13-6400
antibody from Invitrogen Antibodies
Targeting: MAPT
DDPAC, FLJ31424, FTDP-17, MAPTL, MGC138549, MSTD, MTBT1, MTBT2, PPND, PPP1R103, tau
 Western blot
Western blot ELISA
ELISA Immunocytochemistry
Immunocytochemistry Immunoprecipitation
Immunoprecipitation Immunohistochemistry
Immunohistochemistry Flow cytometry
Flow cytometry Immunoelectron microscopy
Immunoelectron microscopy Other assay
Other assayAntibody data
- Antibody Data
- Antigen structure
- References [35]
- Comments [0]
- Validations
- Immunocytochemistry [4]
- Immunohistochemistry [4]
- Flow cytometry [1]
- Other assay [18]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 13-6400 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Tau Monoclonal Antibody (T46)
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- This antibody recognizes an epitope near the C-terminal region (404-441 aa) of human tau. It reacts with all 7 isoforms of human tau (binding-competent and binding-incompetent), A68 (an abnormal phosphorylated derivative of tau), bovine tau, and human NeuroFibrillary Tangles (NFTs). Reactivity is independent of the state of tau phosphorylation. Some cross-reactivity with MAP2 is also observed. This antibody is suitable for immunohistochemical staining of Bouin's-fixed or alcohol-fixed, paraffin-embedded or frozen tissue sections.
- Reactivity
- Human, Mouse, Rat, Bovine, Chicken/Avian, Rabbit, Xenopus
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- T46
- Vial size
- 100 μg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references ATase inhibition rescues age-associated proteotoxicity of the secretory pathway.
Dephosphorylation Passivates the Seeding Activity of Oligomeric Tau Derived From Alzheimer's Brain.
UBE4B, a microRNA-9 target gene, promotes autophagy-mediated Tau degradation.
α- Linolenic acid modulates phagocytosis and endosomal pathways of extracellular Tau in microglia.
Asparagine residue 368 is involved in Alzheimer's disease tau strain-specific aggregation.
Basic Limonoid modulates Chaperone-mediated Proteostasis and dissolve Tau fibrils.
Melatonin Reduces GSK3β-Mediated Tau Phosphorylation, Enhances Nrf2 Nuclear Translocation and Anti-Inflammation.
Phagocytosis of full-length Tau oligomers by Actin-remodeling of activated microglia.
Integrative approach to sporadic Alzheimer's disease: deficiency of TYROBP in a tauopathy mouse model reduces C1q and normalizes clinical phenotype while increasing spread and state of phosphorylation of tau.
Suppression of glioblastoma by a drug cocktail reprogramming tumor cells into neuronal like cells.
Porphyromonas gingivalis in Alzheimer's disease brains: Evidence for disease causation and treatment with small-molecule inhibitors.
Impacts of Acute Hypoxia on Alzheimer's Disease-Like Pathologies in APP(swe)/PS1(dE9) Mice and Their Wild Type Littermates.
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Both secreted and the cellular levels of BDNF attenuated due to tau hyperphosphorylation in primary cultures of cortical neurons.
A Dual Pathogenic Mechanism Links Tau Acetylation to Sporadic Tauopathy.
Conserved Lysine Acetylation within the Microtubule-Binding Domain Regulates MAP2/Tau Family Members.
Mutations in LRRK2 impair NF-κB pathway in iPSC-derived neurons.
Improved proteostasis in the secretory pathway rescues Alzheimer's disease in the mouse.
Globular Glial Mixed Four Repeat Tau and TDP-43 Proteinopathy with Motor Neuron Disease and Frontotemporal Dementia.
Biochemical classification of tauopathies by immunoblot, protein sequence and mass spectrometric analyses of sarkosyl-insoluble and trypsin-resistant tau.
Different pathways of molecular pathophysiology underlie cognitive and motor tauopathy phenotypes in transgenic models for Alzheimer's disease and frontotemporal lobar degeneration.
Tamoxifen inhibits CDK5 kinase activity by interacting with p35/p25 and modulates the pattern of tau phosphorylation.
Total body 100-mGy X-irradiation does not induce Alzheimer's disease-like pathogenesis or memory impairment in mice.
Dissociation of tau toxicity and phosphorylation: role of GSK-3beta, MARK and Cdk5 in a Drosophila model.
Multiple Eph receptors and B-class ephrins regulate midline crossing of corpus callosum fibers in the developing mouse forebrain.
Abeta and tau form soluble complexes that may promote self aggregation of both into the insoluble forms observed in Alzheimer's disease.
Neurodegeneration in heterozygous Niemann-Pick type C1 (NPC1) mouse: implication of heterozygous NPC1 mutations being a risk for tauopathy.
Fyn phosphorylates human MAP-2c on tyrosine 67.
Proteolysis of non-phosphorylated and phosphorylated tau by thrombin.
Caspase-cleavage of tau is an early event in Alzheimer disease tangle pathology.
Pathogenic implications of mutations in the tau gene in pallido-ponto-nigral degeneration and related neurodegenerative disorders linked to chromosome 17.
Pathogenic implications of mutations in the tau gene in pallido-ponto-nigral degeneration and related neurodegenerative disorders linked to chromosome 17.
The neuropathology of a chromosome 17-linked autosomal dominant parkinsonism and dementia ("pallido-ponto-nigral degeneration").
The neuropathology of a chromosome 17-linked autosomal dominant parkinsonism and dementia ("pallido-ponto-nigral degeneration").
Cognitive, neuroimaging, and pathological studies in a patient with Pick's disease.
Murie M, Peng Y, Rigby MJ, Dieterich IA, Farrugia MA, Endresen A, Bhattacharyya A, Puglielli L
Communications biology 2022 Feb 25;5(1):173
Communications biology 2022 Feb 25;5(1):173
Dephosphorylation Passivates the Seeding Activity of Oligomeric Tau Derived From Alzheimer's Brain.
Wu R, Li L, Shi R, Zhou Y, Jin N, Gu J, Tung YC, Liu F, Chu D
Frontiers in molecular neuroscience 2021;14:631833
Frontiers in molecular neuroscience 2021;14:631833
UBE4B, a microRNA-9 target gene, promotes autophagy-mediated Tau degradation.
Subramanian M, Hyeon SJ, Das T, Suh YS, Kim YK, Lee JS, Song EJ, Ryu H, Yu K
Nature communications 2021 Jun 2;12(1):3291
Nature communications 2021 Jun 2;12(1):3291
α- Linolenic acid modulates phagocytosis and endosomal pathways of extracellular Tau in microglia.
Desale SE, Chinnathambi S
Cell adhesion & migration 2021 Dec;15(1):84-100
Cell adhesion & migration 2021 Dec;15(1):84-100
Asparagine residue 368 is involved in Alzheimer's disease tau strain-specific aggregation.
Shimonaka S, Matsumoto SE, Elahi M, Ishiguro K, Hasegawa M, Hattori N, Motoi Y
The Journal of biological chemistry 2020 Oct 9;295(41):13996-14014
The Journal of biological chemistry 2020 Oct 9;295(41):13996-14014
Basic Limonoid modulates Chaperone-mediated Proteostasis and dissolve Tau fibrils.
Gorantla NV, Das R, Chidambaram H, Dubey T, Mulani FA, Thulasiram HV, Chinnathambi S
Scientific reports 2020 Mar 4;10(1):4023
Scientific reports 2020 Mar 4;10(1):4023
Melatonin Reduces GSK3β-Mediated Tau Phosphorylation, Enhances Nrf2 Nuclear Translocation and Anti-Inflammation.
Das R, Balmik AA, Chinnathambi S
ASN neuro 2020 Jan-Dec;12:1759091420981204
ASN neuro 2020 Jan-Dec;12:1759091420981204
Phagocytosis of full-length Tau oligomers by Actin-remodeling of activated microglia.
Das R, Balmik AA, Chinnathambi S
Journal of neuroinflammation 2020 Jan 8;17(1):10
Journal of neuroinflammation 2020 Jan 8;17(1):10
Integrative approach to sporadic Alzheimer's disease: deficiency of TYROBP in a tauopathy mouse model reduces C1q and normalizes clinical phenotype while increasing spread and state of phosphorylation of tau.
Audrain M, Haure-Mirande JV, Wang M, Kim SH, Fanutza T, Chakrabarty P, Fraser P, St George-Hyslop PH, Golde TE, Blitzer RD, Schadt EE, Zhang B, Ehrlich ME, Gandy S
Molecular psychiatry 2019 Sep;24(9):1383-1397
Molecular psychiatry 2019 Sep;24(9):1383-1397
Suppression of glioblastoma by a drug cocktail reprogramming tumor cells into neuronal like cells.
Gao L, Huang S, Zhang H, Hua W, Xin S, Cheng L, Guan W, Yu Y, Mao Y, Pei G
Scientific reports 2019 Mar 5;9(1):3462
Scientific reports 2019 Mar 5;9(1):3462
Porphyromonas gingivalis in Alzheimer's disease brains: Evidence for disease causation and treatment with small-molecule inhibitors.
Dominy SS, Lynch C, Ermini F, Benedyk M, Marczyk A, Konradi A, Nguyen M, Haditsch U, Raha D, Griffin C, Holsinger LJ, Arastu-Kapur S, Kaba S, Lee A, Ryder MI, Potempa B, Mydel P, Hellvard A, Adamowicz K, Hasturk H, Walker GD, Reynolds EC, Faull RLM, Curtis MA, Dragunow M, Potempa J
Science advances 2019 Jan;5(1):eaau3333
Science advances 2019 Jan;5(1):eaau3333
Impacts of Acute Hypoxia on Alzheimer's Disease-Like Pathologies in APP(swe)/PS1(dE9) Mice and Their Wild Type Littermates.
Zhang F, Zhong R, Qi H, Li S, Cheng C, Liu X, Liu Y, Le W
Frontiers in neuroscience 2018;12:314
Frontiers in neuroscience 2018;12:314
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Zhou Y, Shi J, Chu D, Hu W, Guan Z, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2018;10:27
Frontiers in aging neuroscience 2018;10:27
Both secreted and the cellular levels of BDNF attenuated due to tau hyperphosphorylation in primary cultures of cortical neurons.
Atasoy İL, Dursun E, Gezen-Ak D, Metin-Armağan D, Öztürk M, Yılmazer S
Journal of chemical neuroanatomy 2017 Mar;80:19-26
Journal of chemical neuroanatomy 2017 Mar;80:19-26
A Dual Pathogenic Mechanism Links Tau Acetylation to Sporadic Tauopathy.
Trzeciakiewicz H, Tseng JH, Wander CM, Madden V, Tripathy A, Yuan CX, Cohen TJ
Scientific reports 2017 Mar 13;7:44102
Scientific reports 2017 Mar 13;7:44102
Conserved Lysine Acetylation within the Microtubule-Binding Domain Regulates MAP2/Tau Family Members.
Hwang AW, Trzeciakiewicz H, Friedmann D, Yuan CX, Marmorstein R, Lee VM, Cohen TJ
PloS one 2016;11(12):e0168913
PloS one 2016;11(12):e0168913
Mutations in LRRK2 impair NF-κB pathway in iPSC-derived neurons.
López de Maturana R, Lang V, Zubiarrain A, Sousa A, Vázquez N, Gorostidi A, Águila J, López de Munain A, Rodríguez M, Sánchez-Pernaute R
Journal of neuroinflammation 2016 Nov 18;13(1):295
Journal of neuroinflammation 2016 Nov 18;13(1):295
Improved proteostasis in the secretory pathway rescues Alzheimer's disease in the mouse.
Peng Y, Kim MJ, Hullinger R, O'Riordan KJ, Burger C, Pehar M, Puglielli L
Brain : a journal of neurology 2016 Mar;139(Pt 3):937-52
Brain : a journal of neurology 2016 Mar;139(Pt 3):937-52
Globular Glial Mixed Four Repeat Tau and TDP-43 Proteinopathy with Motor Neuron Disease and Frontotemporal Dementia.
Takeuchi R, Toyoshima Y, Tada M, Tanaka H, Shimizu H, Shiga A, Miura T, Aoki K, Aikawa A, Ishizawa S, Ikeuchi T, Nishizawa M, Kakita A, Takahashi H
Brain pathology (Zurich, Switzerland) 2016 Jan;26(1):82-94
Brain pathology (Zurich, Switzerland) 2016 Jan;26(1):82-94
Biochemical classification of tauopathies by immunoblot, protein sequence and mass spectrometric analyses of sarkosyl-insoluble and trypsin-resistant tau.
Taniguchi-Watanabe S, Arai T, Kametani F, Nonaka T, Masuda-Suzukake M, Tarutani A, Murayama S, Saito Y, Arima K, Yoshida M, Akiyama H, Robinson A, Mann DMA, Iwatsubo T, Hasegawa M
Acta neuropathologica 2016 Feb;131(2):267-280
Acta neuropathologica 2016 Feb;131(2):267-280
Different pathways of molecular pathophysiology underlie cognitive and motor tauopathy phenotypes in transgenic models for Alzheimer's disease and frontotemporal lobar degeneration.
Melis V, Zabke C, Stamer K, Magbagbeolu M, Schwab K, Marschall P, Veh RW, Bachmann S, Deiana S, Moreau PH, Davidson K, Harrington KA, Rickard JE, Horsley D, Garman R, Mazurkiewicz M, Niewiadomska G, Wischik CM, Harrington CR, Riedel G, Theuring F
Cellular and molecular life sciences : CMLS 2015 Jun;72(11):2199-222
Cellular and molecular life sciences : CMLS 2015 Jun;72(11):2199-222
Tamoxifen inhibits CDK5 kinase activity by interacting with p35/p25 and modulates the pattern of tau phosphorylation.
Corbel C, Zhang B, Le Parc A, Baratte B, Colas P, Couturier C, Kosik KS, Landrieu I, Le Tilly V, Bach S
Chemistry & biology 2015 Apr 23;22(4):472-482
Chemistry & biology 2015 Apr 23;22(4):472-482
Total body 100-mGy X-irradiation does not induce Alzheimer's disease-like pathogenesis or memory impairment in mice.
Wang B, Tanaka K, Ji B, Ono M, Fang Y, Ninomiya Y, Maruyama K, Izumi-Nakajima N, Begum N, Higuchi M, Fujimori A, Uehara Y, Nakajima T, Suhara T, Ono T, Nenoi M
Journal of radiation research 2014 Jan 1;55(1):84-96
Journal of radiation research 2014 Jan 1;55(1):84-96
Dissociation of tau toxicity and phosphorylation: role of GSK-3beta, MARK and Cdk5 in a Drosophila model.
Chatterjee S, Sang TK, Lawless GM, Jackson GR
Human molecular genetics 2009 Jan 1;18(1):164-77
Human molecular genetics 2009 Jan 1;18(1):164-77
Multiple Eph receptors and B-class ephrins regulate midline crossing of corpus callosum fibers in the developing mouse forebrain.
Mendes SW, Henkemeyer M, Liebl DJ
The Journal of neuroscience : the official journal of the Society for Neuroscience 2006 Jan 18;26(3):882-92
The Journal of neuroscience : the official journal of the Society for Neuroscience 2006 Jan 18;26(3):882-92
Abeta and tau form soluble complexes that may promote self aggregation of both into the insoluble forms observed in Alzheimer's disease.
Guo JP, Arai T, Miklossy J, McGeer PL
Proceedings of the National Academy of Sciences of the United States of America 2006 Feb 7;103(6):1953-8
Proceedings of the National Academy of Sciences of the United States of America 2006 Feb 7;103(6):1953-8
Neurodegeneration in heterozygous Niemann-Pick type C1 (NPC1) mouse: implication of heterozygous NPC1 mutations being a risk for tauopathy.
Yu W, Ko M, Yanagisawa K, Michikawa M
The Journal of biological chemistry 2005 Jul 22;280(29):27296-302
The Journal of biological chemistry 2005 Jul 22;280(29):27296-302
Fyn phosphorylates human MAP-2c on tyrosine 67.
Zamora-Leon SP, Bresnick A, Backer JM, Shafit-Zagardo B
The Journal of biological chemistry 2005 Jan 21;280(3):1962-70
The Journal of biological chemistry 2005 Jan 21;280(3):1962-70
Proteolysis of non-phosphorylated and phosphorylated tau by thrombin.
Arai T, Guo JP, McGeer PL
The Journal of biological chemistry 2005 Feb 18;280(7):5145-53
The Journal of biological chemistry 2005 Feb 18;280(7):5145-53
Caspase-cleavage of tau is an early event in Alzheimer disease tangle pathology.
Rissman RA, Poon WW, Blurton-Jones M, Oddo S, Torp R, Vitek MP, LaFerla FM, Rohn TT, Cotman CW
The Journal of clinical investigation 2004 Jul;114(1):121-30
The Journal of clinical investigation 2004 Jul;114(1):121-30
Pathogenic implications of mutations in the tau gene in pallido-ponto-nigral degeneration and related neurodegenerative disorders linked to chromosome 17.
Clark LN, Poorkaj P, Wszolek Z, Geschwind DH, Nasreddine ZS, Miller B, Li D, Payami H, Awert F, Markopoulou K, Andreadis A, D'Souza I, Lee VM, Reed L, Trojanowski JQ, Zhukareva V, Bird T, Schellenberg G, Wilhelmsen KC
Proceedings of the National Academy of Sciences of the United States of America 1998 Oct 27;95(22):13103-7
Proceedings of the National Academy of Sciences of the United States of America 1998 Oct 27;95(22):13103-7
Pathogenic implications of mutations in the tau gene in pallido-ponto-nigral degeneration and related neurodegenerative disorders linked to chromosome 17.
Clark LN, Poorkaj P, Wszolek Z, Geschwind DH, Nasreddine ZS, Miller B, Li D, Payami H, Awert F, Markopoulou K, Andreadis A, D'Souza I, Lee VM, Reed L, Trojanowski JQ, Zhukareva V, Bird T, Schellenberg G, Wilhelmsen KC
Proceedings of the National Academy of Sciences of the United States of America 1998 Oct 27;95(22):13103-7
Proceedings of the National Academy of Sciences of the United States of America 1998 Oct 27;95(22):13103-7
The neuropathology of a chromosome 17-linked autosomal dominant parkinsonism and dementia ("pallido-ponto-nigral degeneration").
Reed LA, Schmidt ML, Wszolek ZK, Balin BJ, Soontornniyomkij V, Lee VM, Trojanowski JQ, Schelper RL
Journal of neuropathology and experimental neurology 1998 Jun;57(6):588-601
Journal of neuropathology and experimental neurology 1998 Jun;57(6):588-601
The neuropathology of a chromosome 17-linked autosomal dominant parkinsonism and dementia ("pallido-ponto-nigral degeneration").
Reed LA, Schmidt ML, Wszolek ZK, Balin BJ, Soontornniyomkij V, Lee VM, Trojanowski JQ, Schelper RL
Journal of neuropathology and experimental neurology 1998 Jun;57(6):588-601
Journal of neuropathology and experimental neurology 1998 Jun;57(6):588-601
Cognitive, neuroimaging, and pathological studies in a patient with Pick's disease.
Lieberman AP, Trojanowski JQ, Lee VM, Balin BJ, Ding XS, Greenberg J, Morrison D, Reivich M, Grossman M
Annals of neurology 1998 Feb;43(2):259-65
Annals of neurology 1998 Feb;43(2):259-65
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
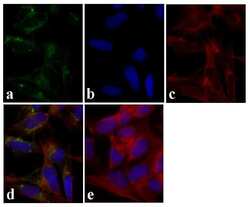
- Experimental details
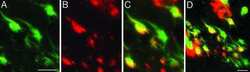
- Immunofluorescent analysis of Tau was done on 70% confluent log phase SHSY5Y cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton™ X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with Tau Mouse Monoclonal Antibody (Product # 13-6400) at 1:250 dilution in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Rabbit Anti-Mouse IgG Secondary Antibody (Product # A-11059) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing cytoplasmic localization. Panel e shows no primary antibody control. The images were captured at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
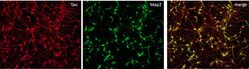
- Experimental details
- Immunofluorescent staining of MAP2 (Green) and Tau (red) on Primary Rat Cortex neurons (E18) (Product # A1084001) cultured for 14 days in the B-27 Plus Neuronal Culture System (Product # A3653401). At day 14 the cells were fixed with 4% paraformaldehyde for 15 min, permeabilized with 0.1% Triton X-100 for 30 min, and blocked with 1% BSA for 30 min at room temperature. Cells were stained with a MAP2 rabbit polyclonal antibody (Product # PA5-17646) at a dilution of 1:250, and a Tau mouse monoclonal antibody clone T46 (Product # 13-6400) at a dilution of 1:100 in 1% BSA staining buffer, overnight at 4C, and then incubated with Alexa Fluor secondary antibodies 488 donkey anti-rabbit (Product # A-21206) and 594 donkey anti-mouse (Product # A-21203) at a dilution of 1:1000 for 30 minutes at room temperature. Wash 3 times with DPBS. Stain with DAPI.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
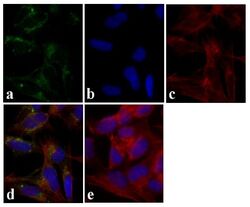
- Experimental details
- Immunofluorescent analysis of Tau was done on 70% confluent log phase SHSY5Y cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton™ X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with Tau Mouse Monoclonal Antibody (Product # 13-6400) at 1:250 dilution in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Rabbit Anti-Mouse IgG Secondary Antibody (Product # A-11059) at a dilution of 1:400 for 30 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing cytoplasmic localization. Panel e shows no primary antibody control. The images were captured at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
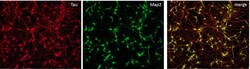
- Experimental details
- Immunofluorescent staining of MAP2 (Green) and Tau (red) on Primary Rat Cortex neurons (E18) (Product # A1084001) cultured for 14 days in the B-27 Plus Neuronal Culture System (Product # A3653401). At day 14 the cells were fixed with 4% paraformaldehyde for 15 min, permeabilized with 0.1% Triton X-100 for 30 min, and blocked with 1% BSA for 30 min at room temperature. Cells were stained with a MAP2 rabbit polyclonal antibody (Product # PA5-17646) at a dilution of 1:250, and a Tau mouse monoclonal antibody clone T46 (Product # 13-6400) at a dilution of 1:100 in 1% BSA staining buffer, overnight at 4C, and then incubated with Alexa Fluor secondary antibodies 488 donkey anti-rabbit (Product # A-21206) and 594 donkey anti-mouse (Product # A-21203) at a dilution of 1:1000 for 30 minutes at room temperature. Wash 3 times with DPBS. Stain with DAPI.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
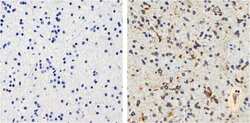
- Experimental details
- Immunohistochemistry analysis of Tau showing staining in the cytoplasm and membrane of paraffin-embedded human astroglioma Tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Tau mouse monoclonal antibody (Product # 13-6400) diluted in 3% BSA-PBS at a dilution of 1:100 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
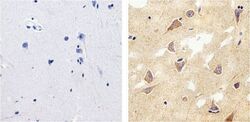
- Experimental details
- Immunohistochemistry analysis of Tau showing staining in the cytoplasm and membrane of paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Tau mouse monoclonal antibody (Product # 13-6400) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
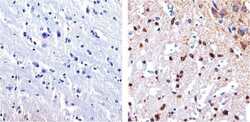
- Experimental details
- Immunohistochemistry analysis of Tau showing staining in the cytoplasm and membrane of paraffin-embedded mouse brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Tau mouse monoclonal antibody (Product # 13-6400) diluted in 3% BSA-PBS at a dilution of 1:100 overnight at 4°C in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
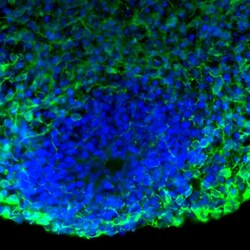
- Experimental details
- Immunofluorescent analysis of Tau in human iPSC-derived forebrain organoids derived at Day 40. The organoids were fixed with 4% PFA for 1 hour at room temperature, followed by incubation with 30% sucrose solution overnight at 4°C. The organoids were then embedded in OCT and cryosectioned at 5 µm, permeabilized with 0.2% Triton X-100 for 20 min, and blocked with 10% donkey serum in PBS for 30 min at room temperature. Organoid slices were stained with a Mouse Tau monoclonal antibody (green; Product # 13-6400) at a dilution of 1:500 in blocking buffer overnight at 4°C, and then incubated with Donkey anti-Mouse Alexa Fluor 488 (Product # R37114) at a dilution of 1:1000 as well as DAPI (blue; 1:25000) in blocking solution at room temperature for 1 hour. Images were taken at 20X magnification. Data courtesy of Dr. Zhexing Wen at Emory University.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
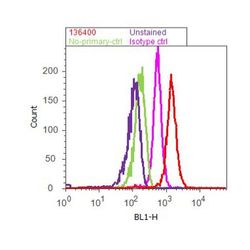
- Flow cytometry analysis of Tau was done on SH-SY5Y cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Tritonª X-100 for 20 minutes, and blocked with 5% BSA for 1 hour at room temperature. Cells were labeled with Tau Mouse Monoclonal Antibody (136400, red histogram) or with mouse isotype control (pink histogram) at 1-3 µg/million cells in 2.5% BSA. After incubation at room temperature for 2-3 hours, the cells were labeled with Alexa Fluor¨ 488 Rabbit Anti-Mouse Secondary Antibody (A11059) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune¨ Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
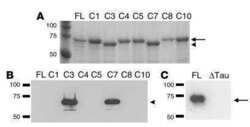
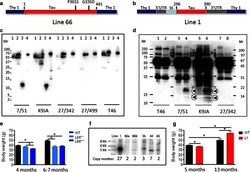
- Fig. 1 Constructs used for microinjection and expression parameters of protein for full-length mutant and truncated tau. a The L66 cDNA construct contains human tau cDNA coding for the longest human CNS tau isoform (htau40; 441 amino acids) with the point mutations to make the amino acid changes P301S and G335D. b The L1 cDNA construct contains human tau cDNA coding for amino acid residues 296-390 with signal sequence and related sequences in murine Thy-1 expression cassette. c Immunoblot analysis of 6-month-old WT ( lane 1 ) and L66 ( lane 2 ) mouse brains with recombinant truncated dGAE ( lane 3 ) and full-length htau40 ( lane 4 ) for reference. The faint 25-kDa band seen in lane 1 was only seen with the wild-type mouse brain and was observed when the secondary anti-mouse antibody was used in the absence of primary tau antibody. The 70-kDa band reflects full-length mutant tau and is detected by all of the antibodies shown. Note that the 55-kDa band, that is not detected by the human-specific mAb 27/499, represents endogenous mouse tau. The position of markers indicates relative molecular mass ( M r ) in kDa. d Immunoblot characterisation of tau in 4-month-old L1 ( even numbers ) and WT mice ( odd numbers ). Protein extracts from whole-brain lysates were separated by SDS-PAGE and immunoblots stained with antibodies 7/51 and K9JA for epitopes within the repeat domain of tau. Immunoreactive bands at 12- and 18-kDa ( arrowheads ) were detected in L1 mice ( lanes 2 , 4 , 6 and 8
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
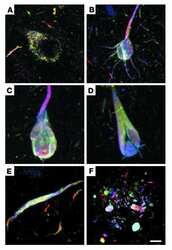
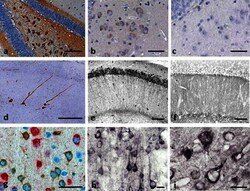
- Fig. 2 Tau immunohistochemistry of transgenic tau L66 ( a , b , e , f , i ) and L1 ( c , d , g , h ) mice. Tau immunoreactivity with mAb 7/51 is strong in L66 with prominent intracellular staining in the hilus/CA4 sectors of the hippocampus ( a ) and in other areas (see Fig. 4 ). In contrast, mAb AT8 immunoreactivity is seen more frequently in elderly mice with staining apparent in long processes of CA1 ( d ). Neurons in visual cortex ( g ) are stained differentially with both mAb 7/51 ( brown ) and AT8 ( red ). Amorphous intracellular tau immunoreactivity, visualised using mAb 7/51, in the hilus/CA4 of L1 mice ( b ) is much weaker than for L66, and absent from the corresponding region in WT mice ( c ). The presence of pS404 phospho-tau immunoreactivity was seen in hippocampus and cortex and was more intense for L1 late in life. At 3 and 9 months old, L1 and WT animals were similar. It was only at 15 months of age that a prominent difference between L1 and wild-type was observed, with CA1 labelling for pS404-Tau greater in L1 ( e ) than WT ( f ). The staining of neurons with this mAb indicates that there are granular accumulations of pS404-Tau immunoreactivity in long processes ( h , i ). Scale bars 50 um ( a , d ); 25 um ( e - g ); 20 um ( b , c ) and 10 um ( h , i )
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
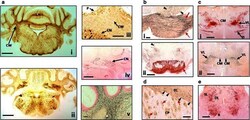
- Fig. 4 Distribution of aggregated tau in L66 transgenic mice. a Staining for astrocytes and axons in the cerebellum of L66 mice aged 6-7 months. a - i Cross-sections through the mid-cerebellar region and underlying brainstem stained for GFAP in L66 shows immunoreactivity within the cerebellar white matter (CW). Cellular staining for phospho-tau using mAb AT8 ( a-ii ) was identified in both the cerebellar cortex and in the deeper cerebellar nuclei ( a - iii , CN), but also some weak staining in processes running through the white matter, and non-specific immunoreactivity in the Purkinje cell layer (P). Deep nuclei were stained also with mAb 7/51, but no processes were immunopositive ( a - iv ). Axons stained darkly by the amino cupric silver stain indicate injury to axonal integrity in L66 mice ( a - v ). b Degeneration of fibres and expression of human tau in pons and ventral tegmentum. Axonal injury was observed in pons using amino cupric silver staining ( b - i ). While black arrows indicate scattered positivity for longitudinally oriented axons in tegmental regions, strong labelling of transverse fibres ( red arrows ) occurred in the cerebral peduncle. Somata/processes were also densely labelled for phospho-tau using AT8 ( b - ii ) in both pontine axons and cells ( red arrow ) and tegmental neurons ( arrowhead ). c Anterior thalamic expression of tau. Dense AT8 labelling ( c - i ) was observed in cells and axons of the ventrolateral thalamic nucleus (VL) and zona incerta (
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
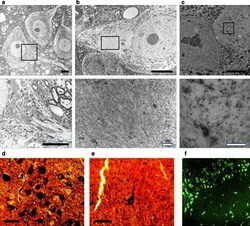
- Fig. 5 Filamentous tau in L66. Electronmicroscopy of spinal cord from a WT and b L66 mice and c anti-tau immunoelectronmicroscopy of L66. Dense bundles of filaments were found in large neurons in L66 but not WT mice and these were immunodecorated with gold particles labelling tau. The lower panel of images are selected magnifications from the upper panel as indicated; scale 5 um ( solid bars ) and 0.5 um ( open bars ). Bielschowsky silver staining for tau aggregates was more abundant in cortical sections from L66 ( d ) than L36 ( e ) mice. In addition, Primulin-stained neurons provide further evidence of the formation of filamentous tau aggregates in cortical tissue from L66 ( f )
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
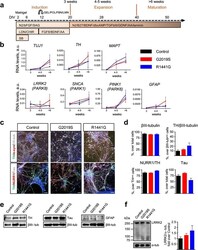
- Fig. 2 Characterization of iPSC-derived DA neurons with LRRK2 mutations. a Diagram showing the DA differentiation protocol used for neural induction of human iPSC lines. b Temporal gene expression analyzed by qRT-PCR at three time points: induction (3 weeks), expansion (4-5 weeks), and maturation (>6 weeks). Each point represents the mean +- SEM of at least two independent differentiation experiments. c Representative images of mature neuronal cultures showing expression of neuronal (betaIII-tubulin, Tau, and alpha-synuclein) and dopaminergic (TH, NURR1) markers. Nuclei were counterstained with Hoechst. Scale bars : 50 mum. d Quantification of immunostainings. Data are represented as mean +- SEM of counts from at least two different lines for each genotype. e Representative western blot analyses of TH, Tau, and GFAP with betaIII-tubulin as loading control in iPSC-derived mature neurons. f Representative immunoblots and quantification of LRRK2 expression in mature neuronal cultures. alpha-tubulin was the loading control and data were normalized to control WT neurons. Bars represent the mean +- SEM of at least two different lines per genotype. DIV days in vitro, GEL gelatin, POL poly-ornithine, FBN fibronectin, LMN laminin, N2 N2 supplement, bFGF basic fibroblast growth factor, SAG smoothened agonist, LDN LDN-193189, CHIR CHIR99021, SB SB431542, BDNF brain-derived neurotrophic factor, AA ascorbic acid, B27 B27 supplement, dbcAMP dibutyryl cyclic adenosine monophosphate, TGFbeta
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
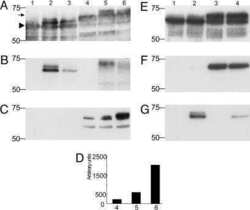
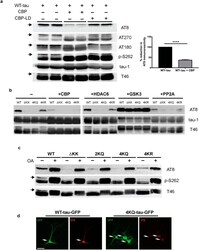
- Figure 1 Tau acetylation modulates tau phosphorylation at specific epitopes. (a-c ) Immunoblot analysis and quantification using the indicated tau antibodies for (a ) cells expressing WT tau-T40 and either an active form of CBP or a catalytically inactive form of CBP (CBP-LD), ( b ) cells co- expressing tau-T40 (WT, DeltaKK, 4KQ, or 4KR) and tau modifying enzymes, and ( c ) cells expressing tau-T40 (WT, DeltaKK, 2KQ, 4KQ, or 4KR) and treated with control (DMSO) or okadaic acid (OA), where indicated. Solid black arrows highlight the ~75 kDa phospho-tau species that is reduced upon acetylation. Statistical significance was assessed using a student t-test (****p < 0.0001). Cropped images from full size immunoblots are provided in panels a-c. Full-length immunoblots are presented in Supplementary Fig. S1 . ( d ) Immunofluorescence microscopy of primary cortical neurons expressing WT-tau-GFP (left) or 4KQ-tau-GFP (right) detecting phosphorylated tau (AT8). White arrows identify transfected neurons. Scale bar, 50 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
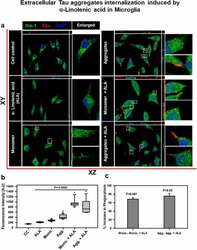
- Figure 2. Extracellular Tau aggregates internalization, induced by alpha-linolenic acid in microglia . Internalization of hTau40 recombinant Tau in Iba-1 positive microglia. (a) Cells were incubated with hTau40 aggregates species, hTau40 monomer species (1 uM) alone, and along with the alpha-linolenic acid (40 uM) for 24 h at 37degC. The cells were fixed after 24 h and stained with anti-Iba-1 antibody (green) and T46 Tau antibody (red) and observed by fluorescence microscopy; scale bar is 20 um. The orthogonal images denote the internalization of Tau, indicated at X and Y plane to visualize the intracellular position. The enlarged panel indicates internalized Tau. The images were taken with the Zeiss fluorescence microscope with Apotome 2.0. (b) Quantification of mean intensity of internalized Tau in microglia cells showing the extent of internalization Tau in microglia cells, which is highly significant ( P < 0.001) compared with cell control (no treatment) and ALA-treated cells. (c) Percentage increase in phagocytosis of hTau40 in microglia after ALA exposure to cells; percentage increase in aggregates compared to aggregates with ALA-exposed groups and in monomer compared to monomer with ALA groups calculated from the intracellular intensity of Tau among the groups; significance is P = 0.02 and 0.007, respectively
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
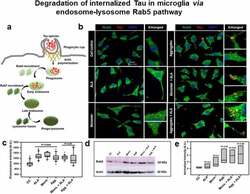
- Figure 3. Degradation of internalized Tau in microglia via endosome-lysosome Rab5 pathway . A microglia cell was exposed to hTau40 monomer and aggregates in the presence and absence of ALA and observed for the levels of Rab5 (green) and Tau (red) by fluorescence microscopy. The degradation of internalized Tau was studied with the early endosomal marker and late endosomal markers. (a) In the pathway, the maturation of phagocytic vesicle takes place that can be marked with the early endosomal marker Rab5 and late endosomal marker Rab7. We observed the colocalization of internalized Tau with endosomal markers (Rab5) to trace the degradation of internalized Tau. (b) The fluorescence microscopy images indicate the levels of endosomal markers and their colocalization with internalized Tau. The enlarged panel indicates specific area in the cells showing colocalization between Rab5 and Tau; scale bar is 20 mum. (c) The quantification of mean intensity of endosomal markers was carried out with ZEN 2.3 software; significance is P < 0.05. (d) Expression analysis of early endosomal marker (Rab5) was observed by western blot after various treatments of hTau40 monomer, aggregates, and ALA after 24 h. (e) Quantification of protein bands is plotted as intensity and normalized with beta-actin as a loading control
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
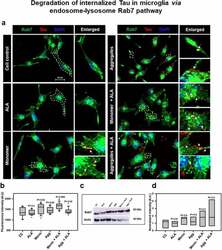
- Figure 4. Degradation of internalized Tau in microglia via endosome-lysosme Rab7 pathway . Fate of internalized Tau was observed with the help of late endosomal marker Rab7 by fluorescence microscopy. (a) The colocalization of internalized Tau was observed with late endosomal marker Rab7, and the enlarged area indicates the specific area of colocalization inside the cell. The white arrowmarks denote the position and colocalization of internalized Tau with Rab7; scale bar is 20 mum. (b) The intensity analysis of Rab7 plotted as a mean intensity of Rab7; significance is P < 0.05. (c) Expression profile of Rab7 was analyzed by western blot after exposure of extracellular Tau. (d) The quantification of protein bands was carried out by calculating the intensity and normalized with beta-actin as a loading control
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
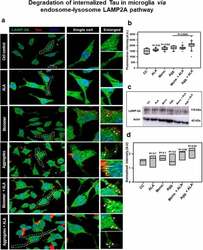
- Figure 5. Degradation of internalized Tau in microglia via endosome-lysosome LAMP-2A pathway . Last step of degaradation pathway includes fusion of late endosome with lysosome studied by fluorescence microscopy. (a) Internalization of extracellular Tau (red) studied with its colocalization with LAMP-2A (green) after 24 h of ALA and extracellular Tau exposure. The images indicate levels of LAMP-2A and its colocalization with Tau. Enlarged panel indicates that specific area from the representative image showing colocalization of Tau and LAMP-2A, indicated with white triangles. Scale bar is 20 mum. (b) Intracellular intensity of LAMP-2A was calculated from immunofluorescence images and plotted as mean intensity inside the cell. The significance is P < 0.05. (c) Levels of LAMP-2A were detected by western blot after ALA and Tau exposure. (d) Quantification of intensity of protein bands, normalized with the beta-actin as a loading control
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
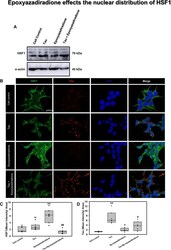
- Figure 7 Activation of HSF1 by epoxyazadiradione. ( A) Treatment of HEK293T cells with Tau and epoxyazadiradione exhibited no changes of HSF1 in western blotting analysis and its quantification. ( B) Extracellular Tau aggregates reduced the cellular HSF1 level and epoxyazadiradione increased HSF1 level in cytosol and nucleus. Tau aggregates were found to adhere in the cell periphery whereas the intracellular Tau level was negligible in HEK293T cells. ( C,D) IF quantification showed a significant increase in intracellular HSF1 and Tau aggregates were observed on the cellular periphery. Quantification was carried out by Zen 2.3 software ( https://www.zeiss.com/microscopy/int/products/light-microscopes/axio-observer-for-biology.html ).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
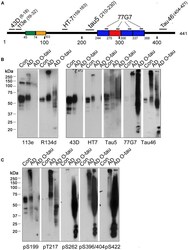
- Experimental details
- FIGURE 1 ADO-tau wasN-terminal truncated C-terminal hyperphosphorylated. (A) The diagram of the longest human tau isoform(tau 441 ) showing the epitopes recognized by various pan-tauantibodies used in this study. (B,C) AD O-tau andthe homogenates from AD and control human brains were analyzed byWestern blots developed with polyclonal or monoclonal pan-tauantibodies (B) and site-specific andphosphorylation-dependent tau antibodies (C) .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
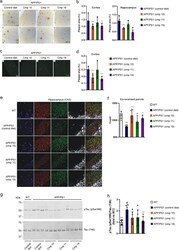
- Experimental details
- ATase inhibitors Compound 10 and Compound 19 reduce Alzheimer's disease pathology in the APP/PS1 mouse. a 6E10 immunohistochemistry in 5 um paraffin-embedded brain slices. b Quantification of ( a ). c Thioflavin-S staining of 5 um paraffin-embedded cortex brain slices. d Quantification of ( c ). e Immunostaining of 5 um paraffin-embedded brain slices for the presynaptic marker synaptophysin and postsynaptic marker Psd-95. NeuN (neuronal marker) is included for anatomic reference. f Quantification of ( e ). Red and green puncta of 1 um diameter were fit with spots and those within 1 um of one another were counted. g Representative Western blot showing levels of pTau (pSer396) in the brain. h Quantification of ( g ). Mice were 10-months old at their end-point. Bars represent mean +- SD. * p < 0.05, ** p < 0.005, # p < 0.0005 via ordinary two-way ANOVA with Dunnett's multiple comparison test (for 6E10 immunohistochemistry and Thioflavin-S staining) or with Tukey's multiple comparison test (for immunostaining of synaptic markers and Western blotting for Tau phosphorylation). In b , d , and f , each data point represents the average of technical replicates (a single brain slice) for a given animal. Black symbol, significance vs WT; Red symbol, significance vs untreated/control diet AT-1 sTg.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
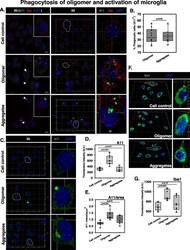
- Experimental details
- Fig. 3 Phagocytosis of Tau oligomers and activation of microglia. a N9 microglia were incubated with extracellular Tau oligomers for 24 h to initiate phagocytosis. The immunostaining study analyzed the presence of A11 + microglia indicating the phenomenon of active internalization of oligomer overburden. b The number of A11+ microglia was equal in amount during the phagocytosis of hTau40 WT oligomers and fibrillar aggregates. c 3D localization studies have shown that the microglia have engulfed an adequate amount of Tau oligomers and low molecular weight aggregates (A11 staining) as compared to the untreated control group. d , e The quantification of total A11 intensity in microglia upon different Tau species treatment group and the A11 intensity per square area have defined the density of A11 fluorescence in treated microglia by ZEN 2.3 image analytical microscopic software. f , g Microglial activation is mediated by the peri-membrane localization of Ca 2+ binding adaptor protein Iba1 in the case of preformed Tau aggregate treatment while the oligomer-treated group showed an increased level of cytosolic localization of Iba1 in microglia. The levels of Iba1 were quantified by ZEN 2.3 image analysis software in N9 microglia treated with Tau species. Significant at the mean difference between treatment groups (X - X`) > Tukey's criterion (T)
 Explore
Explore Validate
Validate Learn
Learn