44-738G
antibody from Invitrogen Antibodies
Targeting: MAPT
DDPAC, FLJ31424, FTDP-17, MAPTL, MGC138549, MSTD, MTBT1, MTBT2, PPND, PPP1R103, tau
Antibody data
- Antibody Data
- Antigen structure
- References [33]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [1]
- Flow cytometry [1]
- Other assay [16]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-738G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-Tau (Thr205) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- Purified from rabbit serum by sequential epitope-specific chromatography, this product contains enough material for 10 mini-blots. This antibody has been negatively preadsorbed using a non-phosphopeptide corresponding to the site of phosphorylation to remove antibody that is reactive with non-phosphorylated Tau. The final product is generated by affinity chromatography using a Tau-derived peptide that is phosphorylated at threonine 205. The antibody has been used in western blotting. Previous lots of this antibody have been used in immunohistochemistry. In western analysis the positive control used was recombinant human Tau treated with GSK3beta.
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
Submitted references Tau aggregates are RNA-protein assemblies that mislocalize multiple nuclear speckle components.
RPS23RG1 modulates tau phosphorylation and axon outgrowth through regulating p35 proteasomal degradation.
Truncation of Tau selectively facilitates its pathological activities.
β-amyloid and tau pathology in the aging feline brain.
Pathological Tau From Alzheimer's Brain Induces Site-Specific Hyperphosphorylation and SDS- and Reducing Agent-Resistant Aggregation of Tau in vivo.
GSKIP-Mediated Anchoring Increases Phosphorylation of Tau by PKA but Not by GSK3beta via cAMP/PKA/GSKIP/GSK3/Tau Axis Signaling in Cerebrospinal Fluid and iPS Cells in Alzheimer Disease.
Subacute to chronic Alzheimer-like alterations after controlled cortical impact in human tau transgenic mice.
Involvement of Activation of Asparaginyl Endopeptidase in Tau Hyperphosphorylation in Repetitive Mild Traumatic Brain Injury.
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Quercetin inhibits okadaic acid-induced tau protein hyperphosphorylation through the Ca2+‑calpain‑p25‑CDK5 pathway in HT22 cells.
Expression of Tau Pathology-Related Proteins in Different Brain Regions: A Molecular Basis of Tau Pathogenesis.
Tau secretion is correlated to an increase of Golgi dynamics.
Specific ion channels contribute to key elements of pathology during secondary degeneration following neurotrauma.
Human Truncated Tau Induces Mature Neurofibrillary Pathology in a Mouse Model of Human Tauopathy.
Dendritic Spine Loss and Chronic White Matter Inflammation in a Mouse Model of Highly Repetitive Head Trauma.
Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment.
Loss of O-GlcNAc glycosylation in forebrain excitatory neurons induces neurodegeneration.
Rapid alteration of protein phosphorylation during postmortem: implication in the study of protein phosphorylation.
Neuronal uptake and propagation of a rare phosphorylated high-molecular-weight tau derived from Alzheimer's disease brain.
Phosphorylation of a splice variant of collapsin response mediator protein 2 in the nucleus of tumour cells links cyclin dependent kinase-5 to oncogenesis.
Cross talk between PI3K-AKT-GSK-3β and PP2A pathways determines tau hyperphosphorylation.
Amyloid and tau pathology of familial Alzheimer's disease APP/PS1 mouse model in a senescence phenotype background (SAMP8).
Specificity of anti-tau antibodies when analyzing mice models of Alzheimer's disease: problems and solutions.
Early alterations in energy metabolism in the hippocampus of APPswe/PS1dE9 mouse model of Alzheimer's disease.
MiR-26b, upregulated in Alzheimer's disease, activates cell cycle entry, tau-phosphorylation, and apoptosis in postmitotic neurons.
An experimental rat model of sporadic Alzheimer's disease and rescue of cognitive impairment with a neurotrophic peptide.
The carboxy-terminal fragment of inhibitor-2 of protein phosphatase-2A induces Alzheimer disease pathology and cognitive impairment.
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Increased tau phosphorylation on mitogen-activated protein kinase consensus sites and cognitive decline in transgenic models for Alzheimer's disease and FTDP-17: evidence for distinct molecular processes underlying tau abnormalities.
Dephosphorylation of tau by protein phosphatase 5: impairment in Alzheimer's disease.
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Interaction of tau isoforms with Alzheimer's disease abnormally hyperphosphorylated tau and in vitro phosphorylation into the disease-like protein.
Lester E, Ooi FK, Bakkar N, Ayers J, Woerman AL, Wheeler J, Bowser R, Carlson GA, Prusiner SB, Parker R
Neuron 2021 May 19;109(10):1675-1691.e9
Neuron 2021 May 19;109(10):1675-1691.e9
RPS23RG1 modulates tau phosphorylation and axon outgrowth through regulating p35 proteasomal degradation.
Zhao D, Zhou Y, Huo Y, Meng J, Xiao X, Han L, Zhang X, Luo H, Can D, Sun H, Huang TY, Wang X, Zhang J, Liu FR, Xu H, Zhang YW
Cell death and differentiation 2021 Jan;28(1):337-348
Cell death and differentiation 2021 Jan;28(1):337-348
Truncation of Tau selectively facilitates its pathological activities.
Gu J, Xu W, Jin N, Li L, Zhou Y, Chu D, Gong CX, Iqbal K, Liu F
The Journal of biological chemistry 2020 Oct 2;295(40):13812-13828
The Journal of biological chemistry 2020 Oct 2;295(40):13812-13828
β-amyloid and tau pathology in the aging feline brain.
Fiock KL, Smith JD, Crary JF, Hefti MM
The Journal of comparative neurology 2020 Jan 1;528(1):108-113
The Journal of comparative neurology 2020 Jan 1;528(1):108-113
Pathological Tau From Alzheimer's Brain Induces Site-Specific Hyperphosphorylation and SDS- and Reducing Agent-Resistant Aggregation of Tau in vivo.
Miao J, Shi R, Li L, Chen F, Zhou Y, Tung YC, Hu W, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2019;11:34
Frontiers in aging neuroscience 2019;11:34
GSKIP-Mediated Anchoring Increases Phosphorylation of Tau by PKA but Not by GSK3beta via cAMP/PKA/GSKIP/GSK3/Tau Axis Signaling in Cerebrospinal Fluid and iPS Cells in Alzheimer Disease.
Ko HJ, Chiou SJ, Wong YH, Wang YH, Lai Y, Chou CH, Wang C, Loh JK, Lieu AS, Cheng JT, Lin YT, Lu PJ, Fann MJ, Huang CF, Hong YR
Journal of clinical medicine 2019 Oct 21;8(10)
Journal of clinical medicine 2019 Oct 21;8(10)
Subacute to chronic Alzheimer-like alterations after controlled cortical impact in human tau transgenic mice.
Zhang Y, Wu F, Iqbal K, Gong CX, Hu W, Liu F
Scientific reports 2019 Mar 7;9(1):3789
Scientific reports 2019 Mar 7;9(1):3789
Involvement of Activation of Asparaginyl Endopeptidase in Tau Hyperphosphorylation in Repetitive Mild Traumatic Brain Injury.
Hu W, Tung YC, Zhang Y, Liu F, Iqbal K
Journal of Alzheimer's disease : JAD 2018;64(3):709-722
Journal of Alzheimer's disease : JAD 2018;64(3):709-722
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Zhou Y, Shi J, Chu D, Hu W, Guan Z, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2018;10:27
Frontiers in aging neuroscience 2018;10:27
Quercetin inhibits okadaic acid-induced tau protein hyperphosphorylation through the Ca2+‑calpain‑p25‑CDK5 pathway in HT22 cells.
Shen XY, Luo T, Li S, Ting OY, He F, Xu J, Wang HQ
International journal of molecular medicine 2018 Feb;41(2):1138-1146
International journal of molecular medicine 2018 Feb;41(2):1138-1146
Expression of Tau Pathology-Related Proteins in Different Brain Regions: A Molecular Basis of Tau Pathogenesis.
Hu W, Wu F, Zhang Y, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2017;9:311
Frontiers in aging neuroscience 2017;9:311
Tau secretion is correlated to an increase of Golgi dynamics.
Mohamed NV, Desjardins A, Leclerc N
PloS one 2017;12(5):e0178288
PloS one 2017;12(5):e0178288
Specific ion channels contribute to key elements of pathology during secondary degeneration following neurotrauma.
O'Hare Doig RL, Chiha W, Giacci MK, Yates NJ, Bartlett CA, Smith NM, Hodgetts SI, Harvey AR, Fitzgerald M
BMC neuroscience 2017 Aug 14;18(1):62
BMC neuroscience 2017 Aug 14;18(1):62
Human Truncated Tau Induces Mature Neurofibrillary Pathology in a Mouse Model of Human Tauopathy.
Zimova I, Brezovakova V, Hromadka T, Weisova P, Cubinkova V, Valachova B, Filipcik P, Jadhav S, Smolek T, Novak M, Zilka N
Journal of Alzheimer's disease : JAD 2016 Sep 6;54(2):831-43
Journal of Alzheimer's disease : JAD 2016 Sep 6;54(2):831-43
Dendritic Spine Loss and Chronic White Matter Inflammation in a Mouse Model of Highly Repetitive Head Trauma.
Winston CN, Noël A, Neustadtl A, Parsadanian M, Barton DJ, Chellappa D, Wilkins TE, Alikhani AD, Zapple DN, Villapol S, Planel E, Burns MP
The American journal of pathology 2016 Mar;186(3):552-67
The American journal of pathology 2016 Mar;186(3):552-67
Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment.
Smolek T, Madari A, Farbakova J, Kandrac O, Jadhav S, Cente M, Brezovakova V, Novak M, Zilka N
The Journal of comparative neurology 2016 Mar 1;524(4):874-95
The Journal of comparative neurology 2016 Mar 1;524(4):874-95
Loss of O-GlcNAc glycosylation in forebrain excitatory neurons induces neurodegeneration.
Wang AC, Jensen EH, Rexach JE, Vinters HV, Hsieh-Wilson LC
Proceedings of the National Academy of Sciences of the United States of America 2016 Dec 27;113(52):15120-15125
Proceedings of the National Academy of Sciences of the United States of America 2016 Dec 27;113(52):15120-15125
Rapid alteration of protein phosphorylation during postmortem: implication in the study of protein phosphorylation.
Wang Y, Zhang Y, Hu W, Xie S, Gong CX, Iqbal K, Liu F
Scientific reports 2015 Oct 29;5:15709
Scientific reports 2015 Oct 29;5:15709
Neuronal uptake and propagation of a rare phosphorylated high-molecular-weight tau derived from Alzheimer's disease brain.
Takeda S, Wegmann S, Cho H, DeVos SL, Commins C, Roe AD, Nicholls SB, Carlson GA, Pitstick R, Nobuhara CK, Costantino I, Frosch MP, Müller DJ, Irimia D, Hyman BT
Nature communications 2015 Oct 13;6:8490
Nature communications 2015 Oct 13;6:8490
Phosphorylation of a splice variant of collapsin response mediator protein 2 in the nucleus of tumour cells links cyclin dependent kinase-5 to oncogenesis.
Grant NJ, Coates PJ, Woods YL, Bray SE, Morrice NA, Hastie CJ, Lamont DJ, Carey FA, Sutherland C
BMC cancer 2015 Nov 10;15:885
BMC cancer 2015 Nov 10;15:885
Cross talk between PI3K-AKT-GSK-3β and PP2A pathways determines tau hyperphosphorylation.
Wang Y, Yang R, Gu J, Yin X, Jin N, Xie S, Wang Y, Chang H, Qian W, Shi J, Iqbal K, Gong CX, Cheng C, Liu F
Neurobiology of aging 2015 Jan;36(1):188-200
Neurobiology of aging 2015 Jan;36(1):188-200
Amyloid and tau pathology of familial Alzheimer's disease APP/PS1 mouse model in a senescence phenotype background (SAMP8).
Porquet D, Andrés-Benito P, Griñán-Ferré C, Camins A, Ferrer I, Canudas AM, Del Valle J, Pallàs M
Age (Dordrecht, Netherlands) 2015 Feb;37(1):9747
Age (Dordrecht, Netherlands) 2015 Feb;37(1):9747
Specificity of anti-tau antibodies when analyzing mice models of Alzheimer's disease: problems and solutions.
Petry FR, Pelletier J, Bretteville A, Morin F, Calon F, Hébert SS, Whittington RA, Planel E
PloS one 2014;9(5):e94251
PloS one 2014;9(5):e94251
Early alterations in energy metabolism in the hippocampus of APPswe/PS1dE9 mouse model of Alzheimer's disease.
Pedrós I, Petrov D, Allgaier M, Sureda F, Barroso E, Beas-Zarate C, Auladell C, Pallàs M, Vázquez-Carrera M, Casadesús G, Folch J, Camins A
Biochimica et biophysica acta 2014 Sep;1842(9):1556-66
Biochimica et biophysica acta 2014 Sep;1842(9):1556-66
MiR-26b, upregulated in Alzheimer's disease, activates cell cycle entry, tau-phosphorylation, and apoptosis in postmitotic neurons.
Absalon S, Kochanek DM, Raghavan V, Krichevsky AM
The Journal of neuroscience : the official journal of the Society for Neuroscience 2013 Sep 11;33(37):14645-59
The Journal of neuroscience : the official journal of the Society for Neuroscience 2013 Sep 11;33(37):14645-59
An experimental rat model of sporadic Alzheimer's disease and rescue of cognitive impairment with a neurotrophic peptide.
Bolognin S, Blanchard J, Wang X, Basurto-Islas G, Tung YC, Kohlbrenner E, Grundke-Iqbal I, Iqbal K
Acta neuropathologica 2012 Jan;123(1):133-51
Acta neuropathologica 2012 Jan;123(1):133-51
The carboxy-terminal fragment of inhibitor-2 of protein phosphatase-2A induces Alzheimer disease pathology and cognitive impairment.
Wang X, Blanchard J, Kohlbrenner E, Clement N, Linden RM, Radu A, Grundke-Iqbal I, Iqbal K
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2010 Nov;24(11):4420-32
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2010 Nov;24(11):4420-32
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Yu Y, Run X, Liang Z, Li Y, Liu F, Liu Y, Iqbal K, Grundke-Iqbal I, Gong CX
Journal of neurochemistry 2009 Mar;108(6):1480-94
Journal of neurochemistry 2009 Mar;108(6):1480-94
Increased tau phosphorylation on mitogen-activated protein kinase consensus sites and cognitive decline in transgenic models for Alzheimer's disease and FTDP-17: evidence for distinct molecular processes underlying tau abnormalities.
Lambourne SL, Sellers LA, Bush TG, Choudhury SK, Emson PC, Suh YH, Wilkinson LS
Molecular and cellular biology 2005 Jan;25(1):278-93
Molecular and cellular biology 2005 Jan;25(1):278-93
Dephosphorylation of tau by protein phosphatase 5: impairment in Alzheimer's disease.
Liu F, Iqbal K, Grundke-Iqbal I, Rossie S, Gong CX
The Journal of biological chemistry 2005 Jan 21;280(3):1790-6
The Journal of biological chemistry 2005 Jan 21;280(3):1790-6
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Haase C, Stieler JT, Arendt T, Holzer M
Journal of neurochemistry 2004 Mar;88(6):1509-20
Journal of neurochemistry 2004 Mar;88(6):1509-20
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Haase C, Stieler JT, Arendt T, Holzer M
Journal of neurochemistry 2004 Mar;88(6):1509-20
Journal of neurochemistry 2004 Mar;88(6):1509-20
Interaction of tau isoforms with Alzheimer's disease abnormally hyperphosphorylated tau and in vitro phosphorylation into the disease-like protein.
Alonso AD, Zaidi T, Novak M, Barra HS, Grundke-Iqbal I, Iqbal K
The Journal of biological chemistry 2001 Oct 12;276(41):37967-73
The Journal of biological chemistry 2001 Oct 12;276(41):37967-73
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- TAU (pT205 Polyclonal Antibody using Western Blot (Product # 44-738G).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of Phospho-Tau pThr205 Antibody was done on 70% confluent log phase SHSY5Y cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton™ X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with Phospho-Tau pThr205 Antibody (Product # 44-738G) at 1µg/mL in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Goat Anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing nuclear and slight cytoplasmic localization. Panel e is a no primary antibody control. The images were captured at 40X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
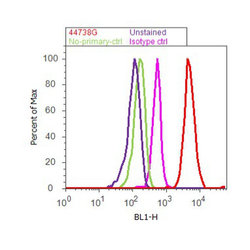
- Experimental details
- Flow cytometry analysis of Tau [pT205] was done on SH-SY5Y cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with Tau [pT205] Rabbit Polyclonal Antibody (44738G, red histogram) or with rabbit isotype control (pink histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Goat Anti-Rabbit Secondary Antibody (A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
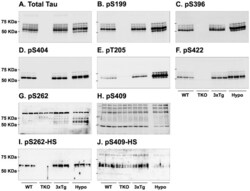
- Experimental details
- Figure 7 Analysis of tau signal with polyclonal antibodies by Western blotting. Proteins were extracted from the cortex of 3 mouse lines: control mice (WT and Hypothermic), Tau KO mice and 3xTg-AD mice. Proteins were separated by SDS-PAGE and then identified with the following polyclonal antibodies: A: Total Tau, B: pS199, C: pS396, D: pS404, E: pT205, F: pS422, G: pS262 and H: pS409. Normal anti-rabbit secondary antibodies were used to detect primary antibodies. The heat stable fraction was used to remove non-specificity: I: pS262 and J: pS409. Quantifications of the blots are available in Figure S5 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
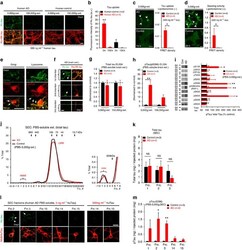
- Experimental details
- Figure 6 Neuronal uptake of PBS-soluble HMW tau derived from human AD brain. ( a , b ) Primary neurons were incubated with AD or control brain extracts (cases were matched for age and postmortem interval ( Supplementary Table S1 )) and immunostained at day 2 ( a ). ( b ) Quantification of fluorescence intensity of human tau staining. One-way ANOVA and a subsequent Tukey-Kramer test. ( c , d ) Tau uptake ( c ) and seeding activity ( d ) assay in HEK-tau-biosensor cells. (Mann-Whitney U -test) ( e ) Subcellular localization of human tau taken up by neurons (PBS-3,000 g , 500 ng ml -1 human tau). ( f ) Neuron-to-neuron transfer of tau in a 3-chamber microfluidic device. AD brain extract (PBS-3,000 g , 500 ng ml -1 human tau) was added to the 1st chamber. Human tau positive neurons were detected in both the 1st and 2nd chamber at day 7 (arrow). ( g , h ) Quantification of total-tau ( g ) and phospho-tau ( h ) levels in AD and control brain extract (ELISA). Unpaired t -test. ( i ) Brain extracts were immunoblotted with phospho-tau specific antibodies recognizing different epitopes. Representative immunoblot and quantification of phospho-tau levels at each epitope. Unpaired t -test. ( j , k ) SEC analysis of PBS-soluble tau from AD and control brain. ( j ) Representative graph of total tau levels (ELISA) in SEC-separated samples. Small peaks for HMW fractions were detected in both groups (right panel). ( k ) Mean total tau levels of HMW SEC fractions. ( l ) Tau uptake from each SEC
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
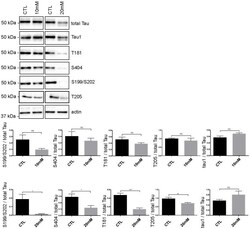
- Experimental details
- Fig 5 Tau phosphorylation is decreased in neurons treated with 20 mM KCl. Immunoblot analysis of Tau phosphorylation in neurons treated either with 10mM or 20mM KCl. The phosphorylation of S199/S202, T181, T205 and S404 was not significantly affected by 10mM of KCl treatment whereas the phosphorylation of all these sites was decreased by 20mM of KCl treatment. The signal of the phospho-antibodies was normalized to that of total Tau (n = 5, mean +- SEM, unpaired t-test two-tailed, * P < 0.05, *** P < 0.001). The amount of dephosphorylated Tau revealed by the Tau-1 antibody was normalized to that of total Tau lysate (n = 5, mean +- SEM, unpaired t-test two-tailed, * P < 0.05).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
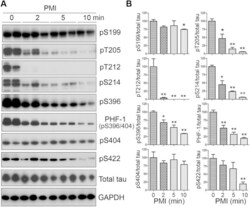
- Experimental details
- Figure 1 Tau is rapidly dephosphorylated site-specifically in mouse brains during postmortem. Mice were scarified by cervical dislocation. The dead animal bodies were kept at room temperature for the indicated periods of time. ( A ) Phosphorylation of tau was analyzed by Western blots developed with phosphorylation-dependent and site-specific tau antibodies indicated at the right side of the blots. ( B ) The levels of tau phosphorylation at individual sites were quantified and normalized with total tau level and presented as mean +- SEM. (n = 3-4). * p < 0.05; ** p < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Both HMW-tau and LMW-tau are selectively hyperphosphorylated in AD brain. (A) AD and control human brain homogenates were analyzed by western blots developed with the indicated site-specific and phosphorylation dependent anti-tau antibodies. (B,C) Blots were analyzed by densitometry. The levels of hyperphosphorylated HMW-tau (B) and LMW-tau (C) are shown as scattered dots with mean +- SD. ** p < 0.01; *** p < 0.001; **** p < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
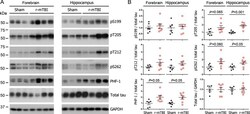
- Experimental details
- Fig. 1 Tau phosphorylation increases at multiple sites in the brain following r-mTBI in 3xTg-AD mice. Adult female 3xTg-AD mice, 4-- months of age, were subjected to repetitive mild closed-head brain impacts, totaling five with 48-h inter-impact intervals, and sacrificed 24 h after the final impact. A) Representative western blots showing the level of indicated proteins/phosphorylation sites. B) Densitometric quantification of the blots. Among the phosphorylation sites examined, tau was hyperphosphorylated at pThr 205 , pSer 262 and PHF-1 (pSer 396/404 ) sites. Data are presented as scatter dot plots with mean+-SEM ( n = 6-7 mice each) and analyzed using unpaired t test, with Welch's correction in the case of unequal variance.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
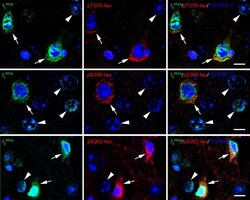
- Experimental details
- Fig. 5 Translocated I 2 PP2A /SET is colocalized with hyperphosphorylated tau in the neuronal cytoplasm in cerebral cortex of 3xTg-AD mice with r-mTBI. Brain sections were fluorescently dual immuno-stained for I 2 PP2A and phospho-tau. Translocated I 2 PP2A colocalized with pThr 205 -tau, pSer 396 -tau and with pSer 262 -tau in the cytoplasm of neurons (arrows). However, there was no somatodendritic phospho-tau staining in neurons where cytoplasmic I 2 PP2A was absent (filled arrowheads). Scale bar = 10 mu m for all images.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
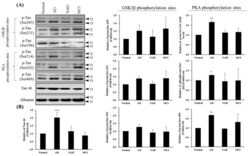
- Figure 4 Western blot of Tau46 (total Tau) and phosphorylated Tau by GSK3beta and PKA kinases from cerebrospinal fluid (CSF) sampling of four groups: normal, Alzheimer's disease (AD), neurological disorders (NAD), and mild cognitive impairment (MCI). ( A ) Western blotting with Tau 46, Tau Ser205, Tau Ser231, and Tau Ser396 by GSK3beta or Tau Ser214, Tau Ser262, and Tau Ser409 by PKA antibodies. Albumin was used as an internal control. ( B ) Statistical analysis. Bar graphs represent the mean +- SD of triplicates. * p < 0.05, ** p < 0.01, *** p < 0.001 compared with the control group.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3 Imaging of CDK5 substrate phosphorylaiton in primary cells. Rat primary cortical neurons were cultured for 6 days in vitro. a Cells were incubated with 10 muM purvalanol A or vehicle for 3 h prior to fixation, permeabolisation and staining with the indicated antibodies. Phosphospecific antibodies were detected by Cy-3 bound 2ry antibodies ( red ) and nuclei were counter-stained with the DNA-binding dye DAPI ( blue ). Scale bar = 60 mum. b The primary neuronal cultures were incubated with either 10 muM purvalanol A, roscovitine or vehicle for 3 h prior to fixation in formaldehyde and embedding in paraffin. Sections were taken from each paraffin block and incubated with the phospho-antibodies as indicated, then biotinylated secondary antibody followed by streptavidin complexed with biotinylated peroxidases which were visualized using DAB staining. Cell nuclei were counterstained with hematoxylin and mounted in DPX. Scale bar = 100 mum. Images are representative of sets from at least three different neuron preparations
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
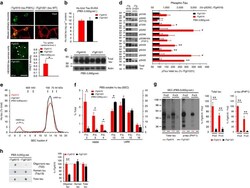
- Figure 2 Lack of PBS-soluble phosphorylated HMW tau species is associated with low tau uptake in primary neurons. ( a , top) Uptake of human tau from brain extracts from rTg4510 and rTg21221 mice by primary neurons (PBS-3,000 g , 500 ng ml -1 human tau). Neurons were immunostained with human tau-specific antibody (green) and total (human and mouse) tau antibody (red). ( a , bottom) Tau uptake assay in HEK-tau-biosensor cells. Brain extracts (10 mug protein) were applied to the cells (lipofectamie (-)). ( n =4) Unpaired t -test. Scale bar, 50 mum. ( b ) Human tau levels in brain extracts (ELISA). ( c ) Immunoblot analysis of PBS-soluble extracts with total tau antibody (DA9). Up-shifted bands in rTg4510 brain suggest phosphorylation of tau (arrow). ( d ) Brain extracts were immunoblotted with phospho-tau specific antibodies recognizing different epitopes. Representative immunoblot and quantification of phospho-tau levels at each epitope. ( n =3-4) Unpaired t -test. ( e , f ) SEC analysis of PBS-soluble tau. ( e ) Representative graph of human tau levels (ELISA) in SEC-separated samples ( f ) Mean human tau levels of HMW (Frc. 2-4) and LMW (Frc. 13-16) SEC fractions. ( n =3-6) Unpaired t -test. ( g ) Immunoblot analysis (SDS-PAGE) of SEC-separated fractions from brain extracts (total tau, DAKO). Quantification of band density is also shown (right graphs) ( n =4). Unpaired t -test. ( h ) Dot blot analysis of PBS-soluble brain extracts with tau oligomer-specific antibody (T22), h
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunohistochemistry
Immunohistochemistry