44-742G
antibody from Invitrogen Antibodies
Targeting: MAPT
DDPAC, FLJ31424, FTDP-17, MAPTL, MGC138549, MSTD, MTBT1, MTBT2, PPND, PPP1R103, tau
Antibody data
- Antibody Data
- Antigen structure
- References [26]
- Comments [0]
- Validations
- Immunocytochemistry [2]
- Immunohistochemistry [2]
- Other assay [11]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-742G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-Tau (Ser214) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 μL
- Storage
- -20°C
Submitted references Enriched environment ameliorates propagation of tau pathology and improves cognition in rat model of tauopathy.
Genome-wide association study and functional validation implicates JADE1 in tauopathy.
Dephosphorylation Passivates the Seeding Activity of Oligomeric Tau Derived From Alzheimer's Brain.
TBK1 interacts with tau and enhances neurodegeneration in tauopathy.
Truncation of Tau selectively facilitates its pathological activities.
β-amyloid and tau pathology in the aging feline brain.
Pathological Tau From Alzheimer's Brain Induces Site-Specific Hyperphosphorylation and SDS- and Reducing Agent-Resistant Aggregation of Tau in vivo.
Tau-Mediated Disruption of the Spliceosome Triggers Cryptic RNA Splicing and Neurodegeneration in Alzheimer's Disease.
GSKIP-Mediated Anchoring Increases Phosphorylation of Tau by PKA but Not by GSK3beta via cAMP/PKA/GSKIP/GSK3/Tau Axis Signaling in Cerebrospinal Fluid and iPS Cells in Alzheimer Disease.
Subacute to chronic Alzheimer-like alterations after controlled cortical impact in human tau transgenic mice.
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Expression of Tau Pathology-Related Proteins in Different Brain Regions: A Molecular Basis of Tau Pathogenesis.
Liraglutide Improves Water Maze Learning and Memory Performance While Reduces Hyperphosphorylation of Tau and Neurofilaments in APP/PS1/Tau Triple Transgenic Mice.
Human Truncated Tau Induces Mature Neurofibrillary Pathology in a Mouse Model of Human Tauopathy.
Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment.
Tau-driven 26S proteasome impairment and cognitive dysfunction can be prevented early in disease by activating cAMP-PKA signaling.
Hypersialylation is a common feature of neurofibrillary tangles and granulovacuolar degenerations in Alzheimer's disease and tauopathy brains.
Rapid alteration of protein phosphorylation during postmortem: implication in the study of protein phosphorylation.
Age-dependent effects of A53T alpha-synuclein on behavior and dopaminergic function.
Pseudomonas aeruginosa exotoxin Y is a promiscuous cyclase that increases endothelial tau phosphorylation and permeability.
The carboxy-terminal fragment of inhibitor-2 of protein phosphatase-2A induces Alzheimer disease pathology and cognitive impairment.
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Tumor-suppressor PTEN affects tau phosphorylation, aggregation, and binding to microtubules.
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Interaction of tau isoforms with Alzheimer's disease abnormally hyperphosphorylated tau and in vitro phosphorylation into the disease-like protein.
Mate V, Smolek T, Kazmerova ZV, Jadhav S, Brezovakova V, Jurkanin B, Uhrinova I, Basheer N, Zilka N, Katina S, Novak P
Frontiers in aging neuroscience 2022;14:935973
Frontiers in aging neuroscience 2022;14:935973
Genome-wide association study and functional validation implicates JADE1 in tauopathy.
Farrell K, Kim S, Han N, Iida MA, Gonzalez EM, Otero-Garcia M, Walker JM, Richardson TE, Renton AE, Andrews SJ, Fulton-Howard B, Humphrey J, Vialle RA, Bowles KR, de Paiva Lopes K, Whitney K, Dangoor DK, Walsh H, Marcora E, Hefti MM, Casella A, Sissoko CT, Kapoor M, Novikova G, Udine E, Wong G, Tang W, Bhangale T, Hunkapiller J, Ayalon G, Graham RR, Cherry JD, Cortes EP, Borukov VY, McKee AC, Stein TD, Vonsattel JP, Teich AF, Gearing M, Glass J, Troncoso JC, Frosch MP, Hyman BT, Dickson DW, Murray ME, Attems J, Flanagan ME, Mao Q, Mesulam MM, Weintraub S, Woltjer RL, Pham T, Kofler J, Schneider JA, Yu L, Purohit DP, Haroutunian V, Hof PR, Gandy S, Sano M, Beach TG, Poon W, Kawas CH, Corrada MM, Rissman RA, Metcalf J, Shuldberg S, Salehi B, Nelson PT, Trojanowski JQ, Lee EB, Wolk DA, McMillan CT, Keene CD, Latimer CS, Montine TJ, Kovacs GG, Lutz MI, Fischer P, Perrin RJ, Cairns NJ, Franklin EE, Cohen HT, Raj T, Cobos I, Frost B, Goate A, White Iii CL, Crary JF
Acta neuropathologica 2022 Jan;143(1):33-53
Acta neuropathologica 2022 Jan;143(1):33-53
Dephosphorylation Passivates the Seeding Activity of Oligomeric Tau Derived From Alzheimer's Brain.
Wu R, Li L, Shi R, Zhou Y, Jin N, Gu J, Tung YC, Liu F, Chu D
Frontiers in molecular neuroscience 2021;14:631833
Frontiers in molecular neuroscience 2021;14:631833
TBK1 interacts with tau and enhances neurodegeneration in tauopathy.
Abreha MH, Ojelade S, Dammer EB, McEachin ZT, Duong DM, Gearing M, Bassell GJ, Lah JJ, Levey AI, Shulman JM, Seyfried NT
The Journal of biological chemistry 2021 Jan-Jun;296:100760
The Journal of biological chemistry 2021 Jan-Jun;296:100760
Truncation of Tau selectively facilitates its pathological activities.
Gu J, Xu W, Jin N, Li L, Zhou Y, Chu D, Gong CX, Iqbal K, Liu F
The Journal of biological chemistry 2020 Oct 2;295(40):13812-13828
The Journal of biological chemistry 2020 Oct 2;295(40):13812-13828
β-amyloid and tau pathology in the aging feline brain.
Fiock KL, Smith JD, Crary JF, Hefti MM
The Journal of comparative neurology 2020 Jan 1;528(1):108-113
The Journal of comparative neurology 2020 Jan 1;528(1):108-113
Pathological Tau From Alzheimer's Brain Induces Site-Specific Hyperphosphorylation and SDS- and Reducing Agent-Resistant Aggregation of Tau in vivo.
Miao J, Shi R, Li L, Chen F, Zhou Y, Tung YC, Hu W, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2019;11:34
Frontiers in aging neuroscience 2019;11:34
Tau-Mediated Disruption of the Spliceosome Triggers Cryptic RNA Splicing and Neurodegeneration in Alzheimer's Disease.
Hsieh YC, Guo C, Yalamanchili HK, Abreha M, Al-Ouran R, Li Y, Dammer EB, Lah JJ, Levey AI, Bennett DA, De Jager PL, Seyfried NT, Liu Z, Shulman JM
Cell reports 2019 Oct 8;29(2):301-316.e10
Cell reports 2019 Oct 8;29(2):301-316.e10
GSKIP-Mediated Anchoring Increases Phosphorylation of Tau by PKA but Not by GSK3beta via cAMP/PKA/GSKIP/GSK3/Tau Axis Signaling in Cerebrospinal Fluid and iPS Cells in Alzheimer Disease.
Ko HJ, Chiou SJ, Wong YH, Wang YH, Lai Y, Chou CH, Wang C, Loh JK, Lieu AS, Cheng JT, Lin YT, Lu PJ, Fann MJ, Huang CF, Hong YR
Journal of clinical medicine 2019 Oct 21;8(10)
Journal of clinical medicine 2019 Oct 21;8(10)
Subacute to chronic Alzheimer-like alterations after controlled cortical impact in human tau transgenic mice.
Zhang Y, Wu F, Iqbal K, Gong CX, Hu W, Liu F
Scientific reports 2019 Mar 7;9(1):3789
Scientific reports 2019 Mar 7;9(1):3789
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Zhou Y, Shi J, Chu D, Hu W, Guan Z, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2018;10:27
Frontiers in aging neuroscience 2018;10:27
Expression of Tau Pathology-Related Proteins in Different Brain Regions: A Molecular Basis of Tau Pathogenesis.
Hu W, Wu F, Zhang Y, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2017;9:311
Frontiers in aging neuroscience 2017;9:311
Liraglutide Improves Water Maze Learning and Memory Performance While Reduces Hyperphosphorylation of Tau and Neurofilaments in APP/PS1/Tau Triple Transgenic Mice.
Chen S, Sun J, Zhao G, Guo A, Chen Y, Fu R, Deng Y
Neurochemical research 2017 Aug;42(8):2326-2335
Neurochemical research 2017 Aug;42(8):2326-2335
Human Truncated Tau Induces Mature Neurofibrillary Pathology in a Mouse Model of Human Tauopathy.
Zimova I, Brezovakova V, Hromadka T, Weisova P, Cubinkova V, Valachova B, Filipcik P, Jadhav S, Smolek T, Novak M, Zilka N
Journal of Alzheimer's disease : JAD 2016 Sep 6;54(2):831-43
Journal of Alzheimer's disease : JAD 2016 Sep 6;54(2):831-43
Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment.
Smolek T, Madari A, Farbakova J, Kandrac O, Jadhav S, Cente M, Brezovakova V, Novak M, Zilka N
The Journal of comparative neurology 2016 Mar 1;524(4):874-95
The Journal of comparative neurology 2016 Mar 1;524(4):874-95
Tau-driven 26S proteasome impairment and cognitive dysfunction can be prevented early in disease by activating cAMP-PKA signaling.
Myeku N, Clelland CL, Emrani S, Kukushkin NV, Yu WH, Goldberg AL, Duff KE
Nature medicine 2016 Jan;22(1):46-53
Nature medicine 2016 Jan;22(1):46-53
Hypersialylation is a common feature of neurofibrillary tangles and granulovacuolar degenerations in Alzheimer's disease and tauopathy brains.
Nagamine S, Yamazaki T, Makioka K, Fujita Y, Ikeda M, Takatama M, Okamoto K, Yokoo H, Ikeda Y
Neuropathology : official journal of the Japanese Society of Neuropathology 2016 Aug;36(4):333-45
Neuropathology : official journal of the Japanese Society of Neuropathology 2016 Aug;36(4):333-45
Rapid alteration of protein phosphorylation during postmortem: implication in the study of protein phosphorylation.
Wang Y, Zhang Y, Hu W, Xie S, Gong CX, Iqbal K, Liu F
Scientific reports 2015 Oct 29;5:15709
Scientific reports 2015 Oct 29;5:15709
Age-dependent effects of A53T alpha-synuclein on behavior and dopaminergic function.
Oaks AW, Frankfurt M, Finkelstein DI, Sidhu A
PloS one 2013;8(4):e60378
PloS one 2013;8(4):e60378
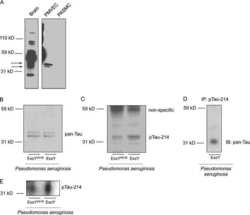
Pseudomonas aeruginosa exotoxin Y is a promiscuous cyclase that increases endothelial tau phosphorylation and permeability.
Ochoa CD, Alexeyev M, Pastukh V, Balczon R, Stevens T
The Journal of biological chemistry 2012 Jul 20;287(30):25407-18
The Journal of biological chemistry 2012 Jul 20;287(30):25407-18
The carboxy-terminal fragment of inhibitor-2 of protein phosphatase-2A induces Alzheimer disease pathology and cognitive impairment.
Wang X, Blanchard J, Kohlbrenner E, Clement N, Linden RM, Radu A, Grundke-Iqbal I, Iqbal K
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2010 Nov;24(11):4420-32
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2010 Nov;24(11):4420-32
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Yu Y, Run X, Liang Z, Li Y, Liu F, Liu Y, Iqbal K, Grundke-Iqbal I, Gong CX
Journal of neurochemistry 2009 Mar;108(6):1480-94
Journal of neurochemistry 2009 Mar;108(6):1480-94
Tumor-suppressor PTEN affects tau phosphorylation, aggregation, and binding to microtubules.
Zhang X, Li F, Bulloj A, Zhang YW, Tong G, Zhang Z, Liao FF, Xu H
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2006 Jun;20(8):1272-4
FASEB journal : official publication of the Federation of American Societies for Experimental Biology 2006 Jun;20(8):1272-4
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Haase C, Stieler JT, Arendt T, Holzer M
Journal of neurochemistry 2004 Mar;88(6):1509-20
Journal of neurochemistry 2004 Mar;88(6):1509-20
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Haase C, Stieler JT, Arendt T, Holzer M
Journal of neurochemistry 2004 Mar;88(6):1509-20
Journal of neurochemistry 2004 Mar;88(6):1509-20
Interaction of tau isoforms with Alzheimer's disease abnormally hyperphosphorylated tau and in vitro phosphorylation into the disease-like protein.
Alonso AD, Zaidi T, Novak M, Barra HS, Grundke-Iqbal I, Iqbal K
The Journal of biological chemistry 2001 Oct 12;276(41):37967-73
The Journal of biological chemistry 2001 Oct 12;276(41):37967-73
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
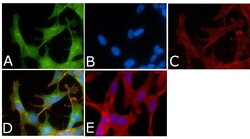
- Experimental details
- Immunofluorescent analysis of Phospho-Tau pSer214 Antibody was done on 70% confluent log phase SHSY5Y cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton™ X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with Phospho-Tau pSer214 Antibody (Product # 44-742G) at 1µg/mL in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Goat Anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing nuclear and cytoplasmic localization. Panel e is a no primary antibody control. The images were captured at 40X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
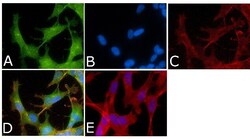
- Experimental details
- Immunofluorescent analysis of Phospho-Tau pSer214 Antibody was done on 70% confluent log phase SHSY5Y cells. The cells were fixed with 4% paraformaldehyde for 15 minutes, permeabilized with 0.25% Triton™ X-100 for 10 minutes, and blocked with 5% BSA for 1 hour at room temperature. The cells were labeled with Phospho-Tau pSer214 Antibody (Product # 44-742G) at 1µg/mL in 1% BSA and incubated for 3 hours at room temperature and then labeled with Alexa Fluor 488 Goat Anti-Rabbit IgG Secondary Antibody (Product # A-11008) at a dilution of 1:400 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor 594 Phalloidin (Product # A12381). Panel d is a merged image showing nuclear and cytoplasmic localization. Panel e is a no primary antibody control. The images were captured at 40X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
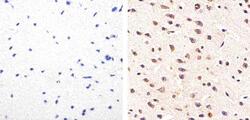
- Experimental details
- Immunohistochemistry analysis of Phospho-Tau (pS214) showing staining in the cytoplasm of paraffin-embedded mouse brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Phospho-Tau (pS214) polyclonal antibody (Product # 44-742G) diluted in 3% BSA-PBS at a dilution of 1:100 overnight at 4ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
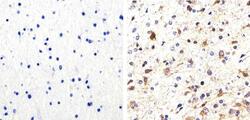
- Experimental details
- Immunohistochemistry analysis of Phospho-Tau (pS214) showing staining in the cytoplasm of paraffin-embedded human astroglioma tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Phospho-Tau (pS214) polyclonal antibody (Product # 44-742G) diluted in 3% BSA-PBS at a dilution of 1:100 overnight at 4ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
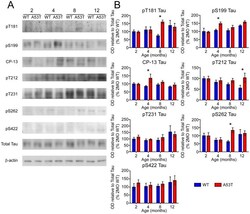
- Figure 6 Screen for additional pJNK-Tau sites in striatum. (A) Phosphorylation of Tau protein at epitopes subject to phosphorylation by JNK was analyzed by immunoblot. (B) Band optical density (OD) from phosphorylation-specific probes relative to total Tau expression is presented as percent of 2 month-old WT (mean +- SEM) and was analyzed by two-way ANOVA with Bonferroni post-hoc tests comparing each A53T group to age-matched controls (*p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
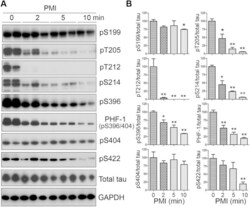
- Figure 1 Tau is rapidly dephosphorylated site-specifically in mouse brains during postmortem. Mice were scarified by cervical dislocation. The dead animal bodies were kept at room temperature for the indicated periods of time. ( A ) Phosphorylation of tau was analyzed by Western blots developed with phosphorylation-dependent and site-specific tau antibodies indicated at the right side of the blots. ( B ) The levels of tau phosphorylation at individual sites were quantified and normalized with total tau level and presented as mean +- SEM. (n = 3-4). * p < 0.05; ** p < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
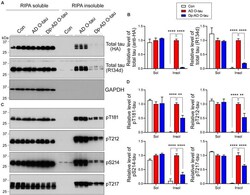
- FIGURE 5 Dephosphorylation of AD O-tau suppressed itsability to template tau aggregation. (A-D) HEK-293FT cellswere transfected with pCI/HA-tau 15 1-3 91 , treated with AD O-tau or Dp-AD O-tau and lysed withRIPA buffer. RIPA-soluble and-insoluble fractions were separated bycentrifugation and analyzed with Western blots developed withantibodies toward HA, total tau (R134d), GAPDH (A) , orvarious phosphorylated tau (C) . The experiment wasperformed in triplicate. Relative levels of total (B) orphosphorylated (D) tau were measured. ** P < 0.01. **** P < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
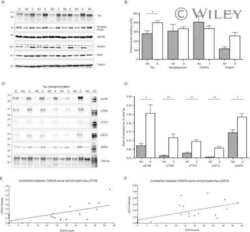
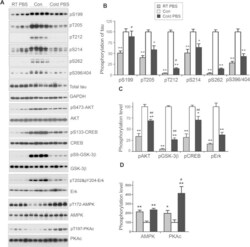
- Figure 5 Perfusion causes dramatic changes of protein phosphorylation in the mouse brain. Mice were transcardially perfused with RT-PBS or cold PBS after sacrificed by cervical dislocation. The forebrains were collected and used for protein phosphorylation analysis by Western blots ( A ). The phosphorylation level was quantified after being normalized with the corresponding protein and presented as mean +- SEM (n = 4) ( B ). * ,# p < 0.05; ** ,## p < 0.01; *perfusion vs control; # cold PBS vs RT-PBS.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
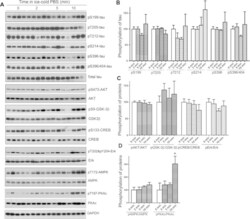
- Figure 6 Keeping tissues in ice-cold buffer prevents the alteration of protein phosphorylation in mouse brain. Mice were sacrificed by dislocation. The fore brains were transferred immediately into ice-cold PBS (0 ) and kept in the ice-cold PBS for various time points. The protein phosphorylation was analyzed by Western blots ( A ). The phosphorylation levels of proteins were normalized by corresponding proteins and are presented as mean +- SEM (n = 4-5) ( B) . * p < 0.05.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Both HMW-tau and LMW-tau are selectively hyperphosphorylated in AD brain. (A) AD and control human brain homogenates were analyzed by western blots developed with the indicated site-specific and phosphorylation dependent anti-tau antibodies. (B,C) Blots were analyzed by densitometry. The levels of hyperphosphorylated HMW-tau (B) and LMW-tau (C) are shown as scattered dots with mean +- SD. ** p < 0.01; *** p < 0.001; **** p < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
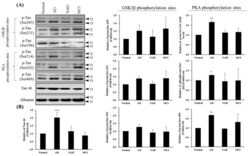
- Figure 4 Western blot of Tau46 (total Tau) and phosphorylated Tau by GSK3beta and PKA kinases from cerebrospinal fluid (CSF) sampling of four groups: normal, Alzheimer's disease (AD), neurological disorders (NAD), and mild cognitive impairment (MCI). ( A ) Western blotting with Tau 46, Tau Ser205, Tau Ser231, and Tau Ser396 by GSK3beta or Tau Ser214, Tau Ser262, and Tau Ser409 by PKA antibodies. Albumin was used as an internal control. ( B ) Statistical analysis. Bar graphs represent the mean +- SD of triplicates. * p < 0.05, ** p < 0.01, *** p < 0.001 compared with the control group.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
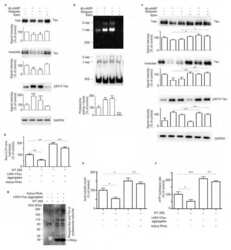
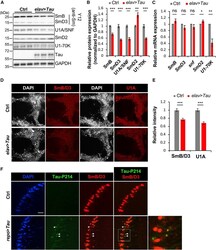
- Figure 2. Disruption of the Spliceosome following Tau Expression in Drosophila (A and B) Pan-neuronal human Tau R406W disrupts expression of multiple spliceosomal proteins in 1-day-old adult fly head homogenates. Western blots (A) were probed for Tau or spliceosome proteins and normalized to the loading control, GAPDH (n = 4 replicates for quantification, B). The Y12 antibody recognizes both SmB and SmD3. (C) mRNA expression was also examined in 1-day-old adult heads (n = 3). (D and E) Whole-mount stains of 1-day-old adult fly brains (D) reveal depletion of SmB/D3 and U1A/SNF protein (red, n = 15 for quantification, E). Nuclei are colabeled with 4',6-diamidino-2-phenylindole (DAPI; grayscale). Scale bar: 20 mum. (F) Glial expression of Tau WT ( repo-GAL4 ) induces cytoplasmic foci (arrowheads) of SmB/D3 (Y12, red) that colocalize with phospho-Tau aggregates (green) in 10-day-old adult brains. Nuclei are labeled with DAPI (blue). Boxed region is magnified at right. Quantification (n > 9) reveals 14.86% +- 2.3% of phospho-Tau aggregates colabeling for Sm proteins; aggregates were not observed in controls. Scale bar: 10 mum. See also Figure S3A . All error bars denote mean +- SEM. *p < 0.05; **p < 0.01; ***p < 0.001; ns, not significant.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry