44-734G
antibody from Invitrogen Antibodies
Targeting: MAPT
DDPAC, FLJ31424, FTDP-17, MAPTL, MGC138549, MSTD, MTBT1, MTBT2, PPND, PPP1R103, tau
Antibody data
- Antibody Data
- Antigen structure
- References [36]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [2]
- Immunohistochemistry [2]
- Flow cytometry [1]
- Other assay [21]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-734G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-Tau (Ser199) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
Submitted references Passive immunization against phosphorylated tau improves features of Huntington's disease pathology.
Medial septum tau accumulation induces spatial memory deficit via disrupting medial septum-hippocampus cholinergic pathway.
RPS23RG1 modulates tau phosphorylation and axon outgrowth through regulating p35 proteasomal degradation.
Chromatin accessibility mapping of the striatum identifies tyrosine kinase FYN as a therapeutic target for heroin use disorder.
Truncation of Tau selectively facilitates its pathological activities.
Pathological Tau From Alzheimer's Brain Induces Site-Specific Hyperphosphorylation and SDS- and Reducing Agent-Resistant Aggregation of Tau in vivo.
Subacute to chronic Alzheimer-like alterations after controlled cortical impact in human tau transgenic mice.
Involvement of Activation of Asparaginyl Endopeptidase in Tau Hyperphosphorylation in Repetitive Mild Traumatic Brain Injury.
Moringa Oleifera Alleviates Homocysteine-Induced Alzheimer's Disease-Like Pathology and Cognitive Impairments.
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Methanolic extract of Tamarix Gallica attenuates hyperhomocysteinemia induced AD-like pathology and cognitive impairments in rats.
Phosphorylation of different tau sites during progression of Alzheimer's disease.
Expression of Tau Pathology-Related Proteins in Different Brain Regions: A Molecular Basis of Tau Pathogenesis.
Role of mammalian target of rapamycin signaling in autophagy and the neurodegenerative process using a senescence accelerated mouse-prone 8 model.
The Deacetylase HDAC6 Mediates Endogenous Neuritic Tau Pathology.
Human Truncated Tau Induces Mature Neurofibrillary Pathology in a Mouse Model of Human Tauopathy.
Dendritic Spine Loss and Chronic White Matter Inflammation in a Mouse Model of Highly Repetitive Head Trauma.
Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment.
Tau phosphorylation regulates the interaction between BIN1's SH3 domain and Tau's proline-rich domain.
Rapid alteration of protein phosphorylation during postmortem: implication in the study of protein phosphorylation.
Neuronal uptake and propagation of a rare phosphorylated high-molecular-weight tau derived from Alzheimer's disease brain.
Cross talk between PI3K-AKT-GSK-3β and PP2A pathways determines tau hyperphosphorylation.
Amyloid and tau pathology of familial Alzheimer's disease APP/PS1 mouse model in a senescence phenotype background (SAMP8).
Specificity of anti-tau antibodies when analyzing mice models of Alzheimer's disease: problems and solutions.
Early alterations in energy metabolism in the hippocampus of APPswe/PS1dE9 mouse model of Alzheimer's disease.
Terminal hypothermic Tau.P301L mice have increased Tau phosphorylation independently of glycogen synthase kinase 3α/β.
Age-dependent effects of A53T alpha-synuclein on behavior and dopaminergic function.
Neurological characterization of mice deficient in GSK3α highlight pleiotropic physiological functions in cognition and pathological activity as Tau kinase.
An experimental rat model of sporadic Alzheimer's disease and rescue of cognitive impairment with a neurotrophic peptide.
Inhibition of JNK by a peptide inhibitor reduces traumatic brain injury-induced tauopathy in transgenic mice.
Asp664 cleavage of amyloid precursor protein induces tau phosphorylation by decreasing protein phosphatase 2A activity.
Methylthioninium chloride (methylene blue) induces autophagy and attenuates tauopathy in vitro and in vivo.
The carboxy-terminal fragment of inhibitor-2 of protein phosphatase-2A induces Alzheimer disease pathology and cognitive impairment.
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Co-localization of hyperphosphorylated tau and caspases in the brainstem of Alzheimer's disease patients.
Dephosphorylation of tau by protein phosphatase 5: impairment in Alzheimer's disease.
Alpaugh M, Masnata M, de Rus Jacquet A, Lepinay E, Denis HL, Saint-Pierre M, Davies P, Planel E, Cicchetti F
Molecular therapy : the journal of the American Society of Gene Therapy 2022 Apr 6;30(4):1500-1522
Molecular therapy : the journal of the American Society of Gene Therapy 2022 Apr 6;30(4):1500-1522
Medial septum tau accumulation induces spatial memory deficit via disrupting medial septum-hippocampus cholinergic pathway.
Wu D, Gao D, Yu H, Pi G, Xiong R, Lei H, Wang X, Liu E, Ye J, Yu H, Gao Y, He T, Jiang T, Sun F, Su J, Song G, Peng W, Yang Y, Wang JZ
Clinical and translational medicine 2021 Jun;11(6):e428
Clinical and translational medicine 2021 Jun;11(6):e428
RPS23RG1 modulates tau phosphorylation and axon outgrowth through regulating p35 proteasomal degradation.
Zhao D, Zhou Y, Huo Y, Meng J, Xiao X, Han L, Zhang X, Luo H, Can D, Sun H, Huang TY, Wang X, Zhang J, Liu FR, Xu H, Zhang YW
Cell death and differentiation 2021 Jan;28(1):337-348
Cell death and differentiation 2021 Jan;28(1):337-348
Chromatin accessibility mapping of the striatum identifies tyrosine kinase FYN as a therapeutic target for heroin use disorder.
Egervari G, Akpoyibo D, Rahman T, Fullard JF, Callens JE, Landry JA, Ly A, Zhou X, Warren N, Hauberg ME, Hoffman G, Ellis R, Ferland JN, Miller ML, Keller E, Zhang B, Roussos P, Hurd YL
Nature communications 2020 Sep 14;11(1):4634
Nature communications 2020 Sep 14;11(1):4634
Truncation of Tau selectively facilitates its pathological activities.
Gu J, Xu W, Jin N, Li L, Zhou Y, Chu D, Gong CX, Iqbal K, Liu F
The Journal of biological chemistry 2020 Oct 2;295(40):13812-13828
The Journal of biological chemistry 2020 Oct 2;295(40):13812-13828
Pathological Tau From Alzheimer's Brain Induces Site-Specific Hyperphosphorylation and SDS- and Reducing Agent-Resistant Aggregation of Tau in vivo.
Miao J, Shi R, Li L, Chen F, Zhou Y, Tung YC, Hu W, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2019;11:34
Frontiers in aging neuroscience 2019;11:34
Subacute to chronic Alzheimer-like alterations after controlled cortical impact in human tau transgenic mice.
Zhang Y, Wu F, Iqbal K, Gong CX, Hu W, Liu F
Scientific reports 2019 Mar 7;9(1):3789
Scientific reports 2019 Mar 7;9(1):3789
Involvement of Activation of Asparaginyl Endopeptidase in Tau Hyperphosphorylation in Repetitive Mild Traumatic Brain Injury.
Hu W, Tung YC, Zhang Y, Liu F, Iqbal K
Journal of Alzheimer's disease : JAD 2018;64(3):709-722
Journal of Alzheimer's disease : JAD 2018;64(3):709-722
Moringa Oleifera Alleviates Homocysteine-Induced Alzheimer's Disease-Like Pathology and Cognitive Impairments.
Mahaman YAR, Huang F, Wu M, Wang Y, Wei Z, Bao J, Salissou MTM, Ke D, Wang Q, Liu R, Wang JZ, Zhang B, Chen D, Wang X
Journal of Alzheimer's disease : JAD 2018;63(3):1141-1159
Journal of Alzheimer's disease : JAD 2018;63(3):1141-1159
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Zhou Y, Shi J, Chu D, Hu W, Guan Z, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2018;10:27
Frontiers in aging neuroscience 2018;10:27
Methanolic extract of Tamarix Gallica attenuates hyperhomocysteinemia induced AD-like pathology and cognitive impairments in rats.
Salissou MTM, Mahaman YAR, Zhu F, Huang F, Wang Y, Xu Z, Ke D, Wang Q, Liu R, Wang JZ, Zhang B, Wang X
Aging 2018 Nov 12;10(11):3229-3248
Aging 2018 Nov 12;10(11):3229-3248
Phosphorylation of different tau sites during progression of Alzheimer's disease.
Neddens J, Temmel M, Flunkert S, Kerschbaumer B, Hoeller C, Loeffler T, Niederkofler V, Daum G, Attems J, Hutter-Paier B
Acta neuropathologica communications 2018 Jun 29;6(1):52
Acta neuropathologica communications 2018 Jun 29;6(1):52
Expression of Tau Pathology-Related Proteins in Different Brain Regions: A Molecular Basis of Tau Pathogenesis.
Hu W, Wu F, Zhang Y, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2017;9:311
Frontiers in aging neuroscience 2017;9:311
Role of mammalian target of rapamycin signaling in autophagy and the neurodegenerative process using a senescence accelerated mouse-prone 8 model.
Wang Y, Ma Q, Ma X, Zhang Z, Liu N, Wang M
Experimental and therapeutic medicine 2017 Aug;14(2):1051-1057
Experimental and therapeutic medicine 2017 Aug;14(2):1051-1057
The Deacetylase HDAC6 Mediates Endogenous Neuritic Tau Pathology.
Tseng JH, Xie L, Song S, Xie Y, Allen L, Ajit D, Hong JS, Chen X, Meeker RB, Cohen TJ
Cell reports 2017 Aug 29;20(9):2169-2183
Cell reports 2017 Aug 29;20(9):2169-2183
Human Truncated Tau Induces Mature Neurofibrillary Pathology in a Mouse Model of Human Tauopathy.
Zimova I, Brezovakova V, Hromadka T, Weisova P, Cubinkova V, Valachova B, Filipcik P, Jadhav S, Smolek T, Novak M, Zilka N
Journal of Alzheimer's disease : JAD 2016 Sep 6;54(2):831-43
Journal of Alzheimer's disease : JAD 2016 Sep 6;54(2):831-43
Dendritic Spine Loss and Chronic White Matter Inflammation in a Mouse Model of Highly Repetitive Head Trauma.
Winston CN, Noël A, Neustadtl A, Parsadanian M, Barton DJ, Chellappa D, Wilkins TE, Alikhani AD, Zapple DN, Villapol S, Planel E, Burns MP
The American journal of pathology 2016 Mar;186(3):552-67
The American journal of pathology 2016 Mar;186(3):552-67
Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment.
Smolek T, Madari A, Farbakova J, Kandrac O, Jadhav S, Cente M, Brezovakova V, Novak M, Zilka N
The Journal of comparative neurology 2016 Mar 1;524(4):874-95
The Journal of comparative neurology 2016 Mar 1;524(4):874-95
Tau phosphorylation regulates the interaction between BIN1's SH3 domain and Tau's proline-rich domain.
Sottejeau Y, Bretteville A, Cantrelle FX, Malmanche N, Demiaute F, Mendes T, Delay C, Alves Dos Alves H, Flaig A, Davies P, Dourlen P, Dermaut B, Laporte J, Amouyel P, Lippens G, Chapuis J, Landrieu I, Lambert JC
Acta neuropathologica communications 2015 Sep 23;3:58
Acta neuropathologica communications 2015 Sep 23;3:58
Rapid alteration of protein phosphorylation during postmortem: implication in the study of protein phosphorylation.
Wang Y, Zhang Y, Hu W, Xie S, Gong CX, Iqbal K, Liu F
Scientific reports 2015 Oct 29;5:15709
Scientific reports 2015 Oct 29;5:15709
Neuronal uptake and propagation of a rare phosphorylated high-molecular-weight tau derived from Alzheimer's disease brain.
Takeda S, Wegmann S, Cho H, DeVos SL, Commins C, Roe AD, Nicholls SB, Carlson GA, Pitstick R, Nobuhara CK, Costantino I, Frosch MP, Müller DJ, Irimia D, Hyman BT
Nature communications 2015 Oct 13;6:8490
Nature communications 2015 Oct 13;6:8490
Cross talk between PI3K-AKT-GSK-3β and PP2A pathways determines tau hyperphosphorylation.
Wang Y, Yang R, Gu J, Yin X, Jin N, Xie S, Wang Y, Chang H, Qian W, Shi J, Iqbal K, Gong CX, Cheng C, Liu F
Neurobiology of aging 2015 Jan;36(1):188-200
Neurobiology of aging 2015 Jan;36(1):188-200
Amyloid and tau pathology of familial Alzheimer's disease APP/PS1 mouse model in a senescence phenotype background (SAMP8).
Porquet D, Andrés-Benito P, Griñán-Ferré C, Camins A, Ferrer I, Canudas AM, Del Valle J, Pallàs M
Age (Dordrecht, Netherlands) 2015 Feb;37(1):9747
Age (Dordrecht, Netherlands) 2015 Feb;37(1):9747
Specificity of anti-tau antibodies when analyzing mice models of Alzheimer's disease: problems and solutions.
Petry FR, Pelletier J, Bretteville A, Morin F, Calon F, Hébert SS, Whittington RA, Planel E
PloS one 2014;9(5):e94251
PloS one 2014;9(5):e94251
Early alterations in energy metabolism in the hippocampus of APPswe/PS1dE9 mouse model of Alzheimer's disease.
Pedrós I, Petrov D, Allgaier M, Sureda F, Barroso E, Beas-Zarate C, Auladell C, Pallàs M, Vázquez-Carrera M, Casadesús G, Folch J, Camins A
Biochimica et biophysica acta 2014 Sep;1842(9):1556-66
Biochimica et biophysica acta 2014 Sep;1842(9):1556-66
Terminal hypothermic Tau.P301L mice have increased Tau phosphorylation independently of glycogen synthase kinase 3α/β.
Maurin H, Lechat B, Borghgraef P, Devijver H, Jaworski T, Van Leuven F
The European journal of neuroscience 2014 Jul;40(2):2442-53
The European journal of neuroscience 2014 Jul;40(2):2442-53
Age-dependent effects of A53T alpha-synuclein on behavior and dopaminergic function.
Oaks AW, Frankfurt M, Finkelstein DI, Sidhu A
PloS one 2013;8(4):e60378
PloS one 2013;8(4):e60378
Neurological characterization of mice deficient in GSK3α highlight pleiotropic physiological functions in cognition and pathological activity as Tau kinase.
Maurin H, Lechat B, Dewachter I, Ris L, Louis JV, Borghgraef P, Devijver H, Jaworski T, Van Leuven F
Molecular brain 2013 May 25;6:27
Molecular brain 2013 May 25;6:27
An experimental rat model of sporadic Alzheimer's disease and rescue of cognitive impairment with a neurotrophic peptide.
Bolognin S, Blanchard J, Wang X, Basurto-Islas G, Tung YC, Kohlbrenner E, Grundke-Iqbal I, Iqbal K
Acta neuropathologica 2012 Jan;123(1):133-51
Acta neuropathologica 2012 Jan;123(1):133-51
Inhibition of JNK by a peptide inhibitor reduces traumatic brain injury-induced tauopathy in transgenic mice.
Tran HT, Sanchez L, Brody DL
Journal of neuropathology and experimental neurology 2012 Feb;71(2):116-29
Journal of neuropathology and experimental neurology 2012 Feb;71(2):116-29
Asp664 cleavage of amyloid precursor protein induces tau phosphorylation by decreasing protein phosphatase 2A activity.
Park SS, Jung HJ, Kim YJ, Park TK, Kim C, Choi H, Mook-Jung IH, Koo EH, Park SA
Journal of neurochemistry 2012 Dec;123(5):856-65
Journal of neurochemistry 2012 Dec;123(5):856-65
Methylthioninium chloride (methylene blue) induces autophagy and attenuates tauopathy in vitro and in vivo.
Congdon EE, Wu JW, Myeku N, Figueroa YH, Herman M, Marinec PS, Gestwicki JE, Dickey CA, Yu WH, Duff KE
Autophagy 2012 Apr;8(4):609-22
Autophagy 2012 Apr;8(4):609-22
The carboxy-terminal fragment of inhibitor-2 of protein phosphatase-2A induces Alzheimer disease pathology and cognitive impairment.
Wang X, Blanchard J, Kohlbrenner E, Clement N, Linden RM, Radu A, Grundke-Iqbal I, Iqbal K
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2010 Nov;24(11):4420-32
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2010 Nov;24(11):4420-32
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Yu Y, Run X, Liang Z, Li Y, Liu F, Liu Y, Iqbal K, Grundke-Iqbal I, Gong CX
Journal of neurochemistry 2009 Mar;108(6):1480-94
Journal of neurochemistry 2009 Mar;108(6):1480-94
Co-localization of hyperphosphorylated tau and caspases in the brainstem of Alzheimer's disease patients.
Wai MS, Liang Y, Shi C, Cho EY, Kung HF, Yew DT
Biogerontology 2009 Aug;10(4):457-69
Biogerontology 2009 Aug;10(4):457-69
Dephosphorylation of tau by protein phosphatase 5: impairment in Alzheimer's disease.
Liu F, Iqbal K, Grundke-Iqbal I, Rossie S, Gong CX
The Journal of biological chemistry 2005 Jan 21;280(3):1790-6
The Journal of biological chemistry 2005 Jan 21;280(3):1790-6
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Peptide Competition. Human recombinant Tau untreated (lane 1) or treated with GSK-3β (1 µg per µg Tau) for 45 minutes (lanes 2-5) was added to background extracts, resolved by SDS-PAGE on a 10% Tris-glycine gel and transferred to PVDF. The membrane was blocked with a 5% BSA-TBST buffer for one hour at room temperature, and then incubated with the Tau (pS199) antibody in a 3% BSA-TBST buffer for two hours at room temperature, following prior incubation with: no peptide (1, 2), the non-phosphopeptide corresponding to the phosphopeptide immunogen (3), a generic phosphoserine-containing peptide (4), or the phosphopeptide immunogen (5). After washing, the membrane was incubated with goat F (ab')2 anti-rabbit IgG HRP conjugate (Product # ALI4404) and signals were detected using the Pierce SuperSignal™ method. The data show that only the peptide corresponding to Tau (pS199) blocks the antibody signal, demonstrating the specificity of the antibody. The data also show the induction of Tau (pS199) phosphorylation by the addition of GSK-3β.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Peptide Competition. Human recombinant Tau untreated (lane 1) or treated with GSK-3β (1 µg per µg Tau) for 45 minutes (lanes 2-5) was added to background extracts, resolved by SDS-PAGE on a 10% Tris-glycine gel and transferred to PVDF. The membrane was blocked with a 5% BSA-TBST buffer for one hour at room temperature, and then incubated with the Tau (pS199) antibody in a 3% BSA-TBST buffer for two hours at room temperature, following prior incubation with: no peptide (1, 2), the non-phosphopeptide corresponding to the phosphopeptide immunogen (3), a generic phosphoserine-containing peptide (4), or the phosphopeptide immunogen (5). After washing, the membrane was incubated with goat F (ab')2 anti-rabbit IgG HRP conjugate (Product # ALI4404) and signals were detected using the Pierce SuperSignal™ method. The data show that only the peptide corresponding to Tau (pS199) blocks the antibody signal, demonstrating the specificity of the antibody. The data also show the induction of Tau (pS199) phosphorylation by the addition of GSK-3β.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Tau (pS199) and Tau (pS214) phosphospecific antibodies (Product # 44-742G); CV-1 cells transfected with full length adult human Tau and treated with okadaic acid to inhibit endogenous PP2A were incubated with Tau (pS199) (left) or Tau (pS214) (right). Data show that differential phosphorylation of Tau can influence the subcellular/cytoskeletal localization of Tau, including whether it remains bound to microtubules or is found soluble in the cytoplasm.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
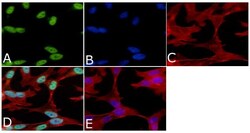
- Immunofluorescent analysis of Phospho-Tau pSer199 Antibody was done on 70% confluent log phase SHSY5Y cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton™ X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with Phospho-Tau pSer199 Antibody (Product # 44-734G) at 1µg/mL in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Goat Anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing nuclear localization. Panel e is a no primary antibody control. The images were captured at 40X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of Phospho-Tau (pS199) showing staining in the cytoplasm of paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Phospho-Tau (pS199) polyclonal antibody (Product # 44-734G) diluted in 3% BSA-PBS at a dilution of 1:100 overnight at 4ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemistry analysis of Phospho-Tau (pS199) showing staining in the cytoplasm of paraffin-embedded mouse brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Phospho-Tau (pS199) polyclonal antibody (Product # 44-734G) diluted in 3% BSA-PBS at a dilution of 1:100 overnight at 4ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Flow cytometry analysis of Tau [pS199] was done on SH-SY5Y cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with Tau [pS199] Rabbit Polyclonal Antibody (44734G, red histogram) or with rabbit isotype control (pink histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Goat Anti-Rabbit Secondary Antibody (A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
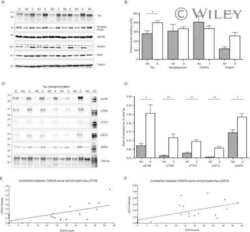
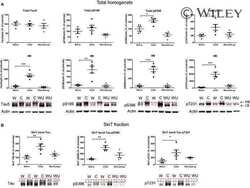
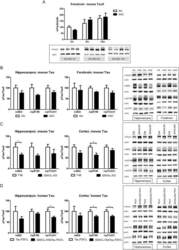
- Figure 10 Biochemical analysis of protein Tau in GSK3alpha deficient mice. A . Biochemical analysis by western blotting of total protein extracts from forebrain of AAC and AA- mice aged 3, 6 and 18 months (n=5/age). Tau protein levels are normalized to actin and expressed relative to control AA- mice at age 3 months. Data (mean+-SEM) are statistically analyzed by two-way Anova (Bonferroni post hoc test), genotype: F (1,24) =3.40, p=0.5093; age: F (2,24) =37.60, p=0.0027; interaction: F (2,24) =0.20, p=0.7759. Lower panels show representative western blots. B . Biochemical analysis by western blotting for phospho-epitopes pS396/404, pS199 and pT231 of endogenous mouse Tau in total protein extracts from hippocampus and forebrain of AAC and control AA- mice. C . Biochemical analysis by western blotting for phospho-epitopes pS396/404, pS199 and pT231 of endogenous mouse Tau in total protein extracts from hippocampus and cortex of GSK3alpha.KO and FVB wild-type mice. D . Biochemical analysis by western blotting for phospho-epitopes pS396/404, pS199 and pT231 of human Tau.P301L in total protein extracts from hippocampus and cortex of GSK3alpha.KOxTau.P301L mice and the parental Tau.P301L mice. In panels B-D , all data are normalized for total Tau and reported relative to the respective control mice. Data (mean+-SEM) are statistically analyzed by unpaired Student's t-test (two-tailed), n=6 or 7 per genotype; * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 Tau phosphorylation precludes the BIN1-Tau interaction in vitro . a . Tau-F5 fragment phosphorylation analysis after in vitro phosphorylation by CDK2/CycA3 kinase. Representative immunoblots using various antibodies against phosphorylation epitopes in Tau. The total amount of Tau-F5 was revealed by the phosphorylation-independent antibody Tau5. The significant shift in molecular weight observed in CDK2/CycA3-treated samples indicates Tau hyperphosphorylation. In contrast to other antibodies, the Tau-1 antibody binds to various non-phosphorylated Tau sites; the signal thus decreases when Tau is hyperphosphorylated [ 31 ]. b . 2D [ 1 H, 15 N] HSQC spectra of 125 muM 15 N CDK-phosphorylated Tau-F5[165-245] free in solution (gray) and with a 1 molar amount of GST-BIN1/SH3 (red, superimposed): Overlaid details of 2D [ 1 H, 15 N] HSQC spectra presented in Additional file 6 . c . 2D [ 1 H, 15 N] HSQC spectra of 60 muM 15 N CDK-phosphorylated 2N4R Tau free in solution (gray) and with a 2 molar amount of GST-BIN1/SH3 (blue, superimposed): Overlaid details of full spectra presented in Additional file 7
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
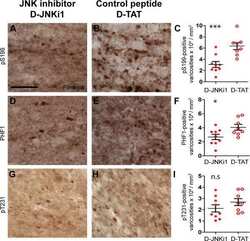
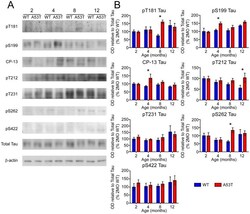
- Figure 6 Screen for additional pJNK-Tau sites in striatum. (A) Phosphorylation of Tau protein at epitopes subject to phosphorylation by JNK was analyzed by immunoblot. (B) Band optical density (OD) from phosphorylation-specific probes relative to total Tau expression is presented as percent of 2 month-old WT (mean +- SEM) and was analyzed by two-way ANOVA with Bonferroni post-hoc tests comparing each A53T group to age-matched controls (*p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
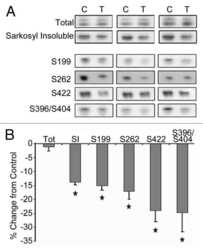
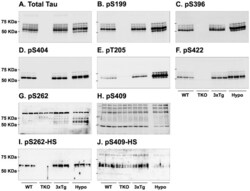
- Figure 7 Analysis of tau signal with polyclonal antibodies by Western blotting. Proteins were extracted from the cortex of 3 mouse lines: control mice (WT and Hypothermic), Tau KO mice and 3xTg-AD mice. Proteins were separated by SDS-PAGE and then identified with the following polyclonal antibodies: A: Total Tau, B: pS199, C: pS396, D: pS404, E: pT205, F: pS422, G: pS262 and H: pS409. Normal anti-rabbit secondary antibodies were used to detect primary antibodies. The heat stable fraction was used to remove non-specificity: I: pS262 and J: pS409. Quantifications of the blots are available in Figure S5 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
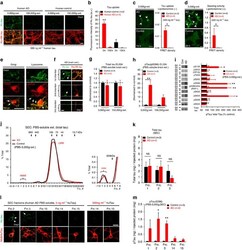
- Figure 6 Neuronal uptake of PBS-soluble HMW tau derived from human AD brain. ( a , b ) Primary neurons were incubated with AD or control brain extracts (cases were matched for age and postmortem interval ( Supplementary Table S1 )) and immunostained at day 2 ( a ). ( b ) Quantification of fluorescence intensity of human tau staining. One-way ANOVA and a subsequent Tukey-Kramer test. ( c , d ) Tau uptake ( c ) and seeding activity ( d ) assay in HEK-tau-biosensor cells. (Mann-Whitney U -test) ( e ) Subcellular localization of human tau taken up by neurons (PBS-3,000 g , 500 ng ml -1 human tau). ( f ) Neuron-to-neuron transfer of tau in a 3-chamber microfluidic device. AD brain extract (PBS-3,000 g , 500 ng ml -1 human tau) was added to the 1st chamber. Human tau positive neurons were detected in both the 1st and 2nd chamber at day 7 (arrow). ( g , h ) Quantification of total-tau ( g ) and phospho-tau ( h ) levels in AD and control brain extract (ELISA). Unpaired t -test. ( i ) Brain extracts were immunoblotted with phospho-tau specific antibodies recognizing different epitopes. Representative immunoblot and quantification of phospho-tau levels at each epitope. Unpaired t -test. ( j , k ) SEC analysis of PBS-soluble tau from AD and control brain. ( j ) Representative graph of total tau levels (ELISA) in SEC-separated samples. Small peaks for HMW fractions were detected in both groups (right panel). ( k ) Mean total tau levels of HMW SEC fractions. ( l ) Tau uptake from each SEC
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
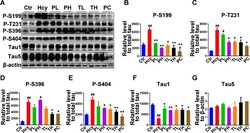
- Fig.3 Treatment with MO mitigated tau phosphorylation induced by HHcy. A) Levels of tau phosphorylation were measured by western blotting in the hippocampus after treatment (see method). B-E) Quantitative analysis of the blots of phosphorylated tau probed with several phosphorylated-tau antibodies, and (F) non-phosphorylated tau probed with Tau1 antibody, all normalized with total tau (Tau5) level. G) Total tau was normalized to beta-actin. The significant increase in tau phosphorylation at several studied sites was decreased by MO administration. The data were expressed as mean+-SD ( n = 6). # p < 0.05, # # p < 0.01 versus control; * p < 0.05, ** p < 0.01 versus Hcy.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
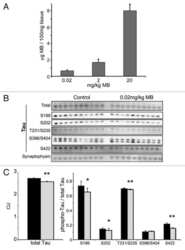
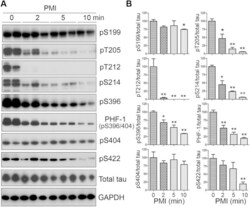
- Figure 1 Tau is rapidly dephosphorylated site-specifically in mouse brains during postmortem. Mice were scarified by cervical dislocation. The dead animal bodies were kept at room temperature for the indicated periods of time. ( A ) Phosphorylation of tau was analyzed by Western blots developed with phosphorylation-dependent and site-specific tau antibodies indicated at the right side of the blots. ( B ) The levels of tau phosphorylation at individual sites were quantified and normalized with total tau level and presented as mean +- SEM. (n = 3-4). * p < 0.05; ** p < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Both HMW-tau and LMW-tau are selectively hyperphosphorylated in AD brain. (A) AD and control human brain homogenates were analyzed by western blots developed with the indicated site-specific and phosphorylation dependent anti-tau antibodies. (B,C) Blots were analyzed by densitometry. The levels of hyperphosphorylated HMW-tau (B) and LMW-tau (C) are shown as scattered dots with mean +- SD. ** p < 0.01; *** p < 0.001; **** p < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Fig. 1 Tau phosphorylation increases at multiple sites in the brain following r-mTBI in 3xTg-AD mice. Adult female 3xTg-AD mice, 4-- months of age, were subjected to repetitive mild closed-head brain impacts, totaling five with 48-h inter-impact intervals, and sacrificed 24 h after the final impact. A) Representative western blots showing the level of indicated proteins/phosphorylation sites. B) Densitometric quantification of the blots. Among the phosphorylation sites examined, tau was hyperphosphorylated at pThr 205 , pSer 262 and PHF-1 (pSer 396/404 ) sites. Data are presented as scatter dot plots with mean+-SEM ( n = 6-7 mice each) and analyzed using unpaired t test, with Welch's correction in the case of unequal variance.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
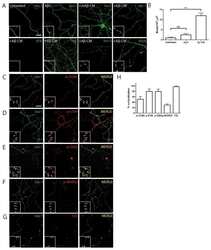
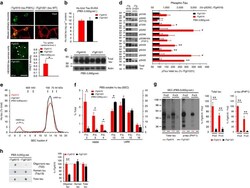
- Figure 2 Lack of PBS-soluble phosphorylated HMW tau species is associated with low tau uptake in primary neurons. ( a , top) Uptake of human tau from brain extracts from rTg4510 and rTg21221 mice by primary neurons (PBS-3,000 g , 500 ng ml -1 human tau). Neurons were immunostained with human tau-specific antibody (green) and total (human and mouse) tau antibody (red). ( a , bottom) Tau uptake assay in HEK-tau-biosensor cells. Brain extracts (10 mug protein) were applied to the cells (lipofectamie (-)). ( n =4) Unpaired t -test. Scale bar, 50 mum. ( b ) Human tau levels in brain extracts (ELISA). ( c ) Immunoblot analysis of PBS-soluble extracts with total tau antibody (DA9). Up-shifted bands in rTg4510 brain suggest phosphorylation of tau (arrow). ( d ) Brain extracts were immunoblotted with phospho-tau specific antibodies recognizing different epitopes. Representative immunoblot and quantification of phospho-tau levels at each epitope. ( n =3-4) Unpaired t -test. ( e , f ) SEC analysis of PBS-soluble tau. ( e ) Representative graph of human tau levels (ELISA) in SEC-separated samples ( f ) Mean human tau levels of HMW (Frc. 2-4) and LMW (Frc. 13-16) SEC fractions. ( n =3-6) Unpaired t -test. ( g ) Immunoblot analysis (SDS-PAGE) of SEC-separated fractions from brain extracts (total tau, DAKO). Quantification of band density is also shown (right graphs) ( n =4). Unpaired t -test. ( h ) Dot blot analysis of PBS-soluble brain extracts with tau oligomer-specific antibody (T22), h
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
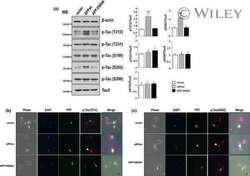
- Figure 2. Rapamycin treatment may promote cell morphology and alleviate the Tau phosphorylation of neurons from SAMP8. (A) Cell morphology (magnification, x200). The neurons derived from SAMR1 appear to be in a normal state (a, Neurons-SAMR1). The neurons extracted from SAMP8 were in poor state with most processes broken and exhibited 'bead-like' changes (indicated by the white arrows) (b, Neurons-SAMP8). When pretreated with 0.5 uM rapamycin, neurons from SAMP8 were partially improved with some smooth and slender processes (indicated by the white arrows; c, Neurons-SAMP8 + 0.5 uM rapamycin). When pretreated with 1.0 uM rapamycin, the cell state was worse than the untreated group and most neurons lacked projections (d, Neurons-SAMP8 + 1.0 uM rapamycin). (B) Western blot analysis was used to investigate Tau phosphorylation. In the neurons-SAMP8 group, the protein expression levels of Tau (pS199) and Tau (pS396) were significantly increased when compared with the control neurons-SAMR1 group. When pretreated with 0.5 uM rapamycin for three days, Tau (pS199) and Tau (pS396) exhibited significantly decreased levels of protein expression when compared with the neurons-SAMP8 group. *P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
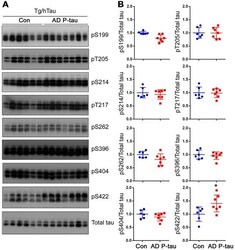
- Experimental details
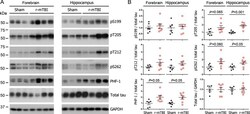
- Figure 3 Tau phosphorylation in the ipsilateral cortex in Tg/hTau mice injected with AD P-tau. Phosphorylation of tau at Ser199, Thr205, Ser214, Thr217, Ser262, Ser396, Ser404 and Ser422 in the ipsilateral cortex was analyzed by Western blots (A) and analyzed with unpaired student t -test. Data are presented as scattered dots with mean +- SD (B) ; * p < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Treatment with TG mitigated Hcy-induced tau hyperphosphorylation. ( A ) Phosphorylation status of tau protein as measured by Western blotting in the hippocampus after Hcy treatment and TG supplementation. ( B ) Total tau (Tau5) normalized to beta-actin. ( C-E ) Quantitative analysis of the blots of phosphorylated tau probed with several phosphorylated-tau antibodies normalized to total tau (Tau5). A significant decrease in tau hyperphosphorylation at several studied sites was seen following TG administration. The data were expressed as mean +- SEM (n = 6). ## P < 0.01, ### P
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot