Antibody data
- Antibody Data
- Antigen structure
- References [13]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [6]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 48-0639-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD63 Monoclonal Antibody (H5C6), eFluor™ 450, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: This H5C6 monoclonal antibody reacts with human CD63, a type III member of the tetraspanin family of transmembrane proteins. CD63 is expressed intracellularly on lysosomes, endosomes, and granules of resting platelets and basophils. However, cell surface expression of CD63 can be detected on activated basophils and platelets, monocytes, macrophages, and granulocytes. This receptor is also expressed on endothelial cells, fibroblasts, and smooth muscle cells. Studies have demonstrated that CD63 associates with integrins (VLA-3 and VLA-6) and TIMP-1 to mediate the allergic response. Applications Reported: This H5C6 antibody has been reported for use in flow cytometric analysis. Applications Tested: This H5C6 antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (1 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. eFluor® 450 is an alternative to Pacific Blue®. eFluor® 450 emits at 445 nm and is excited with the Violet laser (405 nm). Please make sure that your instrument is capable of detecting this fluorochome. Excitation: 405 nm; Emission: 445 nm; Laser: Violet Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- H5C6
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references M2-like tumor-associated macrophages transmit exosomal miR-27b-3p and maintain glioblastoma stem-like cell properties.
Cutting edge approaches for rapid characterization of airway exosomes.
Proteomic and lipidomic analysis of exosomes derived from ovarian cancer cells and ovarian surface epithelial cells.
Ionizing Radiation Increases the Activity of Exosomal Secretory Pathway in MCF-7 Human Breast Cancer Cells: A Possible Way to Communicate Resistance against Radiotherapy.
Unique Lipid Signatures of Extracellular Vesicles from the Airways of Asthmatics.
Urinary exosomes as a novel biomarker for evaluation of α-lipoic acid's protective effect in early diabetic nephropathy.
MicroRNA-containing extracellular vesicles released from endothelial colony-forming cells modulate angiogenesis during ischaemic retinopathy.
Microvesicles derived from human umbilical cord Wharton's jelly mesenchymal stem cells attenuate bladder tumor cell growth in vitro and in vivo.
Marked differences in the signaling requirements for expression of CD203c and CD11b versus CD63 expression and histamine release in human basophils.
Identification of CD63 as a tissue inhibitor of metalloproteinase-1 interacting cell surface protein.
Flow cytometric analysis of in vitro activated basophils, specific IgE and skin tests in the diagnosis of pollen-associated food allergy.
Specific association of CD63 with the VLA-3 and VLA-6 integrins.
Cellular distribution of CD63 antigen in platelets and in three megakaryocytic cell lines.
Zhao G, Ding L, Yu H, Wang W, Wang H, Hu Y, Qin L, Deng G, Xie B, Li G, Qi L
Cell death discovery 2022 Aug 4;8(1):350
Cell death discovery 2022 Aug 4;8(1):350
Cutting edge approaches for rapid characterization of airway exosomes.
Hough KP, Deshane JS
Methods (San Diego, Calif.) 2020 May 1;177:27-34
Methods (San Diego, Calif.) 2020 May 1;177:27-34
Proteomic and lipidomic analysis of exosomes derived from ovarian cancer cells and ovarian surface epithelial cells.
Cheng L, Zhang K, Qing Y, Li D, Cui M, Jin P, Xu T
Journal of ovarian research 2020 Jan 22;13(1):9
Journal of ovarian research 2020 Jan 22;13(1):9
Ionizing Radiation Increases the Activity of Exosomal Secretory Pathway in MCF-7 Human Breast Cancer Cells: A Possible Way to Communicate Resistance against Radiotherapy.
Jabbari N, Nawaz M, Rezaie J
International journal of molecular sciences 2019 Jul 25;20(15)
International journal of molecular sciences 2019 Jul 25;20(15)
Unique Lipid Signatures of Extracellular Vesicles from the Airways of Asthmatics.
Hough KP, Wilson LS, Trevor JL, Strenkowski JG, Maina N, Kim YI, Spell ML, Wang Y, Chanda D, Dager JR, Sharma NS, Curtiss M, Antony VB, Dransfield MT, Chaplin DD, Steele C, Barnes S, Duncan SR, Prasain JK, Thannickal VJ, Deshane JS
Scientific reports 2018 Jul 9;8(1):10340
Scientific reports 2018 Jul 9;8(1):10340
Urinary exosomes as a novel biomarker for evaluation of α-lipoic acid's protective effect in early diabetic nephropathy.
Sun H, Yao W, Tang Y, Zhuang W, Wu D, Huang S, Sheng H
Journal of clinical laboratory analysis 2017 Nov;31(6)
Journal of clinical laboratory analysis 2017 Nov;31(6)
MicroRNA-containing extracellular vesicles released from endothelial colony-forming cells modulate angiogenesis during ischaemic retinopathy.
Dellett M, Brown ED, Guduric-Fuchs J, O'Connor A, Stitt AW, Medina RJ, Simpson DA
Journal of cellular and molecular medicine 2017 Dec;21(12):3405-3419
Journal of cellular and molecular medicine 2017 Dec;21(12):3405-3419
Microvesicles derived from human umbilical cord Wharton's jelly mesenchymal stem cells attenuate bladder tumor cell growth in vitro and in vivo.
Wu S, Ju GQ, Du T, Zhu YJ, Liu GH
PloS one 2013;8(4):e61366
PloS one 2013;8(4):e61366
Marked differences in the signaling requirements for expression of CD203c and CD11b versus CD63 expression and histamine release in human basophils.
MacGlashan D Jr
International archives of allergy and immunology 2012;159(3):243-52
International archives of allergy and immunology 2012;159(3):243-52
Identification of CD63 as a tissue inhibitor of metalloproteinase-1 interacting cell surface protein.
Jung KK, Liu XW, Chirco R, Fridman R, Kim HR
The EMBO journal 2006 Sep 6;25(17):3934-42
The EMBO journal 2006 Sep 6;25(17):3934-42
Flow cytometric analysis of in vitro activated basophils, specific IgE and skin tests in the diagnosis of pollen-associated food allergy.
Ebo DG, Hagendorens MM, Bridts CH, Schuerwegh AJ, De Clerck LS, Stevens WJ
Cytometry. Part B, Clinical cytometry 2005 Mar;64(1):28-33
Cytometry. Part B, Clinical cytometry 2005 Mar;64(1):28-33
Specific association of CD63 with the VLA-3 and VLA-6 integrins.
Berditchevski F, Bazzoni G, Hemler ME
The Journal of biological chemistry 1995 Jul 28;270(30):17784-90
The Journal of biological chemistry 1995 Jul 28;270(30):17784-90
Cellular distribution of CD63 antigen in platelets and in three megakaryocytic cell lines.
Hamamoto K, Ohga S, Nomura S, Yasunaga K
The Histochemical journal 1994 Apr;26(4):367-75
The Histochemical journal 1994 Apr;26(4):367-75
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
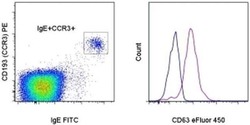
- Experimental details
- Staining of normal human peripheral blood cells with Anti-Human IgE FITC (Product # 11-6986-42), Anti-Human CD193 (CCR3) PE (Product # 12-1939-42) and Mouse IgG1 K Isotype Control eFluor® 450 (Product # 48-4714-82) (blue histogram) or Anti-Human CD63 eFluor® 450 (purple histogram). CD193+IgG+ cells (left) were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
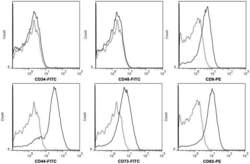
- Experimental details
- Figure 2 hWJMSC-MVs surface expressed molecules analysis. Flow cytometery analysis showed hWJMSC-MVs were positive for some surface expressed molecules typically expressed by MSCs, such as CD9, CD44, CD63, CD73, and negative for CD34, CD45.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
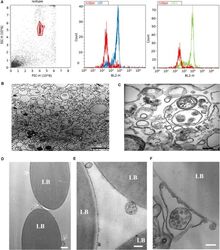
- Experimental details
- Figure 1 Characterization of endothelial colony-forming cells (ECFC)-derived extracellular vesicles (EVs). ( A ) Isolated ECFC-derived EVs were conjugated to CD63-coated latex beads to aid detection by flow cytometry. The gated bead-bound population (left panel) tested positive for EV markers CD9 and CD63. ( B ) Electron micrograph demonstrating the heterogeneity of the EV population (scale bar 1 mum). ( C ) Higher-magnification electron micrograph showing EVs of variable sizes and including a multivesicular body (scale bar 200 nm). ( D-F ) Electron micrographs of EVs bound to latex beads (LB) coated with anti-CD63 antibody. ( D ) White arrows indicate CD63-positive EVs of varying sizes (scale bar 1 mum). ( E ) Higher-magnification micrograph of EVs (scale bar 200 nm). In ( F ), a multivesicular body appears to be encapsulated within a CD63-positive membrane (scale bar 200 nm).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
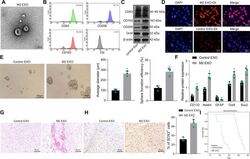
- Experimental details
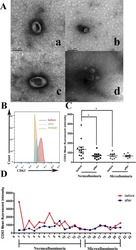
- Fig. 2 M2-TAM-derived exosomes enhance the stemness of GSCs. A TEM images of M2-TAM-derived exosomes. The presence of CD63, CD206, and CD163 in M2-TAM-derived exosomes analyzed by flow cytometric ( B ) and Western blot ( C ) analyses. D Fluorescence images of GSCs co-cultured with Dil-labeled M2-TAM-derived exosomes with nuclei stained with DAPI (blue). E Representative images showing neurosphere formation of GSCs along with the statistics of GSC formation rate and diameter of spheres. F Expression of stem cell-related protein CD133, Nestin, Oct4, Sox2, and GFAP in GSCs measured by Western blot analysis. G HE staining images of xenograft tumors from mice. H The representative immunohistochemical images of Sox2 in xenograft tumors and the percentage of GSCs labeled by Sox2. I Kaplan-Meier survive curve of tumor-bearing mice. n = 10. * p < 0.05 vs. the control EXO group. Measurement data were depicted as mean +- standard deviation, comparison of that between two groups was conducted by unpaired t test. Cell experiments were repeated three times independently.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
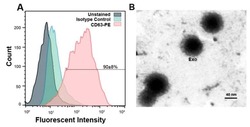
- Experimental details
- Figure 5 ( A ) Flow cytometric data confirmed exosomes by identifying exosomal marker CD63 and 90 +- 8% of exosomes in suspension represented CD63 marker. ( B ) Representative micrograph from transmission electron microscopy showing the nano-sized exosomes (Scale bar 40 nm). Exo: exosomes.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 Exosome isolation and analysis. A&C Representative transmission electron microscopy (TEM) images obtained for exosomes from HOSEPiC and SKOV-3. Scale bar: 100 nm. B&D flow cytometry analysis analyses show intensities for exosomal markers (CD9, CD63). (E) Particle size distribution by NanoSight analysis of exosomes from HOSEPiC and SKOV-3 (F) Western blot analyses show increased intensities for exosomal markers (Alix, TSG101)
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry