44-740G
antibody from Invitrogen Antibodies
Targeting: MAPT
DDPAC, FLJ31424, FTDP-17, MAPTL, MGC138549, MSTD, MTBT1, MTBT2, PPND, PPP1R103, tau
Antibody data
- Antibody Data
- Antigen structure
- References [26]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [1]
- Flow cytometry [1]
- Other assay [18]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-740G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-Tau (Thr212) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- Purified from rabbit serum by sequential epitope-specific chromatography, this product contains enough material for 10 mini-blots. The antibody has been negatively preadsorbed using a non-phosphopeptide corresponding to the site of phosphorylation to remove antibody that is reactive with non-phosphorylated Tau protein. The final product is generated by affinity chromatography using a Tau-derived peptide that is phosphorylated at threonine 212 in isoform Tau (441). For western analysis the positive control used was recombinant human Tau +/- GSK-3beta.
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
Submitted references Dephosphorylation Passivates the Seeding Activity of Oligomeric Tau Derived From Alzheimer's Brain.
Protein phosphatase PPM1B inhibits DYRK1A kinase through dephosphorylation of pS258 and reduces toxic tau aggregation.
Truncation of Tau selectively facilitates its pathological activities.
The green tea polyphenol epigallocatechin-3-gallate (EGCG) restores CDKL5-dependent synaptic defects in vitro and in vivo.
Proline Hydroxylation Primes Protein Kinases for Autophosphorylation and Activation.
Pathological Tau From Alzheimer's Brain Induces Site-Specific Hyperphosphorylation and SDS- and Reducing Agent-Resistant Aggregation of Tau in vivo.
Involvement of Activation of Asparaginyl Endopeptidase in Tau Hyperphosphorylation in Repetitive Mild Traumatic Brain Injury.
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Expression of Tau Pathology-Related Proteins in Different Brain Regions: A Molecular Basis of Tau Pathogenesis.
Human Truncated Tau Induces Mature Neurofibrillary Pathology in a Mouse Model of Human Tauopathy.
Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment.
A chemical with proven clinical safety rescues Down-syndrome-related phenotypes in through DYRK1A inhibition.
Selective inhibition of the kinase DYRK1A by targeting its folding process.
Systematic diversification of benzylidene heterocycles yields novel inhibitor scaffolds selective for Dyrk1A, Clk1 and CK2.
Selectivity Profiling and Biological Activity of Novel β-Carbolines as Potent and Selective DYRK1 Kinase Inhibitors.
Rapid alteration of protein phosphorylation during postmortem: implication in the study of protein phosphorylation.
10-iodo-11H-indolo[3,2-c]quinoline-6-carboxylic acids are selective inhibitors of DYRK1A.
Age-dependent effects of A53T alpha-synuclein on behavior and dopaminergic function.
Asp664 cleavage of amyloid precursor protein induces tau phosphorylation by decreasing protein phosphatase 2A activity.
The carboxy-terminal fragment of inhibitor-2 of protein phosphatase-2A induces Alzheimer disease pathology and cognitive impairment.
Development of a sensitive non-radioactive protein kinase assay and its application for detecting DYRK activity in Xenopus laevis oocytes.
Deletion of tau attenuates heat shock-induced injury in cultured cortical neurons.
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Interaction of tau isoforms with Alzheimer's disease abnormally hyperphosphorylated tau and in vitro phosphorylation into the disease-like protein.
Wu R, Li L, Shi R, Zhou Y, Jin N, Gu J, Tung YC, Liu F, Chu D
Frontiers in molecular neuroscience 2021;14:631833
Frontiers in molecular neuroscience 2021;14:631833
Protein phosphatase PPM1B inhibits DYRK1A kinase through dephosphorylation of pS258 and reduces toxic tau aggregation.
Lee YH, Im E, Hyun M, Park J, Chung KC
The Journal of biological chemistry 2021 Jan-Jun;296:100245
The Journal of biological chemistry 2021 Jan-Jun;296:100245
Truncation of Tau selectively facilitates its pathological activities.
Gu J, Xu W, Jin N, Li L, Zhou Y, Chu D, Gong CX, Iqbal K, Liu F
The Journal of biological chemistry 2020 Oct 2;295(40):13812-13828
The Journal of biological chemistry 2020 Oct 2;295(40):13812-13828
The green tea polyphenol epigallocatechin-3-gallate (EGCG) restores CDKL5-dependent synaptic defects in vitro and in vivo.
Trovò L, Fuchs C, De Rosa R, Barbiero I, Tramarin M, Ciani E, Rusconi L, Kilstrup-Nielsen C
Neurobiology of disease 2020 May;138:104791
Neurobiology of disease 2020 May;138:104791
Proline Hydroxylation Primes Protein Kinases for Autophosphorylation and Activation.
Lee SB, Ko A, Oh YT, Shi P, D'Angelo F, Frangaj B, Koller A, Chen EI, Cardozo T, Iavarone A, Lasorella A
Molecular cell 2020 Aug 6;79(3):376-389.e8
Molecular cell 2020 Aug 6;79(3):376-389.e8
Pathological Tau From Alzheimer's Brain Induces Site-Specific Hyperphosphorylation and SDS- and Reducing Agent-Resistant Aggregation of Tau in vivo.
Miao J, Shi R, Li L, Chen F, Zhou Y, Tung YC, Hu W, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2019;11:34
Frontiers in aging neuroscience 2019;11:34
Involvement of Activation of Asparaginyl Endopeptidase in Tau Hyperphosphorylation in Repetitive Mild Traumatic Brain Injury.
Hu W, Tung YC, Zhang Y, Liu F, Iqbal K
Journal of Alzheimer's disease : JAD 2018;64(3):709-722
Journal of Alzheimer's disease : JAD 2018;64(3):709-722
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Zhou Y, Shi J, Chu D, Hu W, Guan Z, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2018;10:27
Frontiers in aging neuroscience 2018;10:27
Expression of Tau Pathology-Related Proteins in Different Brain Regions: A Molecular Basis of Tau Pathogenesis.
Hu W, Wu F, Zhang Y, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2017;9:311
Frontiers in aging neuroscience 2017;9:311
Human Truncated Tau Induces Mature Neurofibrillary Pathology in a Mouse Model of Human Tauopathy.
Zimova I, Brezovakova V, Hromadka T, Weisova P, Cubinkova V, Valachova B, Filipcik P, Jadhav S, Smolek T, Novak M, Zilka N
Journal of Alzheimer's disease : JAD 2016 Sep 6;54(2):831-43
Journal of Alzheimer's disease : JAD 2016 Sep 6;54(2):831-43
Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment.
Smolek T, Madari A, Farbakova J, Kandrac O, Jadhav S, Cente M, Brezovakova V, Novak M, Zilka N
The Journal of comparative neurology 2016 Mar 1;524(4):874-95
The Journal of comparative neurology 2016 Mar 1;524(4):874-95
A chemical with proven clinical safety rescues Down-syndrome-related phenotypes in through DYRK1A inhibition.
Kim H, Lee KS, Kim AK, Choi M, Choi K, Kang M, Chi SW, Lee MS, Lee JS, Lee SY, Song WJ, Yu K, Cho S
Disease models & mechanisms 2016 Aug 1;9(8):839-48
Disease models & mechanisms 2016 Aug 1;9(8):839-48
Selective inhibition of the kinase DYRK1A by targeting its folding process.
Kii I, Sumida Y, Goto T, Sonamoto R, Okuno Y, Yoshida S, Kato-Sumida T, Koike Y, Abe M, Nonaka Y, Ikura T, Ito N, Shibuya H, Hosoya T, Hagiwara M
Nature communications 2016 Apr 22;7:11391
Nature communications 2016 Apr 22;7:11391
Systematic diversification of benzylidene heterocycles yields novel inhibitor scaffolds selective for Dyrk1A, Clk1 and CK2.
Mariano M, Hartmann RW, Engel M
European journal of medicinal chemistry 2016 Apr 13;112:209-216
European journal of medicinal chemistry 2016 Apr 13;112:209-216
Selectivity Profiling and Biological Activity of Novel β-Carbolines as Potent and Selective DYRK1 Kinase Inhibitors.
Rüben K, Wurzlbauer A, Walte A, Sippl W, Bracher F, Becker W
PloS one 2015;10(7):e0132453
PloS one 2015;10(7):e0132453
Rapid alteration of protein phosphorylation during postmortem: implication in the study of protein phosphorylation.
Wang Y, Zhang Y, Hu W, Xie S, Gong CX, Iqbal K, Liu F
Scientific reports 2015 Oct 29;5:15709
Scientific reports 2015 Oct 29;5:15709
10-iodo-11H-indolo[3,2-c]quinoline-6-carboxylic acids are selective inhibitors of DYRK1A.
Falke H, Chaikuad A, Becker A, Loaëc N, Lozach O, Abu Jhaisha S, Becker W, Jones PG, Preu L, Baumann K, Knapp S, Meijer L, Kunick C
Journal of medicinal chemistry 2015 Apr 9;58(7):3131-43
Journal of medicinal chemistry 2015 Apr 9;58(7):3131-43
Age-dependent effects of A53T alpha-synuclein on behavior and dopaminergic function.
Oaks AW, Frankfurt M, Finkelstein DI, Sidhu A
PloS one 2013;8(4):e60378
PloS one 2013;8(4):e60378
Asp664 cleavage of amyloid precursor protein induces tau phosphorylation by decreasing protein phosphatase 2A activity.
Park SS, Jung HJ, Kim YJ, Park TK, Kim C, Choi H, Mook-Jung IH, Koo EH, Park SA
Journal of neurochemistry 2012 Dec;123(5):856-65
Journal of neurochemistry 2012 Dec;123(5):856-65
The carboxy-terminal fragment of inhibitor-2 of protein phosphatase-2A induces Alzheimer disease pathology and cognitive impairment.
Wang X, Blanchard J, Kohlbrenner E, Clement N, Linden RM, Radu A, Grundke-Iqbal I, Iqbal K
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2010 Nov;24(11):4420-32
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2010 Nov;24(11):4420-32
Development of a sensitive non-radioactive protein kinase assay and its application for detecting DYRK activity in Xenopus laevis oocytes.
Lilienthal E, Kolanowski K, Becker W
BMC biochemistry 2010 May 20;11:20
BMC biochemistry 2010 May 20;11:20
Deletion of tau attenuates heat shock-induced injury in cultured cortical neurons.
Miao Y, Chen J, Zhang Q, Sun A
Journal of neuroscience research 2010 Jan;88(1):102-10
Journal of neuroscience research 2010 Jan;88(1):102-10
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Yu Y, Run X, Liang Z, Li Y, Liu F, Liu Y, Iqbal K, Grundke-Iqbal I, Gong CX
Journal of neurochemistry 2009 Mar;108(6):1480-94
Journal of neurochemistry 2009 Mar;108(6):1480-94
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Haase C, Stieler JT, Arendt T, Holzer M
Journal of neurochemistry 2004 Mar;88(6):1509-20
Journal of neurochemistry 2004 Mar;88(6):1509-20
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Haase C, Stieler JT, Arendt T, Holzer M
Journal of neurochemistry 2004 Mar;88(6):1509-20
Journal of neurochemistry 2004 Mar;88(6):1509-20
Interaction of tau isoforms with Alzheimer's disease abnormally hyperphosphorylated tau and in vitro phosphorylation into the disease-like protein.
Alonso AD, Zaidi T, Novak M, Barra HS, Grundke-Iqbal I, Iqbal K
The Journal of biological chemistry 2001 Oct 12;276(41):37967-73
The Journal of biological chemistry 2001 Oct 12;276(41):37967-73
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of Tau (pT212) was performed by loading 20 µg of Mouse Brain (lane1) and Rat Brain (lane2) tissue lysate using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0322BOX), XCell SureLock™ Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (LC5800), and iBlot® Dry Blotting System (IB21001). Proteins were transferred to a nitrocellulose membrane and blocked with 5 % skim milk for 1 hour at room temperature. Tau (pT212) was detected at ~ 42 kDa using Tau (pT212) Rabbit polyclonal Antibody (Product # 44-740G) at 1:1000 in 5 % skim milk at 4°C overnight on a rocking platform. Goat Anti-Rabbit IgG - HRP Secondary Antibody (G21234) at 1:5000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
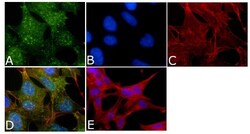
- Experimental details
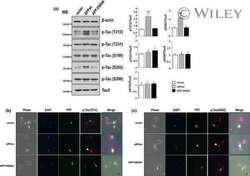
- Immunofluorescent analysis of Phospho-Tau pThr212 Antibody was done on 70% confluent log phase SHSY5Y cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton™ X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with Phospho-Tau pThr212 Antibody (Product # 44-740G) at 1µg/mL in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Goat Anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing nuclear and cytoplasmic localization. Panel e is a no primary antibody control. The images were captured at 40X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
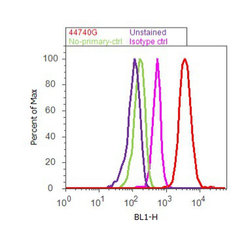
- Experimental details
- Flow cytometry analysis of Tau [pT212] was done on SH-SY5Y cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with Tau [pT212] Rabbit Polyclonal Antibody (44740G, red histogram) or with rabbit isotype control (pink histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Goat Anti-Rabbit Secondary Antibody (A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Direct sandwich ELISA format for the detection of the phosphorylated DYRK substrate peptide tau 207-219 . A , Scheme illustrating the principle of the assay. Bio, biotin; TMB, tetramethylbenzidine; HRP, horseradish peroxidase (coupled to streptavidin). B and C , Titration of phosphorylated and unphosphorylated tau 207-219 . The wells were coated with 100 ng anti tau(pT212) and loaded with dilution series of either phosphorylated or unphosphorylated tau 207-219 . The background signal from wells loaded only with the buffer was subtracted from all values. A representative experiment of three is shown. Panel C presents the same data as in panel B with a linear x-axis to visualize the linear range of the ELISA. The inset shows an enlargement of lower range. D , Titration of phosphorylated and non-phosphorylated tau 207-219 on streptavidin-coated wells. Detection was performed with primary anti-tau(pT212) antibody and secondary goat anti-rabbit antibody coupled to HRP. The graph is representative of two experiments. E , Detection of phosphorylated tau 207-219 in the presence of excess unphosphorylated peptide. Different amounts of phosphorylated tau 207-219 (0.01 pmol, 0.1 pmol, 1 pmol) were mixed with a dilution series of unphosphorylated tau 207-219 (12.5 - 100 pmol). Signals obtained in wells loaded only with the same amount of the unphosphorylated peptide were subtracted from the read-out of the mixtures. The graph is representative of two experiments. I
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
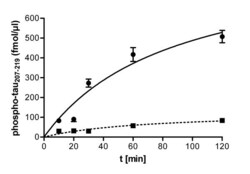
- Figure 4 Immunocomplex kinase assay of xDYRK1B from Xenopus laevis oocytes . Xenopus oocytes were lysed and the lysate was subjected to immunoprecipitation with either DYRK1B antiserum or preimmune serum ( alphaD1B, Pre ). Aliquots from immunocomplex kinase reactions (100 muM ATP, 50 muM tau 207-219 , 30degC, total volume of 50 muL) were taken at different times and the amounts of phosphorylated tau 207-219 were determined by the ELISA method with the help of a standard curve. Error bars represent the difference between the duplicate measurements.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
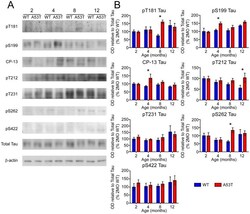
- Figure 6 Screen for additional pJNK-Tau sites in striatum. (A) Phosphorylation of Tau protein at epitopes subject to phosphorylation by JNK was analyzed by immunoblot. (B) Band optical density (OD) from phosphorylation-specific probes relative to total Tau expression is presented as percent of 2 month-old WT (mean +- SEM) and was analyzed by two-way ANOVA with Bonferroni post-hoc tests comparing each A53T group to age-matched controls (*p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
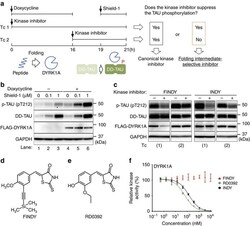
- Figure 1 Cell-based assay to evaluate a transitional intermediate-selective inhibitor of DYRK1A. ( a ) Schematic diagram of the SPHINKS assay. Doxycycline induces DYRK1A expression. Subsequently, Shield-1 stabilizes TAU fused with the destabilization domain of FKBP12 (DD-TAU). DYRK1A phosphorylates DD-TAU over the defined time period (19-21 h). Small molecules from our chemical library were added at the indicated points in Tc 1 and 2. Canonical kinase inhibitors suppress TAU phosphorylation in both Tc 1 and 2. Intermediate-selective inhibitors of DYRK1A should suppress TAU phosphorylation in Tc 1, but not in Tc 2. ( b ) Inducible expression of FLAG-DYRK1A and DD-TAU in HEK293 cells. Doxycycline induced FLAG-DYRK1A expression (lanes 4-6) and Shield-1 stabilized DD-TAU (lanes 2, 3, 5, 6). FLAG-DYRK1A predominantly phosphorylated Thr212 of TAU (p-TAU; lanes 5 and 6). p-TAU, TAU, FLAG and GAPDH were detected by western blot, using their corresponding antibodies. Representative data from the triplicate experiments are shown. ( c ) Identification of FINDY as the intermediate-selective inhibitor of DYRK1A. TAU phosphorylation was suppressed by FINDY (10 muM) in Tc 1, but not in Tc 2. In contrast, the canonical DYRK1A inhibitor INDY (10 muM) suppressed TAU phosphorylation in both Tc 1 and 2. Representative data from the triplicate experiments are shown. ( d ) Structure of FINDY. ( e ) Structure of RD0392, a canonical ATP-competitive inhibitor of DYRK1A. ( f ) FINDY did not inhibit th
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
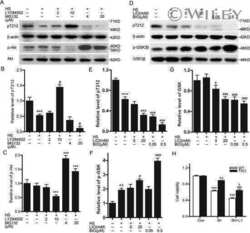
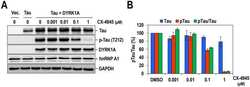
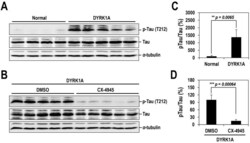
- Fig. 7. Oral administration of CX-4945 suppresses Tau phosphorylation in DYRK1A-overexpressing mice. (A) Hippocampuses from normal and DYRK1A-overexpressing C57BL/6 mice ( n =5 for each) were harvested, and the phosphorylation of Tau was analyzed by western blotting with anti-phosphorylated-Tau (at residue T212; p-Tau) and anti-Tau antibodies. alpha-Tubulin was also analyzed as a loading control. (B) The phosphorylated and total Tau proteins in panel A were quantified, and the mean amount of each protein was calculated. Relative ratios of phosphorylated Tau to total Tau (pTau/Tau) were presented by setting the value from normal mice as 100%. (C) DYRK1A-overexpressing C57BL/6 mice ( n =5 for each group) were administered orally with DMSO or 75 mg/kg of body weight of CX-4945 in PBS. Thirty minutes after oral administration, mice were killed, and the hippocampus was dissected from each mouse. Total cell lysates were prepared and subjected to western blotting with the indicated antibodies. alpha-Tubulin was also analyzed as a loading control. (D) The phosphorylated and total Tau proteins in panel C were quantified, and the mean amount of each protein across the animals was calculated. Relative ratios of phosphorylated Tau to total Tau were calculated by setting the value from DYRK1A mice that had been treated with DMSO as 100%. Data are means+-s.d. (two-tailed Student's t -test).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
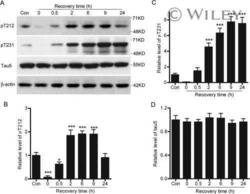
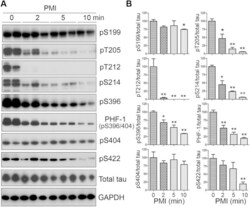
- Figure 1 Tau is rapidly dephosphorylated site-specifically in mouse brains during postmortem. Mice were scarified by cervical dislocation. The dead animal bodies were kept at room temperature for the indicated periods of time. ( A ) Phosphorylation of tau was analyzed by Western blots developed with phosphorylation-dependent and site-specific tau antibodies indicated at the right side of the blots. ( B ) The levels of tau phosphorylation at individual sites were quantified and normalized with total tau level and presented as mean +- SEM. (n = 3-4). * p < 0.05; ** p < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
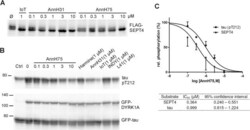
- Fig 5 Inhibition of SEPT4 and tau phosphorylation by DYRK1A. A , HeLa cells transiently expressing FLAG-SEPT4 were treated with AnnH31 or AnnH75 for 5 h before cells were lysed and analysed by immunoblotting with a FLAG-tag antibody. 5-iodotubercidin (IoT) served as positive control. Relative SEPT4 phosphorylation was calculated as the ratio of the intensities of the phosphorylated upper band and the lower band. B , HEK293 cells with constitutive expression of GFP-tau and regulatable expression of GFP-DYRK1A were treated with doxycyclin and the indicated inhibitors for 18 h. Phosphorylation of tau on Thr212 was detected with a phosphospecific antibody. Expression levels of GFP-tau and GFP-DYRK1A were assessed with a GFP antibody. For quantitative evaluation of DYRK1A inhibition, the basal pT212 signal in control cells not treated with doxycyclin (Ctrl) was subtracted from all values. C , Quantitative evaluation of three experiments each for SEPT4 and tau. All data were standardized to the level of phosphorylation in cells untreated with inhibitors. Error bars indicate SEM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
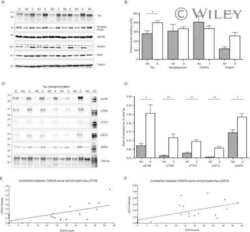
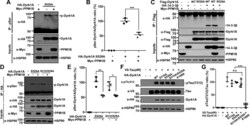
- Figure 3 PPM1B has no effect on phospho-DYRK1A at residues S310 or S529. A and B , HEK293 cells were transfected for 24 h with plasmids encoding HA-DYRK1A-S529A and/or Myc-PPM1B. Cell lysates were immunoprecipitated with anti-p-Ser antibody, followed by immunoblotting with anti-HA antibody. The proper expression of transiently transfected proteins in cell lysates was identified with immunoblot analysis using anti-HA or anti-Myc antibodies. Hsp90 served as a loading control. The ratio of pSer-DYRK1A to DYRK1A is shown in ( B ). All graph data represent the mean +- SD of three independent experiments (*** p < 0.001). C , HEK293 cells were transfected for 24 h with plasmids encoding FLAG-DYRK1A-WT, FLAG-DYRK1A-S529A, HA-14-3-3beta, or Myc-PPM1B alone or in combination. Cell lysates were immunoprecipitated with anti-FLAG antibody, followed by immunoblotting with anti-HA antibody. The proper expression of transiently transfected proteins in cell lysates was identified with immunoblot analysis using anti-Flag, anti-HA, or anti-Myc antibodies. Hsp90 served as a loading control. D , HEK293 cells were transfected for 24 h with plasmids encoding HA-DYRK1A-S529A, HA-DYRK1A-S310/529A, or Myc-PPM1B alone or in combination. Cell lysates were immunoprecipitated with anti-HA antibody, followed by immunoblotting with anti-phospho-serine antibody. The proper expression of transiently transfected proteins in cell lysates was identified with immunoblot analysis using anti-HA or anti-Myc antibodi
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
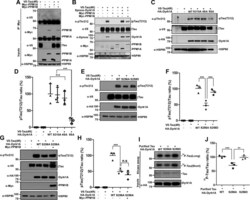
- Figure 5 PPM1B-mediated dephosphorylation of DYRK1A reduces tau phosphorylation at T212. A , HEK293 cells were transfected for 24 h with plasmid encoding V5-Tau, Myc-PPM1A, or Myc-PPM1B alone or in combination. Cell lysates were immunoprecipitated with anti-Myc antibody, followed by immunoblotting with anti-V5 antibody. The proper expression of transiently transfected proteins in cell lysates was identified with immunoblot analysis using anti-V5 or anti-Myc antibodies. Hsp90 served as a loading control. B , HEK293 cells were transfected for 24 h with plasmid encoding V5-Tau, Xpress-DYRK1A, Myc-PPM1A, or Myc-PPM1B alone or in combination. Cell lysates were immunoblotted with anti-pTau antibody (T212). The proper expression of transiently transfected proteins in cell lysates was identified with immunoblot analysis using anti-V5, anti-Xpress, or anti-Myc antibodies. C , HEK293 cells were transfected for 24 h with plasmid V5-Tau, HA-DYRK1A-WT, HA-DYRK1A-S310A, HA-DYRK1A-4SA, or HA-DYRK1A-5SA alone or in combination. Cell lysates were immunoblotted with anti-pTau antibody (T212). The proper expression of transiently transfected proteins in cell lysates was identified with immunoblot analysis using anti-V5 or anti-HA antibodies. D , All graph data represent the mean +- SD of three independent experiments (*** p < 0.001; n.s., not significant). E , HEK293 cells were transfected for 24 h with plasmid encoding V5-Tau, HA-DYRK1A-WT, HA-DYRK1A-S258A, or HA-DYRK1A-S258D alone or in combi
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
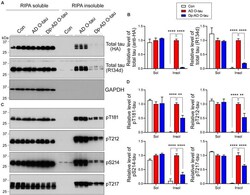
- FIGURE 5 Dephosphorylation of AD O-tau suppressed itsability to template tau aggregation. (A-D) HEK-293FT cellswere transfected with pCI/HA-tau 15 1-3 91 , treated with AD O-tau or Dp-AD O-tau and lysed withRIPA buffer. RIPA-soluble and-insoluble fractions were separated bycentrifugation and analyzed with Western blots developed withantibodies toward HA, total tau (R134d), GAPDH (A) , orvarious phosphorylated tau (C) . The experiment wasperformed in triplicate. Relative levels of total (B) orphosphorylated (D) tau were measured. ** P < 0.01. **** P < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Both HMW-tau and LMW-tau are selectively hyperphosphorylated in AD brain. (A) AD and control human brain homogenates were analyzed by western blots developed with the indicated site-specific and phosphorylation dependent anti-tau antibodies. (B,C) Blots were analyzed by densitometry. The levels of hyperphosphorylated HMW-tau (B) and LMW-tau (C) are shown as scattered dots with mean +- SD. ** p < 0.01; *** p < 0.001; **** p < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
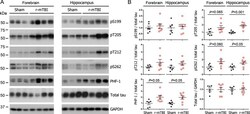
- Experimental details
- Fig. 1 Tau phosphorylation increases at multiple sites in the brain following r-mTBI in 3xTg-AD mice. Adult female 3xTg-AD mice, 4-- months of age, were subjected to repetitive mild closed-head brain impacts, totaling five with 48-h inter-impact intervals, and sacrificed 24 h after the final impact. A) Representative western blots showing the level of indicated proteins/phosphorylation sites. B) Densitometric quantification of the blots. Among the phosphorylation sites examined, tau was hyperphosphorylated at pThr 205 , pSer 262 and PHF-1 (pSer 396/404 ) sites. Data are presented as scatter dot plots with mean+-SEM ( n = 6-7 mice each) and analyzed using unpaired t test, with Welch's correction in the case of unequal variance.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
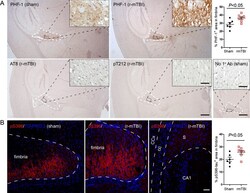
- Experimental details
- Fig. 2 Tau is hyperphosphorylated in fimbria of the hippocampus in 3xTg-AD mice after r-mTBI. A) Representative images showing immunohistochemical staining of sagittal brain sections with antibodies PHF-1, AT8 (pSer 202 /pThr 205 ) or anti-pThr 212 -tau. Fimbria showed easily visible PHF-1 immunoreactivity, and quantification revealed significantly more immunoreactivity in r-mTBI group than sham control group. No staining was detected by AT8 or anti-pThr 212 -tau. No primary antibody control for PHF-1 staining exhibited no staining. B) Immunofluorescence staining with anti-pSer 396 -tau. Immunoreactivity was mostly seen in fimbria, though subiculum also showed sparse staining. In addition, quantification which was performed by using images taken with a 20x objective lens showed more immunoreactivity in fimbria in r-mTBI group than in sham controls. Data are expressed as scattered dot plots with mean+-SEM ( n = 6-7 mice/group) and analyzed with unpaired Student t test. Scale bar = 500 mu m (A), 50 mu m (insert and B).
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot