Antibody data
- Antibody Data
- Antigen structure
- References [3]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [3]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA1-134 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- RhoA Monoclonal Antibody (1B8-1C7)
- Antibody type
- Monoclonal
- Antigen
- Recombinant full-length protein
- Description
- By Western blot, MA1-134 detects recombinant RhoA, but not recombinant RhoB or recombinant RhoC. By Western blot on mouse and rat kidney whole tissue lysates, MA1-134 detects a band of ~22kD corresponding to the expected molecular weight of RhoA. A nonspecific band at ~55kD was also detected in both kidney lysates.
- Reactivity
- Human, Mouse, Rat
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 1B8-1C7
- Vial size
- 100 μg
- Concentration
- 1 mg/mL
- Storage
- -20°C
Submitted references S100A4 is activated by RhoA and catalyses the polymerization of non-muscle myosin, adhesion complex assembly and contraction in airway smooth muscle.
Curvotaxis directs cell migration through cell-scale curvature landscapes.
Influence of Inherent Mechanophenotype on Competitive Cellular Adherence.
Zhang W, Gunst SJ
The Journal of physiology 2020 Oct;598(20):4573-4590
The Journal of physiology 2020 Oct;598(20):4573-4590
Curvotaxis directs cell migration through cell-scale curvature landscapes.
Pieuchot L, Marteau J, Guignandon A, Dos Santos T, Brigaud I, Chauvy PF, Cloatre T, Ponche A, Petithory T, Rougerie P, Vassaux M, Milan JL, Tusamda Wakhloo N, Spangenberg A, Bigerelle M, Anselme K
Nature communications 2018 Sep 28;9(1):3995
Nature communications 2018 Sep 28;9(1):3995
Influence of Inherent Mechanophenotype on Competitive Cellular Adherence.
Shah MK, Garcia-Pak IH, Darling EM
Annals of biomedical engineering 2017 Aug;45(8):2036-2047
Annals of biomedical engineering 2017 Aug;45(8):2036-2047
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
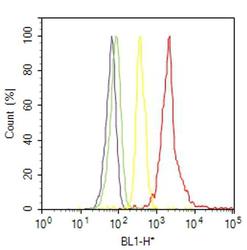
- Experimental details
- Flow cytometry analysis of RhoA was done on A-431 cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with RhoA Mouse Monoclonal Antibody (MA1134, red histogram) or with mouse isotype control (yellow histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Rabbit Anti-Mouse Secondary Antibody (A11059) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
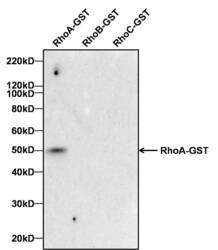
- Experimental details
- Antibody specificity for modified targets can be established by testing the detection of the target protein along with closely related proteins (belonging to the same family). Relevant related proteins (RhoB and RhoC) when tested along with RhoA recombinant protein using RhoA Mouse Monoclonal Antibody (Product # MA1-134) showed that the antibody specifically detected RhoA.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
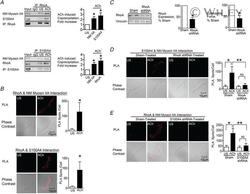
- Experimental details
- 4 Figure The interaction between S100A4 with NM myosin II in response to ACh is stimulated by the activation of RhoA A , RhoA or S100A4 was immunoprecipitated from extracts of SM tissues and immunocomplexes were blotted for RhoA, S100A4 and NM myosin IIA. Stimulation for 5 min with 10 -5 M ACh significantly increased the amount of S100A4 ( n = 6, p = 0.0007) and NM myosin IIA ( n = 4, p = 0.0073) in RhoA immunoprecipitates. ACh stimulation increased the amount of RhoA ( n = 6, p = 0.0005) and NM myosin IIA ( n = 9, p = 0.0001) in S100A4 immunoprecipitates. B , in situ PLA was used to visualize the interaction of RhoA with NM myosin IIA and of RhoA with S100A4 in freshly dissociated tracheal SM cells. Stimulation with ACh caused a significant increase in the number of PLA spots at the cell membrane (RhoA and NM myosin IIA, n = 16 cells for both US and ACh, p = 0.0001); S100A4 and RhoA, n = 19 for US and n = 25 for ACh, p = 0.0001). C , tracheal SM tissues were depleted of RhoA protein using shRNA and RhoA was quantified in tissue extracts by immunoblotting ( n = 8, p = 0.0001). Depletion of RhoA significantly inhibited tension development in response to 10 -5 M ACh ( n = 8, P < 0.0001). D , PLA was used to visualize interactions between S100A4 and NM myosin IIA in cells dissociated from sham-treated and RhoA-depleted tissues. ACh stimulation of sham-treated cells resulted in a significant increase in the number of interactions between S100A4 and NM myosin IIA (US, n = 27; ACh,
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
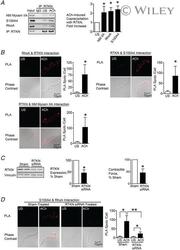
- Experimental details
- 5 Figure Rhotekin couples RhoA to S100A4 and NM myosin II in SM cells and tissues stimulated with ACh A , rhotekin (RTKN) was immunoprecipitated from tissue extracts and immunocomplexes were blotted for RhoA, S100A4, RTKN and NM myosin IIA. Stimulation for 5 min with 10 -5 M ACh significantly increased the coprecipitation of NM myosin IIA ( n = 6, p = 0.0070), RhoA ( n = 3, p = 0.0359) and S100A4 ( n = 9, p = 0.0001) with RTKN. B , in situ PLA was used to determine the interaction of RTKN and RhoA, S100A4 and NM myosin IIA in freshly dissociated tracheal SM cells. Stimulation with ACh caused a significant increase in the number of PLA complexes between all four proteins (RTKN-RhoA, n = 20, p = 0.0001; RTKN-S100A4, n = 20, p = 0.0001; RTKN-NM myosin IIA, n = 31, p = 0.0001). C , tracheal SM tissues were treated with RTKN siRNA. Depletion of RTKN significantly inhibited protein expression ( n = 10, p = 0.0001) and also inhibited tension development in response to 10 -5 M ACh stimulation ( n = 16, p = 0.0001). D , PLA shows interactions between S100A4 and RhoA in cells dissociated from sham-treated tissues and from RTKN-depleted tissues. ACh stimulation of sham-treated cells resulted in a significant increase in the interactions between S100A4 and RhoA (US, n = 16; ACh, n = 20). RTKN depletion inhibited the ACh-induced increase of the interaction between S100A4 and RhoA ( p = 0.0001) (US, n = 18; ACh, n = 19). Data analysed by a paired Student's t test ( A and C ), an unpaired S
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Flow cytometry
Flow cytometry