receptor signaling dynamics.
Garamszegi N, Garamszegi SP, Samavarchi-Tehrani P, Walford E, Schneiderbauer MM, Wrana JL, Scully SP
Oncogene 2010 Apr 22;29(16):2368-80
Oncogene 2010 Apr 22;29(16):2368-80
Interleukin-1 beta regulates proximal tubular cell transforming growth factor beta-1 signalling.
Luo DD, Fielding C, Phillips A, Fraser D
Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association 2009 Sep;24(9):2655-65
Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association 2009 Sep;24(9):2655-65
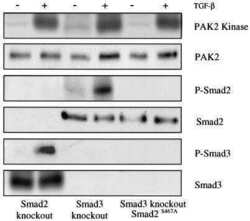
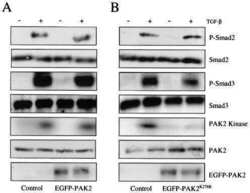
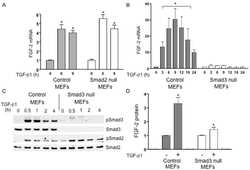
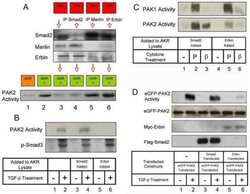
Erbin and the NF2 tumor suppressor Merlin cooperatively regulate cell-type-specific activation of PAK2 by TGF-beta.
Wilkes MC, Repellin CE, Hong M, Bracamonte M, Penheiter SG, Borg JP, Leof EB
Developmental cell 2009 Mar;16(3):433-44
Developmental cell 2009 Mar;16(3):433-44
A positive role for Myc in TGFbeta-induced Snail transcription and epithelial-to-mesenchymal transition.
Smith AP, Verrecchia A, Fagà G, Doni M, Perna D, Martinato F, Guccione E, Amati B
Oncogene 2009 Jan 22;28(3):422-30
Oncogene 2009 Jan 22;28(3):422-30
SKI knockdown inhibits human melanoma tumor growth in vivo.
Chen D, Lin Q, Box N, Roop D, Ishii S, Matsuzaki K, Fan T, Hornyak TJ, Reed JA, Stavnezer E, Timchenko NA, Medrano EE
Pigment cell & melanoma research 2009 Dec;22(6):761-72
Pigment cell & melanoma research 2009 Dec;22(6):761-72
Smad ubiquitination regulatory factor-2 in the fibrotic kidney: regulation, target specificity, and functional implication.
Tan R, He W, Lin X, Kiss LP, Liu Y
American journal of physiology. Renal physiology 2008 May;294(5):F1076-83
American journal of physiology. Renal physiology 2008 May;294(5):F1076-83
Hepatocyte-specific Smad7 expression attenuates TGF-beta-mediated fibrogenesis and protects against liver damage.
Dooley S, Hamzavi J, Ciuclan L, Godoy P, Ilkavets I, Ehnert S, Ueberham E, Gebhardt R, Kanzler S, Geier A, Breitkopf K, Weng H, Mertens PR
Gastroenterology 2008 Aug;135(2):642-59
Gastroenterology 2008 Aug;135(2):642-59
VE-cadherin is a critical endothelial regulator of TGF-beta signalling.
Rudini N, Felici A, Giampietro C, Lampugnani M, Corada M, Swirsding K, Garrè M, Liebner S, Letarte M, ten Dijke P, Dejana E
The EMBO journal 2008 Apr 9;27(7):993-1004
The EMBO journal 2008 Apr 9;27(7):993-1004
Profibrogenic transforming growth factor-beta/activin receptor-like kinase 5 signaling via connective tissue growth factor expression in hepatocytes.
Weng HL, Ciuclan L, Liu Y, Hamzavi J, Godoy P, Gaitantzi H, Kanzler S, Heuchel R, Ueberham U, Gebhardt R, Breitkopf K, Dooley S
Hepatology (Baltimore, Md.) 2007 Oct;46(4):1257-70
Hepatology (Baltimore, Md.) 2007 Oct;46(4):1257-70
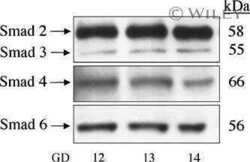
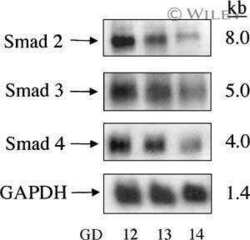
Localization of TGF-beta signaling intermediates Smad2, 3, 4, and 7 in developing and mature human and mouse kidney.
Banas MC, Parks WT, Hudkins KL, Banas B, Holdren M, Iyoda M, Wietecha TA, Kowalewska J, Liu G, Alpers CE
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2007 Mar;55(3):275-85
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 2007 Mar;55(3):275-85
TGFbeta1 regulation of vimentin gene expression during differentiation of the C2C12 skeletal myogenic cell line requires Smads, AP-1 and Sp1 family members.
Wu Y, Zhang X, Salmon M, Lin X, Zehner ZE
Biochimica et biophysica acta 2007 Mar;1773(3):427-39
Biochimica et biophysica acta 2007 Mar;1773(3):427-39
Modulation of tumor induction and progression of oncogenic K-ras-positive tumors in the presence of TGF- b1 haploinsufficiency.
Pandey J, Umphress SM, Kang Y, Angdisen J, Naumova A, Mercer KL, Jacks T, Jakowlew SB
Carcinogenesis 2007 Dec;28(12):2589-96
Carcinogenesis 2007 Dec;28(12):2589-96
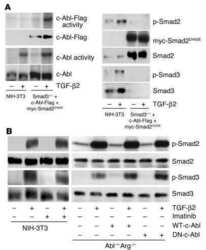
Transforming growth factor beta activation of c-Abl is independent of receptor internalization and regulated by phosphatidylinositol 3-kinase and PAK2 in mesenchymal cultures.
Wilkes MC, Leof EB
The Journal of biological chemistry 2006 Sep 22;281(38):27846-54
The Journal of biological chemistry 2006 Sep 22;281(38):27846-54
Requirement of Smad3 and CREB-1 in mediating transforming growth factor-beta (TGF beta) induction of TGF beta 3 secretion.
Liu G, Ding W, Neiman J, Mulder KM
The Journal of biological chemistry 2006 Oct 6;281(40):29479-90
The Journal of biological chemistry 2006 Oct 6;281(40):29479-90
BMP-7 opposes TGF-beta1-mediated collagen induction in mouse pulmonary myofibroblasts through Id2.
Izumi N, Mizuguchi S, Inagaki Y, Saika S, Kawada N, Nakajima Y, Inoue K, Suehiro S, Friedman SL, Ikeda K
American journal of physiology. Lung cellular and molecular physiology 2006 Jan;290(1):L120-6
American journal of physiology. Lung cellular and molecular physiology 2006 Jan;290(1):L120-6
The mechanism of nuclear export of Smad3 involves exportin 4 and Ran.
Kurisaki A, Kurisaki K, Kowanetz M, Sugino H, Yoneda Y, Heldin CH, Moustakas A
Molecular and cellular biology 2006 Feb;26(4):1318-32
Molecular and cellular biology 2006 Feb;26(4):1318-32
Cutting edge: deficiency in the E3 ubiquitin ligase Cbl-b results in a multifunctional defect in T cell TGF-beta sensitivity in vitro and in vivo.
Wohlfert EA, Gorelik L, Mittler R, Flavell RA, Clark RB
Journal of immunology (Baltimore, Md. : 1950) 2006 Feb 1;176(3):1316-20
Journal of immunology (Baltimore, Md. : 1950) 2006 Feb 1;176(3):1316-20
Cutting edge: deficiency in the E3 ubiquitin ligase Cbl-b results in a multifunctional defect in T cell TGF-beta sensitivity in vitro and in vivo.
Wohlfert EA, Gorelik L, Mittler R, Flavell RA, Clark RB
Journal of immunology (Baltimore, Md. : 1950) 2006 Feb 1;176(3):1316-20
Journal of immunology (Baltimore, Md. : 1950) 2006 Feb 1;176(3):1316-20
Activin A mediates growth inhibition and cell cycle arrest through Smads in human breast cancer cells.
Burdette JE, Jeruss JS, Kurley SJ, Lee EJ, Woodruff TK
Cancer research 2005 Sep 1;65(17):7968-75
Cancer research 2005 Sep 1;65(17):7968-75
Ultraviolet irradiation induces Smad7 via induction of transcription factor AP-1 in human skin fibroblasts.
Quan T, He T, Voorhees JJ, Fisher GJ
The Journal of biological chemistry 2005 Mar 4;280(9):8079-85
The Journal of biological chemistry 2005 Mar 4;280(9):8079-85
Repression of bone morphogenetic protein and activin-inducible transcription by Evi-1.
Alliston T, Ko TC, Cao Y, Liang YY, Feng XH, Chang C, Derynck R
The Journal of biological chemistry 2005 Jun 24;280(25):24227-37
The Journal of biological chemistry 2005 Jun 24;280(25):24227-37
Smad7 abrogates transforming growth factor-beta1-mediated growth inhibition in COLO-357 cells through functional inactivation of the retinoblastoma protein.
Boyer Arnold N, Korc M
The Journal of biological chemistry 2005 Jun 10;280(23):21858-66
The Journal of biological chemistry 2005 Jun 10;280(23):21858-66
Inactivation of TGF-beta signaling in hepatocytes results in an increased proliferative response after partial hepatectomy.
Romero-Gallo J, Sozmen EG, Chytil A, Russell WE, Whitehead R, Parks WT, Holdren MS, Her MF, Gautam S, Magnuson M, Moses HL, Grady WM
Oncogene 2005 Apr 21;24(18):3028-41
Oncogene 2005 Apr 21;24(18):3028-41
Induction by transforming growth factor-beta1 of epithelial to mesenchymal transition is a rare event in vitro.
Brown KA, Aakre ME, Gorska AE, Price JO, Eltom SE, Pietenpol JA, Moses HL
Breast cancer research : BCR 2004;6(3):R215-31
Breast cancer research : BCR 2004;6(3):R215-31
Signaling "cross-talk" between TGF-beta1 and ECM signals in chondrocytic cells.
Schneiderbauer MM, Dutton CM, Scully SP
Cellular signalling 2004 Oct;16(10):1133-40
Cellular signalling 2004 Oct;16(10):1133-40
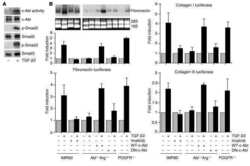
Imatinib mesylate inhibits the profibrogenic activity of TGF-beta and prevents bleomycin-mediated lung fibrosis.
Daniels CE, Wilkes MC, Edens M, Kottom TJ, Murphy SJ, Limper AH, Leof EB
The Journal of clinical investigation 2004 Nov;114(9):1308-16
The Journal of clinical investigation 2004 Nov;114(9):1308-16
Transforming growth factor beta-mediated transcriptional repression of c-myc is dependent on direct binding of Smad3 to a novel repressive Smad binding element.
Frederick JP, Liberati NT, Waddell DS, Shi Y, Wang XF
Molecular and cellular biology 2004 Mar;24(6):2546-59
Molecular and cellular biology 2004 Mar;24(6):2546-59
Integration of TGF-beta/Smad and Jagged1/Notch signalling in epithelial-to-mesenchymal transition.
Zavadil J, Cermak L, Soto-Nieves N, Böttinger EP
The EMBO journal 2004 Mar 10;23(5):1155-65
The EMBO journal 2004 Mar 10;23(5):1155-65
Differential trafficking of transforming growth factor-beta receptors and ligand in polarized epithelial cells.
Murphy SJ, Doré JJ, Edens M, Coffey RJ, Barnard JA, Mitchell H, Wilkes M, Leof EB
Molecular biology of the cell 2004 Jun;15(6):2853-62
Molecular biology of the cell 2004 Jun;15(6):2853-62
Smad-binding defective mutant of transforming growth factor beta type I receptor enhances tumorigenesis but suppresses metastasis of breast cancer cell lines.
Tian F, Byfield SD, Parks WT, Stuelten CH, Nemani D, Zhang YE, Roberts AB
Cancer research 2004 Jul 1;64(13):4523-30
Cancer research 2004 Jul 1;64(13):4523-30
Identification of a gadd45beta 3' enhancer that mediates SMAD3- and SMAD4-dependent transcriptional induction by transforming growth factor beta.
Major MB, Jones DA
The Journal of biological chemistry 2004 Feb 13;279(7):5278-87
The Journal of biological chemistry 2004 Feb 13;279(7):5278-87
Loss of Smad3 in acute T-cell lymphoblastic leukemia.
Wolfraim LA, Fernandez TM, Mamura M, Fuller WL, Kumar R, Cole DE, Byfield S, Felici A, Flanders KC, Walz TM, Roberts AB, Aplan PD, Balis FM, Letterio JJ
The New England journal of medicine 2004 Aug 5;351(6):552-9
The New England journal of medicine 2004 Aug 5;351(6):552-9
Involvement of c-Src kinase in the regulation of TGF-beta1-induced apoptosis.
Park SS, Eom YW, Kim EH, Lee JH, Min DS, Kim S, Kim SJ, Choi KS
Oncogene 2004 Aug 19;23(37):6272-81
Oncogene 2004 Aug 19;23(37):6272-81
Intracellular dynamics of Smad-mediated TGFbeta signaling.
Greene RM, Nugent P, Mukhopadhyay P, Warner DR, Pisano MM
Journal of cellular physiology 2003 Nov;197(2):261-71
Journal of cellular physiology 2003 Nov;197(2):261-71
Cellular response to hypoxia involves signaling via Smad proteins.
Zhang H, Akman HO, Smith EL, Zhao J, Murphy-Ullrich JE, Batuman OA
Blood 2003 Mar 15;101(6):2253-60
Blood 2003 Mar 15;101(6):2253-60
Cell-type-specific activation of PAK2 by transforming growth factor beta independent of Smad2 and Smad3.
Wilkes MC, Murphy SJ, Garamszegi N, Leof EB
Molecular and cellular biology 2003 Dec;23(23):8878-89
Molecular and cellular biology 2003 Dec;23(23):8878-89
Reduction in Smad2/3 signaling enhances tumorigenesis but suppresses metastasis of breast cancer cell lines.
Tian F, DaCosta Byfield S, Parks WT, Yoo S, Felici A, Tang B, Piek E, Wakefield LM, Roberts AB
Cancer research 2003 Dec 1;63(23):8284-92
Cancer research 2003 Dec 1;63(23):8284-92
Dynamic regulation of Smad expression during mesenchyme to epithelium transition in the metanephric kidney.
Oxburgh L, Robertson EJ
Mechanisms of development 2002 Mar;112(1-2):207-11
Mechanisms of development 2002 Mar;112(1-2):207-11
Rapamycin potentiates transforming growth factor beta-induced growth arrest in nontransformed, oncogene-transformed, and human cancer cells.
Law BK, Chytil A, Dumont N, Hamilton EG, Waltner-Law ME, Aakre ME, Covington C, Moses HL
Molecular and cellular biology 2002 Dec;22(23):8184-98
Molecular and cellular biology 2002 Dec;22(23):8184-98
Rapamycin potentiates transforming growth factor beta-induced growth arrest in nontransformed, oncogene-transformed, and human cancer cells.
Law BK, Chytil A, Dumont N, Hamilton EG, Waltner-Law ME, Aakre ME, Covington C, Moses HL
Molecular and cellular biology 2002 Dec;22(23):8184-98
Molecular and cellular biology 2002 Dec;22(23):8184-98
Requirement of Ras/MAPK pathway activation by transforming growth factor beta for transforming growth factor beta 1 production in a Smad-dependent pathway.
Yue J, Mulder KM
The Journal of biological chemistry 2000 Oct 6;275(40):30765-73
The Journal of biological chemistry 2000 Oct 6;275(40):30765-73
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of SMAD3 was performed by loading 35 µg of PC-3 (lane1), U-87 MG (lane2) and a549 (lane3) cell lysate using Novex®NuPAGE® 12 % Bis-Tris gel (Product # NP0341BOX), XCell SureLock Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (LC5800), and iBlot® Dry Blotting System (IB21001). Proteins were transferred to a nitrocellulose membrane and blocked with 5% skim milk for 1 hour at room temperature. SMAD3 was detected at ~ 52 kDa using SMAD3 Rabbit Polyclonal Antibody (Product # 51-1500) at 1-3 µg/mL in 5% skim milk at 4°C overnight on a rocking platform. Goat Anti-Rabbit IgG - HRP Secondary Antibody (G21234) at 1:5000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
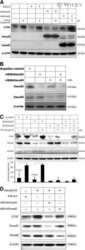
- Knockdown of SMAD3 was achieved by transfecting A549 with SMAD3 specific siRNAs (Silencer® select Product # s8400). Western blot analysis (Fig. a) was performed using whole cell extracts from the SMAD3 knockdown cells (lane 3), non-specific scrambled siRNA transfected cells (lane 2) and untransfected cells (lane 1). The blots were probed with SMAD3 Polyclonal Antibody (Product # 51-1500, 1 µg/mL) and Goat anti-Rabbit IgG (Heavy Chain) Superclonal™ Secondary Antibody, HRP conjugate (Product # A27036, 0.25 µg/mL, 1:4000 dilution). Densitometric analysis of this western blot is shown in histogram (Fig. b). Absence of signal upon siRNA mediated knock down confirms that antibody is specific to SMAD3.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of SMAD3 (Fig. a) was performed by loading 20 µg of HeLa Control (lane 1), HeLa SMAD3 knockout (lane 2) whole cell extracts. SMAD3 was detected at 50 kDa using SMAD3 polyclonal Antibody (Product # 51-1500, 1µg/mL) and Goat anti-Rabbit IgG (Heavy Chain) Superclonal™ Secondary Antibody HRP conjugate (Product # A27036, 1:4000 dilution). Densitometric analysis of this western blot is shown in histogram (Fig. b). Loss of signal in CRISPR mediated knockout (KO) confirms that antibody is specific to SMAD3.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
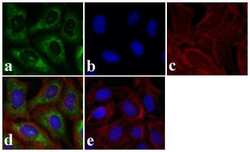
- Experimental details
- Immunofluorescence analysis of SMAD3 was done on 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with SMAD3 Rabbit polyclonal Antibody (Product # 51-1500) at 2 µg/mL in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Goat Anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing cytoplasmic localization. Panel e shows no primary antibody control. The images were captured at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
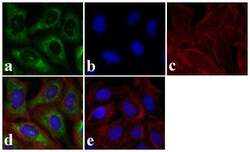
- Experimental details
- Immunofluorescence analysis of SMAD3 was done on 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with SMAD3 Rabbit polyclonal Antibody (Product # 51-1500) at 2 µg/mL in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Goat Anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing cytoplasmic localization. Panel e shows no primary antibody control. The images were captured at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
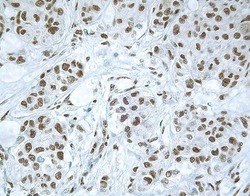
- Experimental details
- Immunohistochemical staining of human breast carcinoma using Rb anti-Smad3 (PAD: LPC3) (Product # 51-1500).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
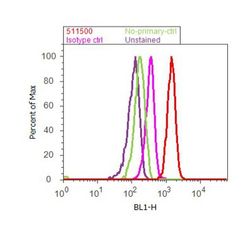
- Experimental details
- Flow cytometry analysis of SMAD3 was done on A549 cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Tritonª X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with SMAD3 Rabbit Polyclonal Antibody (511500, red histogram) or with rabbit isotype control (pink histogram) at 3-5 µg/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor¨ 488 Goat Anti-Rabbit Secondary Antibody (A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune¨ Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
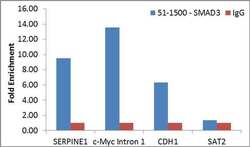
- Experimental details
- Enrichment of endogenous SMAD3 protein at specific gene loci using Anti-SMAD3 Antibody: Chromatin Immunoprecipitation (ChIP) was performed using Anti-SMAD3 Rabbit Polyclonal Antibody (Product # 51-1500, 4 µg) on sheared chromatin from 2 million MCF7 cells treated with TGFb (7 ng/mL for 1 hour) using the MAGnify ChIP system kit (Product # 49-2024). Normal Rabbit IgG was used as a negative IP control. The purified DNA was analyzed by qPCR with PCR primer pairs over SERPINE1, c-Myc Intron 1 and CDH1 (active) and SAT2 satellite repeats (inactive). Data is presented as fold enrichment of the antibody signal versus the negative control IgG using the comparative CT method.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
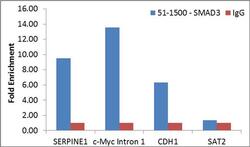
- Experimental details
- Enrichment of endogenous SMAD3 protein at specific gene loci using Anti-SMAD3 Antibody: Chromatin Immunoprecipitation (ChIP) was performed using Anti-SMAD3 Rabbit Polyclonal Antibody (Product # 51-1500, 4 µg) on sheared chromatin from 2 million MCF7 cells treated with TGFb (7 ng/mL for 1 hour) using the MAGnify ChIP system kit (Product # 49-2024). Normal Rabbit IgG was used as a negative IP control. The purified DNA was analyzed by qPCR with PCR primer pairs over SERPINE1, c-Myc Intron 1 and CDH1 (active) and SAT2 satellite repeats (inactive). Data is presented as fold enrichment of the antibody signal versus the negative control IgG using the comparative CT method.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
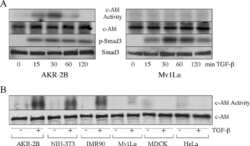
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
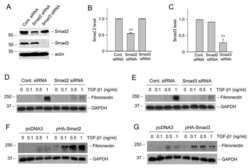
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
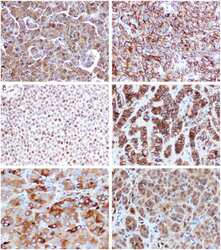
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
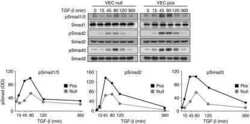
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
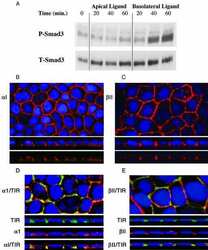
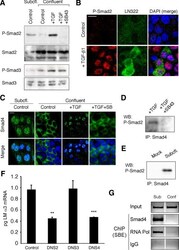
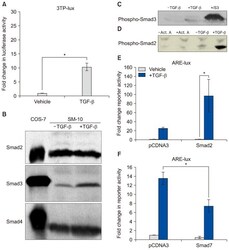
- Figure 4. Phospho-Smad2 and Smad4 regulate LM alpha3 subunit gene transcription. (A) Differential phosphorylation of Smads. Subconfluent (Subcfl.) or confluent cultures either untreated (control) or treated with 5 ng/ml TGF-beta1 for 6 h in the absence or presence of the TbetaR-I inhibitor SB431542 (+TGF-beta1 or + TGF-beta1+SB43, respectively) were analyzed by Western blotting for phospho-Smad2 (P-Smad2), total Smad2, P-Smad3, and total Smad3. (B) Phospho-Smad2 is localized to the nuclei after TGF-beta1 treatment in confluent cells. Untreated (control) or TGF-beta1-treated confluent cells (+TGF-beta1) were stained with antibodies against P-Smad2 (red) and LM-332 (beta3 subunit; green) and analyzed by confocal fluorescence microscopy. Nuclear staining with DAPI, blue. Bar, 10 mum. (C) Smad 4 is also localized to the nuclei in subconfluent and TGF-beta1-treated confluent cells. Subconfluent MDCK cell cultures without added exogenous TGF-beta1 and confluent cultures either untreated (control) or treated with of TGF-beta1 in the presence or absence of SB431542 (SB) were stained with antibodies against Smad4 (green) and analyzed by confocal fluorescence microscopy. Nuclear staining with DAPI, blue. Bar, 10 mum. (D) Phosho-Smad2 and Smad4 form a complex dependent on TbetaR-I signaling. Confluent cultures treated with TGF-beta1 to induce LM-332 expression in the absence or presence of the TbetaR-I inhibitor SB431542 (+TGF or +TGF+SB43, respectively) were extracted in RIPA buffer. E
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
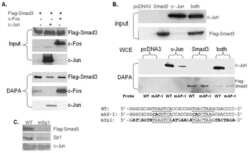
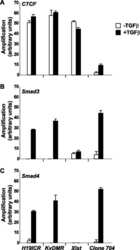
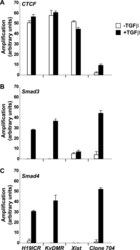
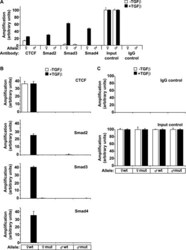
- FIGURE 6. CTCF and Smads co-occupy CTCF-binding loci such as the H19 ICR. ChIP-Q-PCR analysis of four genomic fragments corresponding to three known imprinted loci, H19 ICR, KvDMR , and Xist , and a control intergenic region, clone 704 . ChIP was performed in NMuMG cells stimulated with TGFbeta1 for 4 h using specific antibodies against CTCF ( A ), Smad3 ( B ), and Smad4 ( C ). Q-PCR data show the average values and standard deviations calculated from triplicate determinations.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
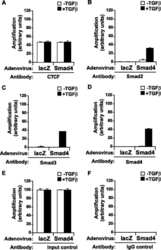
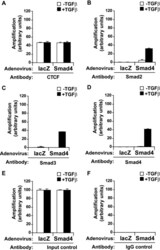
- FIGURE 7. Co-occupation of the H19 ICR by CTCF and Smads requires Smad4. A-D , MDA-MB-468 cells that lack Smad4 were infected with adenoviruses expressing control, LacZ ( left bars ), or wild type Smad4 ( right bars ) and were stimulated with TGFbeta1 for 4 h (+) or left untreated (-) prior to ChIP with specific antibodies for CTCF ( A ), Smad2 ( B ), Smad3 ( C ), and Smad4 ( D ). Immunoprecipitated chromatin was amplified with primers specific for the H19 ICR. E and F , input chromatin controls ( E ) and nonspecific IgG controls ( F ) are also shown after PCR amplification for the same samples analyzed in A-D .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
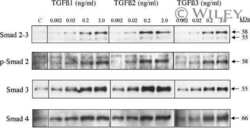
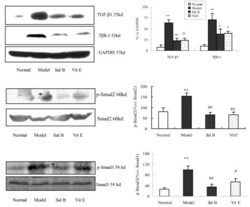
- Figure 4 The Effect of Sal B on TGF-beta1, TbetaRI, Smad2, p-Smad2 and p-Smad3 expression in kidneys of model rats . Western blot analysis showed significantly increased TGF-beta1, TbetaR-I, p-Smad2 and p-Smad3 expressions in rats of the model group. In contrast, Sal B treatment attenuated HgCl 2 -induced upregulation of TGF-beta1, TbetaR-I, p-Smad2 and p-Smad3 expressions. The values are represented as the density of TGF-beta1 or TbetaR-I vs . GAPDH (%), or p-Smad2 vs . Smad2 (%), or p-Smad3 vs . Smad3 (%). ** P < 0.01 vs . normal; * P < 0.05 vs . normal; ## P < 0.01 vs . model; # P < 0.05 vs . model.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
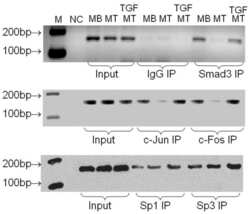
- Figure 4 Effect of TGF-beta1 on Necl-2 mRNA stability and promoter activity. (A) and (B) Analysis of Necl-2 mRNA stability was performed by actinomycin D (ActD) assay. Cells were pre-treated with ActD (5 ug/ml) for 2 h before vehicle or TGF-beta1 treatment. RT-PCR (A) and real-time PCR (B) were performed to determine Necl-2 mRNA level. (C) Progressive 5'-deletion analysis of mouse Necl-2 promoter was performed between nt -502 and -1. A series 5'-deletion constructs and pEGFP vector were co-transfected into GC-1 spg cells followed by TGF-beta1 treatment (5 ng/ml, 18 h). (D) Three putative cis -acting elements including MyoD, CCAATa and CCAATb motifs are located within the region between nt -159 and -1 (upper panel). Site-directed mutagenic constructs containing single, double or triple mutations and pEGFP vector were co-transfected into GC-1 spg cells followed by TGF-beta1 treatment. pEGFP activity was used to normalize transfection efficiency. Promoter activity was represented as the fold change when compared with pGL-3 vector. (E) pGL-3 vector, p(-159/-1)Luc and pEGFP vector were co-transfected with si-Smad3 (#1/#2, 20 nM) or/and si-Smad4 (#1/#2, 20 nM) for 48 h followed by TGF-beta1 treatment. pEGFP activity was used to normalize transfection efficiency. Smad3 and Smad4 protein levels were examined by Western blotting. (F) Wild-type and single mutated constructs of p(-159/-1)Luc were co-transfected with pcDNA3.1 vector, Smad3 or/and Smad4 expression constructs int
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
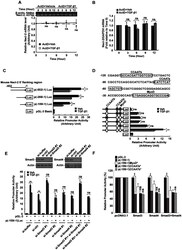
- Fig. 4 TGF- beta triggers Smad2 phosphorylation and transactivation. (A) 3TP luciferase reporter activity in SM10s cells treated with vehicle control or TGF- beta 2. Cells were transiently transfected with 3TP-lux and pRLSV40 using Metafectene. Twenty-four hours post-transfection, cells were treated with vehicle or TGF- beta 2 (5 ng/ml) for 72 hours. Luciferase activity was analyzed using the dual luciferase assay system. The 3TP-lux transactivation values were normalized to the constitutively active reporter (pRLSV40). (B) Western blotting analysis of Smad2, Smad3, and Smad4 in TGF- beta 2 or vehicle-treated SM10 cells. COS-7 cells were used as a positive control. (C) Western blotting analysis of Phospho-Smad3 in TGF- beta 2 or vehicle-treated SM10 cells. COS-7 cells transiently transfected with the plasmid, pXIX-Smad3 and treated with TGF- beta were used as a positive control. (D) Western blotting analysis of Phospho-Smad2 in the TGF- beta 2, Activin A, or vehicle-treated SM10 cells. (E) ARE luciferase reporter activity in SM10 cells transiently transfected with pCDNA3 control or pRK5-Smad2 and treated with vehicle control or TGF- beta 2. Cells were transiently transfected with ARE-lux, pLv-CMV-hFAST-1, and pRLSV40 using Metafectene. Twenty-four hours post-transfection, cells were treated with vehicle or TGF- beta 2 (5 ng/ml) for 72 hours. Luciferase activity was analyzed using the dual luciferase assay system. The ARE-lux transactivation values were normalized to the const
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 4. miR-127 regulates the expression of TGF-beta1 and Smad3. (A) Reverse transcription-quantitative polymerase chain reaction detected the expression of miR-127 after siRNA transfection. (B) Western blot analysis demonstrated alterations in TGF-beta1 and Smad3 in the H9C2 cells treated with 0, 2.5, 5 and 10 uM si-mR-127. (C) Immunofluorescence indicated that the TGF-beta1 and Smad3 signaling pathway was inhibited when miR-127 was inhibited by si-miR-127. ***P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
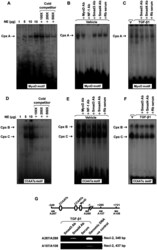
- Figure 5 EMSA of MyoD and CCAATa motifs and ChIP assay. (A) and (D) Dose-dependent and competition assay for EMSA of MyoD motif (A) and CCAATa motif (D). Double stranded oligonucleotides containing the respective motif were radiolabeled with [gamma- 32 P]ATP and incubated with nuclear extract (1-15 ug) alone or in the presence of cold competitors (100- to 500-fold excess). (B-C) and (E-F) Labeled probes were incubated with vehicle or TGF-beta1 treated nuclear extract (15 ug for MyoD motif and 10 ug for CCAATa motif) in the presence of specified antibodies or rabbit serum (Rb serum). (G) A schematic drawing illustrating the relative location of MyoD and CCAAT cis -acting motifs in the Necl-2 promoter and chromatin immunoprecipitation assay. TGF-beta1-treated genomic DNA-protein samples were immunopreciptated with antibodies against Smad3 and Smad4 (2 ug) or rabbit serum. Precipitated DNA-protein complexes were subjected to DNA purification. The promoter region and the open reading frame of Necl-2 gene were amplified using specific primer pairs no. A287/A288 (for promoter region) and no. A107/A108 (for open reading frame), respectively, followed by agarose gel electrophoresis. Rb, Rabbit. Cpx, complex.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
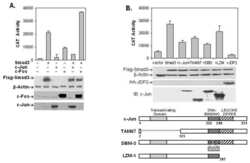
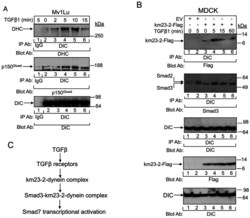
- Fig 5 TGFss regulates the tethering of the specific km23-2-Smad3 cargo to the other dynein components. A: MDCK cells were incubated in SF medium for 1h prior to addition of TGFss1 (5 ng/ml) for the indicated times. Cell lysates were subjected to IP using a monoclonal anti-DIC Ab and were separated by 4-12% NuPAGE, followed by immunoblot analysis using an anti-DHC Ab (top panel) or an anti-p150 Ab (2nd panel). The same membrane was re-blotted with anti-DIC (bottom panel) to show equal protein loading. The results shown are representative of three similar experiments. B: MDCK cells were transiently transfected with either EV or km23-2-Flag as indicated. 24h after transfection, cells were incubated in SF medium for 1h prior to addition of TGFss1 (5 ng/ml) for the indicated times. Cell lysates were subjected to IP using a monoclonal anti-DIC Ab and were separated by 4-12% NuPAGE, followed by immunoblot analysis using an anti-Flag Ab (top panel) and an anti-Smad3 Ab (2nd panel). The same membrane was re-blotted with anti-DIC (3rd panel) to show equal protein loading. Western blot analysis (two bottom panels) with anti-Flag or anti-DIC demonstrates equal protein expression of km23-2 and DIC. The results shown are representative of two similar experiments. C: A schematic diagram illustrating the role of km23-1 in Smad3-dependent Smad7 transcriptional activation after TGFss stimulation.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
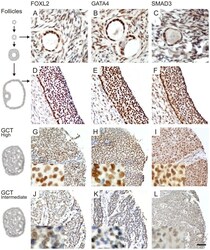
- Figure 4 FOXL2, GATA4, and SMAD3 proteins are expressed in normal follicles and GCTs. Proliferating granulosa cells of primary (A-C) and antral (D-F) follicles express high levels of the transcription factors FOXL2, GATA4 and SMAD3. For GCTs, High- (G-I) and intermediate-level (J-L) expressing examples are shown. FOXL2 and GATA4 expression localized into the nucleus, while low levels of SMAD3 expression was also detected in the cytoplasm. Higher magnifications are shown in insets. Scale bar = 100 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
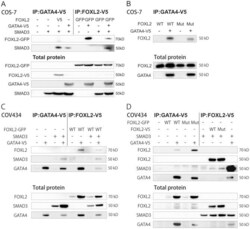
- Figure 1 FOXL2, GATA4, and SMAD3 interaction in the transcription complex. Both COS-7 and COV434 cells were transfected for 48 h. All cell lysates were immunoprecipitated using V5 epitope. A) COS-7 cells transfected with V5 tagged GATA4, V5 or GFP tagged wild type (WT) FOXL2 or SMAD3 overexpression vectors as indicated. B) COS-7 cells transfected with V5 tagged GATA4 and untagged wild type (WT) or C134W mutated (MUT) FOXL2 overexpression vectors. C) COV434 cells transfected with V5 tagged GATA4, GFP tagged wild type FOXL2 or SMAD3 overexpression vectors. D) COV434 cells transfected with V5 tagged GATA4, SMAD3 and wild type or C134W mutated FOXL2 overexpression vectors. Immunoprecipitated proteins were detected using antibodies against FOXL2, SMAD3 and GATA4. Total proteins are shown as controls for transfections and were detected using antibodies against FOXL2, GATA4 and SMAD3. Similar results were obtained in at least three independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
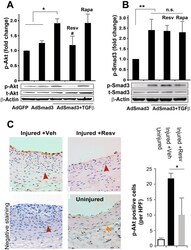
- Figure 7 Inhibitory effect of resveratrol on TGFbeta/Smad3-stimulated Akt activation in vitro and in vivo in balloon-injured rat carotid arteries. ( A , B ) Rat aortic SMCs were infected with AdGFP (control) or AdSmad3 followed by pre-incubation with vehicle (DMSO), resveratrol (50 muM), or rapamycin (200 nM) for 2 h, and then treated with solvent or TGFbeta1 (5 ng/mL). For Western blotting analyses of p-Akt ( A ) and p-Smad3 ( B ), cells were collected after TGFbeta1 treatment for 2 h and 0.5 h, respectively. Quantification: mean +- SEM of three independent experiments; normalization to total Akt (t-Akt) or to total Smad3 (t-Smad3); *P < 0.05 compared to each of the first 2 conditions; **P < 0.05 compared to AdSmad3 only; # P < 0.05 compared to AdSmad3 + TGFbeta1 without pre-incubation with a drug; n.s., not significant. ( C ). Rat carotid arteries were treated with either vehicle or resveratrol and collected on day 14 after angioplasty. Shown on the left are representative sections of p-Akt Immunostaining. Arrows indicate external elastic lamina (EEL); arrowheads mark internal elastic lamina (IEL). Scale bar = 20 mum. Negative staining: IgG instead of a primary antibody was used. Quantification: mean +- SEM of 4-5 animals; *P < 0.05 compared to vehicle control; HPF, high power field.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
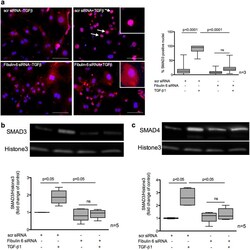
- Experimental details
- Figure 4 Nuclear localization of SMAD3/4 in TGF-beta stimulated CF. nCF transfected with scr siRNA or Fibulin-6 siRNA, with and without TGF-beta stimulation are fixed and stained for SMAD3 expression or are processed for nuclear lysate preparation. ( a ) Cells are stained with SMAD3 (red) and DAPI (blue) (Scale bar = 100 mum). In scr siRNA transfected CF translocation of SMAD3 into the nucleus can be observed upon TGF-beta stimulation. Arrows indicate SMAD3 positive nuclei. In fibulin-6 KD CF SMAD3 staining is almost absent in nuclei. High magnification inserts show nuclear SMAD3 staining in scr siRNA transfected CF and cytoplasmic SMAD3 staining in fibulin-6 siRNA transfected CF. A corresponding graph shows quantitative measurement of SMAD3 positive nuclei compared to the total number of cells (n = 3, p < 0.0001, nonparametric Mann-Whitney U test). The experiment was repeated three times with 10 pictures analyzed each time. Western blot showing ( b ) SMAD3 and ( c ) SMAD4 nuclear signal in TGF-beta stimulated nuclear lysates compared to the non-stimulated set. Westerm blot and corresponding densitometric analysis show increased localization of ( b ) SMAD3 and ( c ) SMAD4 in nuclear extracts after TGF-beta stimulation, which is absent in fibulin-6 KD CF (n = 5, p < 0.05 nonparametric Mann-Whitney U test).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
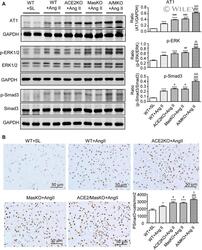
- Experimental details
- 5 Figure Double deletion of ACE2 and Mas receptor enhances Ang II-induced activation of AT1, ERK1/2, and Smad3 signalling in the hypertensive kidney. A, Western blot analysis of Ang II receptor type 1 (AT1), phospho-ERK1/2 (p-ERK1/2), phospho-Smad3 (p-Smad3). B, Immunohistochemistry of p-Smad3 nuclear translocation. Data represent the mean +- SEM for groups of 6-12 mice. * P < .05, *** P < .001 vs. WT + SL; # P < .05, ## P < .01, ### P < .001 vs. WT + Ang II; $ P < .05, $$ P < .01, $$$ P < .001 vs. ACE2 KO + Ang II and/or Mas KO + Ang II. Scale bar, 50 mum
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
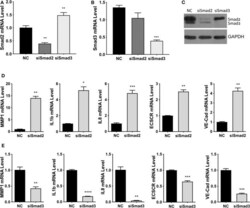
- Experimental details
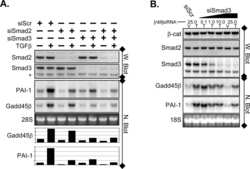
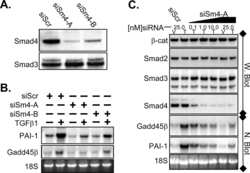
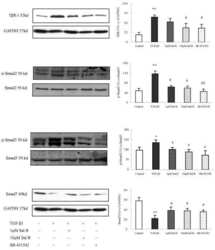
- Figure 4 Smad2 and Smad3 exert opposite effects on the expression of key genes involved in enEVT differentiation. HTR-8/SVneo cells were transfected with a siRNA targeting Smad2 (siSmad2) or Smad3 (siSmad3) or a non-targeting control (NC). Total RNA or protein was extracted. (A) Validation of Smad2 siRNA (siSmad2) by qPCR. (B) Validation of siSmad3 using qPCR. (C) Validation of siSmad2 and siSmad3 by Western blotting using an antibody detecting both total Smad2 and total Smad3. (D) Knockdown of Smad2 significantly increased the mRNA levels of MMP1, IL1b, IL8, ECSCR, and VE-cad. (E) Knockdown of Smad3 significantly reduced the mRNA levels of MMP1, IL1b, IL8, ECSCR, and VE-cad. Data represent mean +- SEM ( n = 3). * p < 0.05; ** p < 0.01; *** p < 0.001; and **** p < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
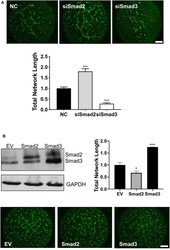
- Experimental details
- Figure 6 Smad2 suppresses while Smad3 promotes the formation of endothelial-like networks by trophoblasts. (A) HTR-8/SVneo cells transfected with siSmad2, siSmad3, or non-targeting control (NC) were seeded on matrigel-coated wells. After 16 h, cells were stained with Calcein AM and total network length was measured. siSmad2 significantly promoted, while siSmad3 significantly suppressed, the ability of trophoblasts to form network structures. (B) Overexpression of Smad2 significantly reduced while Smad3 significantly enhanced the total network length compared to empty vector control (EV). HTR-8/SVneo cells were transfected with EV, Smad2-, or Smad3-expressing plasmids. Overexpression of Smad2 and Smad3 was confirmed by Western blotting. Representative network formation and quantitative graphs (mean +- SEM, n = 3) were shown. * p < 0.05 and *** p < 0.001. Scale bar = 1 mm.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- FIGURE 8. Co-occupation of the H19 ICR by CTCF and Smads requires intact binding sites for CTCF on DNA. A , CTCF and Smads bind only to the maternally inherited allele of the H19 ICR. Primary hepatocytes were stimulated with TGFbeta1 for 4 h ( black bars ) or left untreated ( white bars ) prior to ChIP with the indicated antibodies. Immunoprecipitated chromatin was amplified with primers specific for the H19 ICR, which can discriminate between the maternally inherited allele () and the paternally inherited allele (). Input chromatin and nonspecific IgG controls are also shown after PCR amplification. B , MEFs were stimulated with TGFbeta1 for 4 h ( black bars ) or left untreated ( white bars ) prior to ChIP. The genotype of the amplified allele is shown as wild type or mutant with respect to the H19 ICR point mutations engineered in the specific sequences where CTCF binds. C , nonspecific IgG controls and input chromatin controls for the same samples analyzed in B are shown. In all of the panels, the Q-PCR data show the average values and standard deviations calculated from triplicate determinations.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
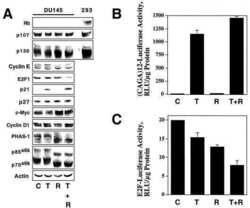
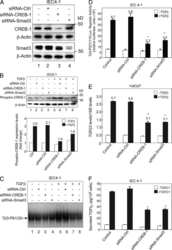
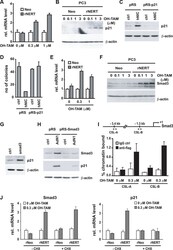
- Figure 4 p21 WAF1/Cip1 mediates Notch1-induced growth arrest and is controlled by Notch1 through a Smad3 dependent mechanism ( A and B ) PC3 cells stably infected with a retroviral vector expressing the flag tagged-activated Notch1 protein fused to the human estrogen receptor (rNERT), or control virus (Neo) were treated for 3 days with 4-hydroxytamoxifen at the indicated concentrations. Expression of p21 WAF1/Cip1 was analyzed by real time RT-PCR (using 36beta4 mRNA for normalization) (A) and by immuno-blotting (B). ( C ) PC3 cells were stably infected with a retrovirus expressing an shRNA against p21 WAF1/Cip1 (pRS-p21 ndeg1) or empty vector control (pRS). Cells were subsequently transfected with an expression vector for activated Notch1 (hNIC) together with a neomycin resistance gene, or empty vector control (ctrl). Twenty four hours later, cells were analyzed by immuno-blotting for p21 WAF1/Cip1 expression, with beta-actin for normalization. ( D ) PC3 cells plus/minus p21 WAF1/Cip1 knock-down and activated Notch1 expression as in the previous panel were replated (at 8 hours after transfection) at low density in triplicate dishes (1000 cells per 6 cm dish) in the presence of neomycin for selection of transfected cells. Numbers of colonies per dish were counted 10 days later. ( E and F ) The same PC3 cells with inducible Notch1 activity as in (A and B) were analyzed for Smad3 expression by real time RT-PCR (using 36beta4 mRNA for normalization) (E) and immunoblotting (F). (
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
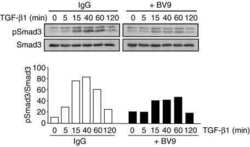
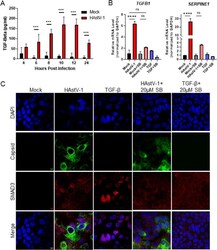
- 10.1371/journal.ppat.1009716.g006 Fig 6 TGF-beta activity increases during HAstV-1 infection. (A) Supernatants were collected from Caco-2 infected with HAstV-1 or mock infected from 4 to 24 hpi. Supernatants were then assayed for active TGF-beta using the PAI assay as described previously []. Error bars indicate standard deviations from two independent experiments performed in duplicate, and asterisks show statistical significance as measured by two-way ANOVA followed by Sidak's multiple comparisons test as follows: *, P < 0.05; **, P < 0.01; ***, P < 0.001. (B) TGFB1 and SERPINE1 , which is specifically activated by TGF-beta, mRNA levels were measured in mock infected, HAstV-1 infected (MOI 5), or TGF-beta (20ng/ml) treated Caco-2 cells at 24 hpi. Error bars indicate standard deviations from two independent experiments performed in duplicate, and asterisks show statistical significance as measured by ordinary one-way ANOVA followed by Tukey's multiple comparisons test as follows: ****, P < 0.0001. (C) Caco-2 cells mock infected, HAstV-1 infected, or treated with TGF-beta with and without 20muM SB431542 treatment were stained for SMAD3 (red), astrovirus capsid protein (green) and DAPI (blue). Images are representative of two independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
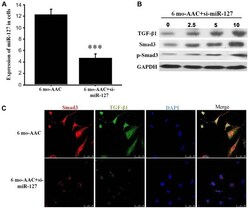
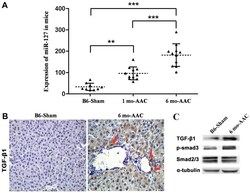
- Figure 3. Overexpression of miR-127 promotes myocardial failure. (A) Reverse transcription-quantitative polymerase chain reaction was used to detect the expression of miR-127 in the DOX-induced and control mice. (B) Immunohistochemistry indicated alterations of TGF-beta1 expression in the induced cardiomyocytes. Magnification, x40. (C) Western blot analysis detected the expression of TGF-beta1 and Smad following induction and in the control group. **P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
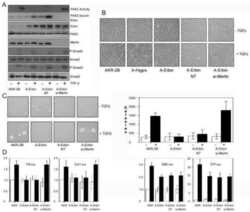
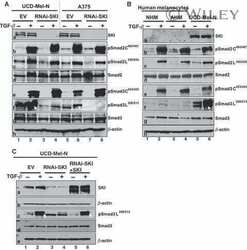
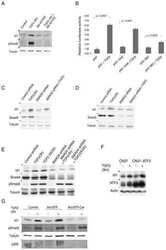
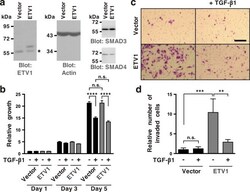
- Figure 4 Inhibition of ETV1-dependent invasion by TGF-beta1 in RWPE-1 cells. ( a ) Western blot analysis of ETV1 expression in stably transduced RWPE-1 cells. *Non-specific band. ETV1 and actin blots were derived from the same gel that was cut at ~50 kDa and the two resulting parts were then separately processed for western blotting utilizing anti-ETV1 or anti-actin antibodies. SMAD3 and SMAD4 blots were derived from different gels. Full-length blots are presented in Supplementary Fig. S2 . ( b ) Cell growth in the presence or absence of TGF-beta1 (n = 3). ( c ) Representative images from cell invasion assays. Scale bar = 200 um. ( d ) Quantification of cell invasion (n = 3). Statistical significance was determined with one-way ANOVA (Tukey's multiple comparisons test); n.s., not significant; **P < 0.01; ***P < 0.001; ****P < 0.0001. Shown are means with standard deviations.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
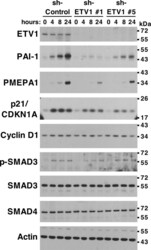
- Figure 5 Regulation of TGF-beta-dependent gene expression by ETV1. PC3 prostate cancer cells stably expressing control shRNA or two different ETV1 shRNAs were treated with TGF-beta1 for 0, 4, 8 or 24 hours. Western blots for indicated proteins are shown. Full-length blots are presented in Supplementary Fig. S4 . Blots for PAI-1 and p21 were derived from the same gel cut at ~34 kDa. Similarly, blots for SMAD4 and PMEPA1 were derived from another gel cut at ~43 kDa. A third gel was utilized for cyclin D1 blotting. The ETV1 blot was derived from the stripped PAI-1 blot, the p-SMAD3 blot from the stripped SMAD4 blot, the SMAD3 blot from the stripped cyclin D1 blot, and the actin blot from the stripped SMAD3 blot.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
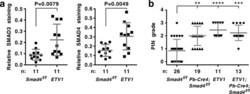
- Figure 6 Analysis of ETV1 transgenic mice at 16.1-25.5 months of age. ( a ) Immunohistochemical analysis of SMAD3 and SMAD4 expression in prostates. ETV1 transgenic mice were compared to Smad4 f/f mice that are indistinguishable from wild-type mice. Means with standard deviations are shown. Statistical significance was assessed with an unpaired, two-tailed t test. ( b ) Indicated mice were analyzed for PIN formation. Kruskal-Wallis test (Dunn's multiple comparisons test). **P < 0.01; ***P < 0.001; ****P < 0.0001. Shown are means with standard deviations. Supplementary Fig. S6 provides data for each mouse separately and shows that the average age between the four groups of mice was not statistically different.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
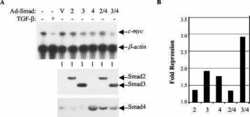
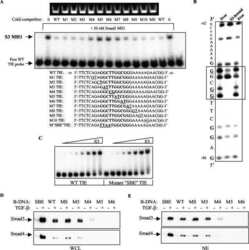
- FIGURE 1. Smad3 interacts with the zinc finger domain of CTCF. A , immunoprecipitation ( IP ) of FLAG-CTCF followed by immunoblotting ( IB ) for the indicated proteins after transfection of HEK293T cells with the indicated plasmids. Total cell lysates ( TCL ) demonstrate the input Smad levels. An asterisk indicates a nonspecific protein. B , co-immunoprecipitation of endogenous CTCF with endogenous Smad3 from HepG2 cells in the absence of TGFbeta stimulation. Immunoblots of total cell lysates are shown as input, and flag represents immunoprecipitation with a nonspecific antibody. C , GST pulldown assay of transfected Myc-Smad3 with the indicated GST fragments of CTCF. An aliquot of transfected HEK293T cells shows the Smad3 expression used as input, and Ponceau staining shows the recombinant CTCF fragments. An asterisk indicates a nonspecific protein. Molecular size markers (in kDa) are shown in the bottom panel. D , schematic diagram of Smad2, its alternatively spliced isoform Smad2Deltaex3, Smad3, and Smad4. The MH1, linker, and MH2 domains are highlighted, along with the C-terminal di-serine motif that is phosphorylated by the TGFbeta type I receptor. Within the MH1 domain, features of the proteins discussed in this paper are emphasized: the N-terminal Smad2-specific peptide insert GAG, the exon 3 corresponding peptide insert TID, and the juxtaposed DNA-binding domain ( DBD ) of all Smads. Splicing of exon 3 is indicated, and the lack of the GAG insert from the sequence of
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA