MN1010
antibody from Invitrogen Antibodies
Targeting: MAPT
DDPAC, FLJ31424, FTDP-17, MAPTL, MGC138549, MSTD, MTBT1, MTBT2, PPND, PPP1R103, tau
Antibody data
- Antibody Data
- Antigen structure
- References [24]
- Comments [0]
- Validations
- Western blot [1]
- Other assay [16]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MN1010 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Tau Monoclonal Antibody (BT2)
- Antibody type
- Monoclonal
- Antigen
- Purifed from natural sources
- Description
- MN1010 recognizes normal Tau from human, rat, monkey, and bovine brain. No cross reactivity with PHF-Tau has been observed. The epitope of this antibody has been mapped on human Tau between residue 194 and 198 (numbering according to human Tau40), corresponding to the amnio acid sequence RSGYS. MN1010 detects Tau which has a predicted molecular weight of approximately 79 kDa. This product is a Low Endotoxin formulation. Purity is >95% as determined by SDS-PAGE.
- Reactivity
- Human, Rat, Bovine
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- BT2
- Vial size
- 100 µg
- Concentration
- 0.2 mg/mL
- Storage
- -20° C, Avoid Freeze/Thaw Cycles
Submitted references TFEB regulates lysosomal exocytosis of tau and its loss of function exacerbates tau pathology and spreading.
Systemic delivery of a specific antibody targeting the pathological N-terminal truncated tau peptide reduces retinal degeneration in a mouse model of Alzheimer's Disease.
Genome-wide CRISPR screen identifies protein pathways modulating tau protein levels in neurons.
Development and Validation of a High Sensitivity Assay for Measuring p217 + tau in Cerebrospinal Fluid.
Highly sensitive quantification of Alzheimer's disease biomarkers by aptamer-assisted amplification.
Chronic PD-1 Checkpoint Blockade Does Not Affect Cognition or Promote Tau Clearance in a Tauopathy Mouse Model.
A post-translational modification signature defines changes in soluble tau correlating with oligomerization in early stage Alzheimer's disease brain.
Probing Conformational Dynamics of Tau Protein by Hydrogen/Deuterium Exchange Mass Spectrometry.
Detection of Aggregation-Competent Tau in Neuron-Derived Extracellular Vesicles.
A validated antibody panel for the characterization of tau post-translational modifications.
Membrane association and release of wild-type and pathological tau from organotypic brain slice cultures.
Variable tau accumulation in murine models with abnormal prion protein deposits.
Passive immunization with phospho-tau antibodies reduces tau pathology and functional deficits in two distinct mouse tauopathy models.
Analysis of tau post-translational modifications in rTg4510 mice, a model of tau pathology.
Insulin therapy modulates mitochondrial dynamics and biogenesis, autophagy and tau protein phosphorylation in the brain of type 1 diabetic rats.
Characterization of novel CSF Tau and ptau biomarkers for Alzheimer's disease.
Early postnatal lead exposure induces tau phosphorylation in the brain of young rats.
In vivo microdialysis reveals age-dependent decrease of brain interstitial fluid tau levels in P301S human tau transgenic mice.
Long term changes in phospho-APP and tau aggregation in the 3xTg-AD mice following cerebral ischemia.
Decrease in brain soluble amyloid precursor protein β (sAPPβ) in Alzheimer's disease cortex.
Increased association between rough endoplasmic reticulum membranes and mitochondria in transgenic mice that express P301L tau.
Detailed immunohistochemical characterization of temporal and spatial progression of Alzheimer's disease-related pathologies in male triple-transgenic mice.
Suppression of Parkin enhances nigrostriatal and motor neuron lesion in mice over-expressing human-mutated tau protein.
Assembly of alpha4beta2 nicotinic acetylcholine receptors assessed with functional fluorescently labeled subunits: effects of localization, trafficking, and nicotine-induced upregulation in clonal mammalian cells and in cultured midbrain neurons.
Xu Y, Du S, Marsh JA, Horie K, Sato C, Ballabio A, Karch CM, Holtzman DM, Zheng H
Molecular psychiatry 2021 Oct;26(10):5925-5939
Molecular psychiatry 2021 Oct;26(10):5925-5939
Systemic delivery of a specific antibody targeting the pathological N-terminal truncated tau peptide reduces retinal degeneration in a mouse model of Alzheimer's Disease.
Latina V, Giacovazzo G, Cordella F, Balzamino BO, Micera A, Varano M, Marchetti C, Malerba F, Florio R, Ercole BB, La Regina F, Atlante A, Coccurello R, Di Angelantonio S, Calissano P, Amadoro G
Acta neuropathologica communications 2021 Mar 9;9(1):38
Acta neuropathologica communications 2021 Mar 9;9(1):38
Genome-wide CRISPR screen identifies protein pathways modulating tau protein levels in neurons.
Sanchez CG, Acker CM, Gray A, Varadarajan M, Song C, Cochran NR, Paula S, Lindeman A, An S, McAllister G, Alford J, Reece-Hoyes J, Russ C, Craig L, Capre K, Doherty C, Hoffman GR, Luchansky SJ, Polydoro M, Dolmetsch R, Elwood F
Communications biology 2021 Jun 14;4(1):736
Communications biology 2021 Jun 14;4(1):736
Development and Validation of a High Sensitivity Assay for Measuring p217 + tau in Cerebrospinal Fluid.
Triana-Baltzer G, Van Kolen K, Theunis C, Moughadam S, Slemmon R, Mercken M, Galpern W, Sun H, Kolb H
Journal of Alzheimer's disease : JAD 2020;77(4):1417-1430
Journal of Alzheimer's disease : JAD 2020;77(4):1417-1430
Highly sensitive quantification of Alzheimer's disease biomarkers by aptamer-assisted amplification.
Chan HN, Xu D, Ho SL, He D, Wong MS, Li HW
Theranostics 2019;9(10):2939-2949
Theranostics 2019;9(10):2939-2949
Chronic PD-1 Checkpoint Blockade Does Not Affect Cognition or Promote Tau Clearance in a Tauopathy Mouse Model.
Lin Y, Rajamohamedsait HB, Sandusky-Beltran LA, Gamallo-Lana B, Mar A, Sigurdsson EM
Frontiers in aging neuroscience 2019;11:377
Frontiers in aging neuroscience 2019;11:377
A post-translational modification signature defines changes in soluble tau correlating with oligomerization in early stage Alzheimer's disease brain.
Ercan-Herbst E, Ehrig J, Schöndorf DC, Behrendt A, Klaus B, Gomez Ramos B, Prat Oriol N, Weber C, Ehrnhoefer DE
Acta neuropathologica communications 2019 Dec 3;7(1):192
Acta neuropathologica communications 2019 Dec 3;7(1):192
Probing Conformational Dynamics of Tau Protein by Hydrogen/Deuterium Exchange Mass Spectrometry.
Huang RY, Iacob RE, Sankaranarayanan S, Yang L, Ahlijanian M, Tao L, Tymiak AA, Chen G
Journal of the American Society for Mass Spectrometry 2018 Jan;29(1):174-182
Journal of the American Society for Mass Spectrometry 2018 Jan;29(1):174-182
Detection of Aggregation-Competent Tau in Neuron-Derived Extracellular Vesicles.
Guix FX, Corbett GT, Cha DJ, Mustapic M, Liu W, Mengel D, Chen Z, Aikawa E, Young-Pearse T, Kapogiannis D, Selkoe DJ, Walsh DM
International journal of molecular sciences 2018 Feb 27;19(3)
International journal of molecular sciences 2018 Feb 27;19(3)
A validated antibody panel for the characterization of tau post-translational modifications.
Ercan E, Eid S, Weber C, Kowalski A, Bichmann M, Behrendt A, Matthes F, Krauss S, Reinhardt P, Fulle S, Ehrnhoefer DE
Molecular neurodegeneration 2017 Nov 21;12(1):87
Molecular neurodegeneration 2017 Nov 21;12(1):87
Membrane association and release of wild-type and pathological tau from organotypic brain slice cultures.
Croft CL, Wade MA, Kurbatskaya K, Mastrandreas P, Hughes MM, Phillips EC, Pooler AM, Perkinton MS, Hanger DP, Noble W
Cell death & disease 2017 Mar 16;8(3):e2671
Cell death & disease 2017 Mar 16;8(3):e2671
Variable tau accumulation in murine models with abnormal prion protein deposits.
Piccardo P, King D, Brown D, Barron RM
Journal of the neurological sciences 2017 Dec 15;383:142-150
Journal of the neurological sciences 2017 Dec 15;383:142-150
Passive immunization with phospho-tau antibodies reduces tau pathology and functional deficits in two distinct mouse tauopathy models.
Sankaranarayanan S, Barten DM, Vana L, Devidze N, Yang L, Cadelina G, Hoque N, DeCarr L, Keenan S, Lin A, Cao Y, Snyder B, Zhang B, Nitla M, Hirschfeld G, Barrezueta N, Polson C, Wes P, Rangan VS, Cacace A, Albright CF, Meredith J Jr, Trojanowski JQ, Lee VM, Brunden KR, Ahlijanian M
PloS one 2015;10(5):e0125614
PloS one 2015;10(5):e0125614
Analysis of tau post-translational modifications in rTg4510 mice, a model of tau pathology.
Song L, Lu SX, Ouyang X, Melchor J, Lee J, Terracina G, Wang X, Hyde L, Hess JF, Parker EM, Zhang L
Molecular neurodegeneration 2015 Mar 26;10:14
Molecular neurodegeneration 2015 Mar 26;10:14
Insulin therapy modulates mitochondrial dynamics and biogenesis, autophagy and tau protein phosphorylation in the brain of type 1 diabetic rats.
Santos RX, Correia SC, Alves MG, Oliveira PF, Cardoso S, Carvalho C, Duarte AI, Santos MS, Moreira PI
Biochimica et biophysica acta 2014 Jul;1842(7):1154-66
Biochimica et biophysica acta 2014 Jul;1842(7):1154-66
Characterization of novel CSF Tau and ptau biomarkers for Alzheimer's disease.
Meredith JE Jr, Sankaranarayanan S, Guss V, Lanzetti AJ, Berisha F, Neely RJ, Slemmon JR, Portelius E, Zetterberg H, Blennow K, Soares H, Ahlijanian M, Albright CF
PloS one 2013;8(10):e76523
PloS one 2013;8(10):e76523
Early postnatal lead exposure induces tau phosphorylation in the brain of young rats.
Rahman A, Khan KM, Al-Khaledi G, Khan I, Attur S
Acta biologica Hungarica 2012 Dec;63(4):411-25
Acta biologica Hungarica 2012 Dec;63(4):411-25
In vivo microdialysis reveals age-dependent decrease of brain interstitial fluid tau levels in P301S human tau transgenic mice.
Yamada K, Cirrito JR, Stewart FR, Jiang H, Finn MB, Holmes BB, Binder LI, Mandelkow EM, Diamond MI, Lee VM, Holtzman DM
The Journal of neuroscience : the official journal of the Society for Neuroscience 2011 Sep 14;31(37):13110-7
The Journal of neuroscience : the official journal of the Society for Neuroscience 2011 Sep 14;31(37):13110-7
Long term changes in phospho-APP and tau aggregation in the 3xTg-AD mice following cerebral ischemia.
Koike MA, Garcia FG, Kitazawa M, Green KN, Laferla FM
Neuroscience letters 2011 May 9;495(1):55-9
Neuroscience letters 2011 May 9;495(1):55-9
Decrease in brain soluble amyloid precursor protein β (sAPPβ) in Alzheimer's disease cortex.
Wu G, Sankaranarayanan S, Hsieh SH, Simon AJ, Savage MJ
Journal of neuroscience research 2011 Jun;89(6):822-32
Journal of neuroscience research 2011 Jun;89(6):822-32
Increased association between rough endoplasmic reticulum membranes and mitochondria in transgenic mice that express P301L tau.
Perreault S, Bousquet O, Lauzon M, Paiement J, Leclerc N
Journal of neuropathology and experimental neurology 2009 May;68(5):503-14
Journal of neuropathology and experimental neurology 2009 May;68(5):503-14
Detailed immunohistochemical characterization of temporal and spatial progression of Alzheimer's disease-related pathologies in male triple-transgenic mice.
Mastrangelo MA, Bowers WJ
BMC neuroscience 2008 Aug 12;9:81
BMC neuroscience 2008 Aug 12;9:81
Suppression of Parkin enhances nigrostriatal and motor neuron lesion in mice over-expressing human-mutated tau protein.
Menéndez J, Rodríguez-Navarro JA, Solano RM, Casarejos MJ, Rodal I, Guerrero R, Sánchez MP, Avila J, Mena MA, de Yébenes JG
Human molecular genetics 2006 Jul 1;15(13):2045-58
Human molecular genetics 2006 Jul 1;15(13):2045-58
Assembly of alpha4beta2 nicotinic acetylcholine receptors assessed with functional fluorescently labeled subunits: effects of localization, trafficking, and nicotine-induced upregulation in clonal mammalian cells and in cultured midbrain neurons.
Nashmi R, Dickinson ME, McKinney S, Jareb M, Labarca C, Fraser SE, Lester HA
The Journal of neuroscience : the official journal of the Society for Neuroscience 2003 Dec 17;23(37):11554-67
The Journal of neuroscience : the official journal of the Society for Neuroscience 2003 Dec 17;23(37):11554-67
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot was performed using Anti-Tau Monoclonal Antibody (BT2) (Product # MN1010) and 40-80 kDa bands corresponding to Tau were observed in Mouse and Rat Brain but not in Mouse Kidney or Mouse and Rat Lung tissues. Tissue extracts (30 µg lysate) of Mouse Brain (Lane 1), Rat Brain (Lane 2), Mouse Kidney (Lane 3), Mouse Lung (Lane 4) or Rat Lung (Lane 5) were electrophoresed using NuPAGE™ 10% Bis-Tris Protein Gel (Product # NP0301BOX). Resolved proteins were then transferred onto a Nitrocellulose membrane (Product # IB23001) by iBlot® 2 Dry Blotting System (Product # IB21001). The blot was probed with the primary antibody (1:500) and detected by chemiluminescence with Goat anti-Mouse IgG (H+L) Superclonal™ Recombinant Secondary Antibody, HRP (Product # A28177,1:4000) using the iBright FL 1000 (Product # A32752). Chemiluminescent detection was performed using Novex® ECL Chemiluminescent Substrate Reagent Kit (Product # WP20005). Additional band at 25 kDa corresponds to tissue IgG and has been marked with an asterisk.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 2 Age-dependent changes in tau pathology in rTg4510 mice. A. AT8 p-tau staining in 3- and 6-month old rTg4510 brain sections. Scale bar = 500 mum. B-E. Age-dependent change in B. hippocampal soluble total tau, C. hippocampal soluble AT8 p-tau, D. hippocampal insoluble total tau, and E. hippocampal insoluble AT8 p-tau. Individual comparisons were vs. 3.5-month old mice using ANOVA with a Dunnett's post-hoc test (*p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
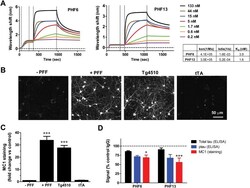
- Experimental details
- Fig 1 Immunodepletion with p-Tau antibodies reduces tau pathology in a primary neuronal model of tau aggregation. A. Kinetics of association and dissociation of PHF6 antibody and PHF13 antibody. The table summarizes the calculated association rates, dissociation rates, and K D values for PHF6 and PHF13. The vertical dotted lines demarcate, from left to right--peptide loading, wash/equilibration, antibody association and antibody dissociation. B. Representative images from Triton-insoluble MC1 staining of primary neurons that received no PFF treatment (- PFF), treatment with 100 nM K18PL PFF (+ PFF), rTg4510 brain extract (Tg4510) or tTA brain extract (tTA). C. Change in Triton-insoluble MC1 staining intensity in primary neurons treated as in B, relative to untreated cultures (n = 6 wells/condition; ANOVA with Dunnett's test vs. no treatment). D. Immunodepletion of rTg4510 extracts with PHF6 or PHF13 led to significant declines in Triton-insoluble MC1staining (Red bar) in neurons relative to control antibody (ANOVA with Dunnett's test vs. Control IgG). Also displayed are total tau (black bar) and p-tau levels (blue bar) measured by ELISA after immunodepletion (*p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 6 Heavy PrP accumulation and complete absence of p.tau immunopositivity in the hippocampus of 101LL mice inoculated with 101L recombinant PrP fibrils. PrP deposits in the corpus callosum and hippocampus of 101LL mice inoculated with recombinant PrP fibrils (a, b) insert in (b) thioflavin staining of the dentate gyrus showing amyloid staining. Absence of p.tau accumulation in adjacent sections probed with anti-phosphorylated tau antibody AT8 (c, d). Images obtained after staining with anti-PrP antibody 6H4 (a, b) and anti-p.tau antibody AT8 (c, d) and counterstained with haematoxylin. Scale bar = 500 mum (c) corresponding to panels a and c; scale bar = 200 mum (d) corresponding to panels (b,d). Thioflavin-s fluorescent plaques insert in panel b (magnification 20 x). Fig. 6
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2. TFEB mediates the release of truncated mutant tau in primary neurons. (a) Schematic of tau protein and epitopes of tau antibodies used for ELISA: Tau5 (aa 210-241), K9JA (aa 243-441), and BT2 (aa 194-198). (b-d) Extracellular (b) and intracellular (c) tau levels from cultured media or cell lysates, respectively, of WT and TFEB cKO neurons at 18 days in vitro (DIV) and quantified by ELISA (Tau5/BT2 combination). (d) Extra/intracellular tau ratio. Two tailed Student's t-test. (n=6 of 2 experiments). (e-g) Extracellular (e) and intracellular (f) tau levels from cultured media or cell lysates of AAV-tau-P301L infected WT and TFEB cKO neurons at 18 days in vitro (DIV) and quantified by ELISA (Tau5/BT2 or K9JA/BT2 combination). (g) Extra/intracellular tau ratio. Two ways ANOVA followed by Sidak's multiple comparisons test. (n=6 of 2 experiments). ( h ) Biochemical characterization of lysosomes isolated by anti-FLAG immunoprecipitation (LysoIP) using antibodies against: lysosomal membrane LAMP1, lysosomal lumen CTSD and non-lysosomal cytosolic protein CDK5 and nuclear protein HDAC9. ( i-k ) Lysosomal (i) and cytosolic (j) tau levels from AAV-tau-P301L infected WT and TFEB cKO neurons at 18 days in vitro (DIV) and quantified by ELISA (Tau5/BT2 or K9JA/BT2 combination). (k) Lysosomal/cytosolic tau ratio. Two tailed Student's t-test. (n=4 of 2 experiments). Data are presented as mean +- SEM. NS, not significant; **P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 Pathological N-terminal tau truncation occurs in eyes of symptomatic Tg2576 mice and is successfully immunodepleted by 12A12mAb systemic delivery. a Study design. 6-month-old Tg2576 Alzheimer's disease (AD) mice were intravenously (i.v.) injected with 12A12mAb or mouse IgG (isotype control). On day 15, mice were sacrificed and eyes were used for biochemical (Western blotting) and morphological (immunofluorescence) evaluations. Wild type (WT) mice immunized with vehicle (saline) or mouse IgG under the same experimental conditions (antibody dosage, time of treatment, administration route) were used as controls. Picture was assembled by means of Biorender online software ( https://biorender.com ). Western blotting analyses ( b ) and semi-quantitative densitometric analysis (n = 6) ( c ) carried out on soluble extracts from three experimental groups (wild-type, Tg2576 and Tg2576 + mAb) showing the presence of the NH 2 htau peptide in retina and vitreous body of Tg2576 mice and its 12A12mAb-mediated neutralization following i.v. administration. Filters were probed with three different tau antibodies reacting against different epitopes located around the extremity and middle N-terminal end of protein, including caspase-cleaved protein (CCP)-NH 2 tau (26-36aa) [, ], BT2 (194-198aa) and DC39N1 (45-73aa). beta-actin was used as loading control. Arrows on the right side indicate the molecular weight (kDa) of bands calculated from migration of standard proteins. Statistically sig
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Detection of tau fragments in human CSF. Human control and AD CSF subjected to RP-HPLC, fractions collected and run on SDS-PAGE gels followed by western-blotting with different tau antibodies. A) HT7 (mid domain antibody). B) Tau 12 (N-terminal antibody). C) K9JA (C-terminal microtubule repeat domain antibody). D) IgG1 isotype control. On each blot, human recombinant tau441 (tau) is included in lane 1 and molecular weight markers (mw) in lane 2 followed by the HPLC fractions from 1 to 6 or HPLC fractions 7 to 11. Fractions 1 and 2 were pooled and run as a single sample, while fractions 3-10 were run as individual samples. Control CSF (C) and AD CSF (D) samples for each fraction were run side by side for comparison.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
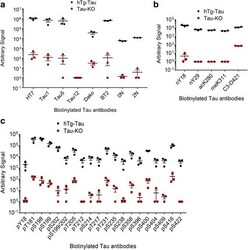
- Experimental details
- Fig. 9 An electrochemiluminescence ELISA detects tau PTMs in hTg-Tau mouse brain lysates. Sandwich ELISA assays using biotinylated antibodies for capture and sulfo-tagged Tau-12 antibody for detection demonstrate strong and specific signals for all antibodies in the panel. a Tau-12 cannot be used as capture and detection antibody at the same time, while all other total tau and tau isoform-specific antibodies in the panel are suitable to detect tau in hTg-Tau brain lysate. b All non-phospho-PTM modifications in the panel can be detected in hTg-Tau brain lysate. c All phospho-specific tau antibodies in the panel give signal from hTg-Tau brain lysate
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 Exosomes from human iPSC-derived neurons contain detectable levels of full-length, aggregation-competent tau. ( A ) Flow chart describing the centrifugation procedure used to isolate exosomes from conditioned medium (CM). The speed, duration and temperature of each centrifugation step are indicated to the right of the arrows. Immediately after collection, CM was centrifuged at 300x g to remove dead cells and then 2000 g to remove cellular debris. The clarified CM was then centrifuged at 10,000x g to pellet microvesicles. The resulting supernatant (S3) was centrifuged at 100,000x g for 70 min to pellet exosomes, and these were washed once in PBS. The wash supernatant was discarded and the final exosomal pellet was lysed in glycine/MPER buffer. CM from two separate iN cultures, designated Experiment (Exp) 1 and Exp 2, was collected and processed as described. A portion of exosomal pellet (EP) lysate was analyzed by Western blotting alongside thecell lysates (CL) of iNs from which the exosomes were derived ( B ). Antibodies used are indicated on the left and the migration of molecular weight standards (in kDa) is on the right. Tau was measured in CM (2000 g (blue) and 100,000 g (red) supernatants and the lysed 100,000 g exosome pellet (purple) using our mid-region ( C ) and full-length (FL) ( D ) ELISAs. Concentrations represent the amount of analyte present in 1 mL of starting iN CM and account for dilution and concentration steps. Results for each culture (Exp
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Exosomes from human CSF contain detectable amounts of full-length, aggregation-competent tau. ( A ) CSF samples from six healthy controls were processed by serial centrifugation, as described in Figure 3 A. Free tau was measured in CSF in 10,000 g supernatant (blue) 100,000 g supernatant (red), and the 100,000 g pellet (purple). The majority of tau is free-floating, while a small amount of tau is recovered in the exosomal pellet of 4 out of 6 samples. Values represent the mean of technical replicates. ( B , C ) Two pools of CSF, designated Pool A and Pool B, were processed as above and the 2,000 g supernatants (blue) 100,000 g supernatants (red), and 100,000 g pellets (purple) analyzed using our mid-region ELISA ( B ) and FL Simoa assay ( C ). Concentrations represent the amount of analyte present in 1 mL of starting cerebrospinal fluid (CSF) and are corrected to account for dilutions incurred during the various processing steps. Results in ( A ) represent the mean of two technical replicates, and results in ( B , C ) indicate the mean +- SD of technical replicates of two independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Blood-derived neuronal exosomes contain several different forms of tau. ( A ) Our in-house BT2-Tau5 ELISA detected tau in all plasma neuron-derived exosomes studied. On the day of testing the LLoQ for the assay was 7.81 pg/mL. ( B ) Full-length tau was detected with an in-house Simoa-based assay which on the day of testing had an LLoQ of 0.74 pg/mL. (C) The concentration of ptau-181 was measured with the Innotest commercial kit, which, on the day of testing, had an LLoQ of 24.5 pg/mL. For each assay, the LLoQ is indicated with a red line.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3 Oligomerization of tau can be monitored with Tau12-Tau12 or T22-Tau12 ELISA. a) Fluorescence intensities of ThT assays showing aggregation of recombinant full-length tau over time. Seeds alone (tau aa256-368), buffer alone and full-length tau alone were used as controls. The signal for tau with seeds increases exponentially until app. 8 h of incubation ( n = 3). Analysis of aggregates by b) Tau12-Tau12 ELISA assay and c) T22-Tau12 ELISA assay. Both methods yield a higher signal for tau with seeds after 48 h of incubation, which is abolished after boiling in SDS-containing buffer ( n = 3). d) Dot blot analysis of native samples with T22 antibody: seeds alone, tau alone and tau with seeds at 0 h and 48 h. Two-way Anova for b and c: **, p < 0.01, ***, p < 0.001, ****, p < 0.0001
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 ELISA assays and atomic force microscopy (AFM) reveal more abundant tau oligomers in Braak III-IV ECs. Comparison of ELISA counts from Braak 0-I controls with a) Braak II entorhinal cortices (EC), hippocampi (Hip), and temporal cortices (TC) and b, c) Braak III-IV EC, Hip and TC, using Tau12-Tau12 ( a, b ) or T22-Tau12 ( c ) assays. d) Representative AFM images of eluates after Tau12 immunoprecipitation; left: eluate without brain lysate (negative control), middle: eluate of Braak 0 EC, right: eluate of Braak IV EC. Scale bars represent 200 nm. e) Relative frequencies of cluster heights detected from AFM scans of two Braak 0 and two Braak IV EC tissue samples shows increase of clusters > 10 nm in Braak IV samples. Number of clusters detected: Braak 0-I: 1343, Braak III-IV: 1053. Student's t-tests: *, p < 0.05, ***, p < 0.001
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 PD-1 treatment did not enhance tau clearance. (A-F) PD-1 antibody treatment had no effect on soluble phospho-tau (PHF-1), total tau (Tau-5), total human tau (CP27) or ratios thereof on Western blots of tauopathy mouse brain homogenates, compared to control group. (A,G) PD-1 treatment had no effect on insoluble Western blot tau levels in the brain. (H,I) Enzyme-linked immunosorbent assay (ELISA) analyses of the same brain homogenates analyzed by Western blots confirmed lack of efficacy of the PD-1 therapy in clearing tau from the brain. Each bar represents the group average +/- SEM. As shown in individual scatterplots, 10 PD-1 treated- and eight control IgG mice were analyzed in each blot or ELISA assay. The total insoluble tau ELISA data failed normality test (IgG group) and was, therefore, analyzed by the Mann-Whitney test. All the other data passed the normality test and were, therefore, analyzed by unpaired t -test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 Quantification of (A) Abeta 42 , (B) tau 441 and (C) p-tau 181 in human CSF samples using the developed assay (left) and a commercially available ELISA kit (right). Error bars, standard error of mean; n = 3. Average net intensity = ((1x1 square pixel of 50 individual MNCs) - (1x1 square pixel of 50 individual background area on the TIRFM images)) / 50.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA