Antibody data
- Antibody Data
- Antigen structure
- References [5]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 62-0399-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD39 Monoclonal Antibody (eBioA1 (A1)), Super Bright™ 436, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- The eBioA1 monoclonal antibody reacts with human CD39 also known as ectonucleoside triphosphate diphosphohydrolase 1 (ENTPD1) or NTPDase. CD39 is an integral membrane protein with two transmembrane domains and exists as a homotetramer. It is the most prominent ectoenzyme of the immune system. The function of CD39 is to effectively remove toxic extracellular ATP by converting it to ADP or AMP. CD39 is thought to work together with CD73 to hydrolyze ATP and has been well characterized on Langerhans cells. Expression of CD39 was originally identified on activated lymphocytes. Expression is also found on a subset of T cells, B cells and dendritic cells as well as weak staining on monocytes and granulocytes. Recently, CD39 and CD73 have been found on regulatory T cells (Treg). Expression of CD39 on Treg may facilitate their entry into inflamed areas where high levels of ATP are present. Expression of CD39 on Foxp3+CD4+ cells ranges from 25-45%. This eBioA1 (A1) antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (0.25 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Super Bright 436 can be excited with the violet laser line (405 nm) and emits at 436 nm. We recommend using a 450/50 bandpass filter, or equivalent. Please make sure that your instrument is capable of detecting this fluorochrome. When using two or more Super Bright dye-conjugated antibodies in a staining panel, it is recommended to use Super Bright Complete Staining Buffer (Product # SB-4401) to minimize any non-specific polymer interactions. Please refer to the datasheet for Super Bright Staining Buffer for more information. Excitation: 405 nm; Emission: 436 nm; Laser: Violet Laser Super Bright Polymer Dyes are sold under license from Becton, Dickinson and Company.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- eBioA1 (A1)
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Pinpointing the tumor-specific T cells via TCR clusters.
Neoadjuvant anti-OX40 (MEDI6469) therapy in patients with head and neck squamous cell carcinoma activates and expands antigen-specific tumor-infiltrating T cells.
Circulating Exosomes Inhibit B Cell Proliferation and Activity.
Immune Suppressive Effects of Plasma-Derived Exosome Populations in Head and Neck Cancer.
Molecular profiling of driver events in metastatic uveal melanoma.
Goncharov MM, Bryushkova EA, Sharaev NI, Skatova VD, Baryshnikova AM, Sharonov GV, Karnaukhov V, Vakhitova MT, Samoylenko IV, Demidov LV, Lukyanov S, Chudakov DM, Serebrovskaya EO
eLife 2022 Apr 4;11
eLife 2022 Apr 4;11
Neoadjuvant anti-OX40 (MEDI6469) therapy in patients with head and neck squamous cell carcinoma activates and expands antigen-specific tumor-infiltrating T cells.
Duhen R, Ballesteros-Merino C, Frye AK, Tran E, Rajamanickam V, Chang SC, Koguchi Y, Bifulco CB, Bernard B, Leidner RS, Curti BD, Fox BA, Urba WJ, Bell RB, Weinberg AD
Nature communications 2021 Feb 16;12(1):1047
Nature communications 2021 Feb 16;12(1):1047
Circulating Exosomes Inhibit B Cell Proliferation and Activity.
Schroeder JC, Puntigam L, Hofmann L, Jeske SS, Beccard IJ, Doescher J, Laban S, Hoffmann TK, Brunner C, Theodoraki MN, Schuler PJ
Cancers 2020 Jul 29;12(8)
Cancers 2020 Jul 29;12(8)
Immune Suppressive Effects of Plasma-Derived Exosome Populations in Head and Neck Cancer.
Beccard IJ, Hofmann L, Schroeder JC, Ludwig S, Laban S, Brunner C, Lotfi R, Hoffmann TK, Jackson EK, Schuler PJ, Theodoraki MN
Cancers 2020 Jul 21;12(7)
Cancers 2020 Jul 21;12(7)
Molecular profiling of driver events in metastatic uveal melanoma.
Karlsson J, Nilsson LM, Mitra S, Alsén S, Shelke GV, Sah VR, Forsberg EMV, Stierner U, All-Eriksson C, Einarsdottir B, Jespersen H, Ny L, Lindnér P, Larsson E, Olofsson Bagge R, Nilsson JA
Nature communications 2020 Apr 20;11(1):1894
Nature communications 2020 Apr 20;11(1):1894
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of normal human peripheral blood cells with Anti-Human CD19 APC (Product # 17-0199-42) and Mouse IgG1 K Isotype Control Super Bright 436 (Product # 62-4714-82) (left) or Anti-Human CD39 Super Bright 436 (right). Cells in the lymphocyte gate were used for analysis.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
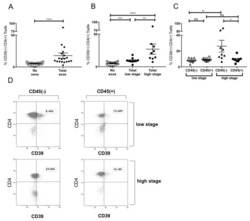
- Figure 5 Induction of CD39+ T reg differentiation by plasma-derived exosomes. ( A ) Expression of CD39 after incubation with total exosome fraction of patients of all stages ( B ) Expression of CD39 after incubation with total exosomes of low stage vs. high stage patients. ( C ) Expression of CD39 after incubation with CD45(-) and CD45(+) exosomes of low stage and high stage patients. Note that only incubation with CD45(-) exosomes showed significant stage-dependent differences. ; n = 18. p values were determined with Mann-Whitney test, with * p < 0.05, ** p < 0.005, **** p < 0.0001. Bars represent standard error of mean (SEM). ( D ) Representative flow cytometry plots depicting CD39 expression of CD4+ T cells after incubation with CD45(-) (left) or CD45(+) (right) exosomes of UICC low stage (top) or high stage (bottom) patients.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
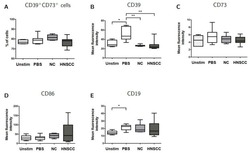
- Figure 5 B cells were harvested after 2 days of co-culture with either NC or HNSCC exosomes or PBS and stained for FACS analysis. ( A ) Frequency of CD39 + CD73 + regulatory B cells. ( B ) The expression of CD39 on B cells was reduced after co-culture with NC or HNSCC exosomes. ( C ) Expression of CD73 on B cells. ( D ) Expression of CD86 on B cells. ( E ) The expression of CD19 on B cells was increased by stimulation with CD40L and IL-4. **: p < 0.01; *: p < 0.05, n = 8 (HNSCC), n = 6 (NC), n = 5 (Unstim). Unstim = Unstimulated B cells, NC = no cancer (exosomes from blood plasma of healthy volunteers), HNSCC, exosomes from blood plasma of HNSCC patients.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Analysis of tumor-infiltrating lymphocytes. a Proportions of CD8 + and CD4 + T cells from biopsy material, and proportions of these that were positive for PD-1 and CD39. Sample UM22 was derived from a patient that has previously been treated with chemotherapy, possibly affecting TIL proportions in this sample. b Assessment of T-cell reactivity against MART-1, gp100 and NY-ESO-1 in yTIL cultures. Proportions found to be reactive are indicated. Samples tested were those with the HLA-A*02:01 genotype, as this genotype is known to present MART-1 and gp100. Samples with this genotype (Supplementary Data 7 ) that are not shown were also tested and found to be negative. c Analysis of relative levels of PDCD1 (PD-1) and ENTPD1 (CD39) expression among different CD8 + T-cell clonotypes, determined by single-cell RNA-seq of yTILs. Clonotypes with one pair of alpha and beta chain were included, and the ones with greatest expression of both markers are highlighted. Point sizes are proportional to clonotype frequency. Gray color indicates clones that were negative for either PDCD1 or ENTPD1 , whereas other colors indicate different clonotypes that correspond to those in Supplementary Fig. 7e . d Expression of T-cell markers and checkpoint receptors in bulk RNA-seq data from biopsies (batch-corrected log 2 RPKM normalized values). yTILs young TILs, TILs isolated and expanded from a biopsy with a low dose of IL-2.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
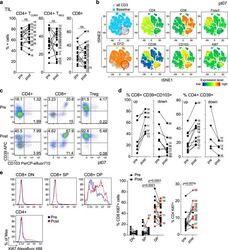
- Fig. 2 Changes in TIL composition after OX40 administration. TIL from a pretreatment biopsy and a surgical specimen after OX40 therapy were assessed for lymphocyte composition and activation markers. The gating strategy is outlined in Supplementary Fig. 3 . a Percentages of CD4+ Tconv cells, CD4+ Treg cells, and CD8+ T cells in each patient before and after OX40 administration, N = 17 patients. b tSNE analysis of the pre and post specimens from patient HNOX07, gated on CD3+ cells. Blue represents the baseline sample, orange the day of surgery sample and gray is the concatenated file. The red circle indicates the population of cells expressing both CD103 and CD39. tSNE analysis was performed on N = 4 patients, one representative patient is shown here, two more patients are shown in Supplementary Fig. 4 . c Flow cytometric analysis of the expression of CD103 and CD39 in CD4+ Tconv cells, CD8+ cells, and CD4+ Treg cells in one immune-responding head and neck squamous cell carcinoma (HNSCC) patient pre- and post OX40 therapy. d Summary of the flow cytometric analysis in (c), left panel depicts CD8+ CD103+ CD39+ T cells and the right panel depicts CD4+ CD39+ T cells; patients with an increase are shown on the left, patients with a decrease are on the right. e Expression of Ki-67 was assessed among memory CD4+ TIL and CD8+ TIL subsets (DN, SP, and DP) in biopsy (pre) and DOS (post) tissue ( N = 17 patients). Blue histograms indicate pre, red indicate post tissues. The left graph sh
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
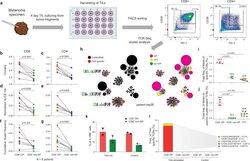
- Figure 2. T cell receptor (TCR) clusters in CD39 + PD1 + tumor-infiltrating lymphocytes (TILs). ( a ) The experimental workflow. ( b-g ) TCRbeta repertoire analysis for CD8 + ( b, d, f ) and CD4 + ( c, e, g ) double-positive (DP) and non-DP TIL subsets sorted from metastatic lymph nodes of eight melanoma patients. Panels show repertoire clonality calculated as [1 - Normalized Shannon-Wiener index] ( b, c ), normalized counts ( d, e ), and cumulative frequency of cluster-related clonotypes, that is, total weight of all clusters as a proportion of TCRbeta repertoire ( f, g ). Paired t-test. ( h ) TCRbeta clusters identified in repertoires obtained from fresh-frozen tumor (FFT) samples, and sorted CD8 + DP and non-DP TILs for HLA-A*02 patient mp26. VDJdb-matched TAA-specific clusters are colored in red. ( i , j ) Cumulative frequency of ( i ) VDJdb-matched TAA-specific clonotypes and ( j ) VDJdb-matched TAA-specific cluster-related clonotypes within CD8 + DP, CD8 + non-DP, and FFT TCRbeta repertoires of patient mp26. One-way ANOVA, Bonferroni multiple comparisons correction. ( k , l ) Proportion of CD137 + cells among CD8 + T cells ( k ) and proportion of VDJdb-matched TAA-specific clonotypes in sorted CD137 + CD8 + T cells ( l ) in DP and non-DP TILs from patient mp26 that were cultured and re-stimulated with TAA-loaded or control dendritic cells. Figure 2--figure supplement 1. ( a ) Fluorescence-activated cell sorting (FACS) gating for sorting of CD8 + and CD4 + CD39 + PD-1 +
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry