44-744
antibody from Invitrogen Antibodies
Targeting: MAPT
DDPAC, FLJ31424, FTDP-17, MAPTL, MGC138549, MSTD, MTBT1, MTBT2, PPND, PPP1R103, tau
Antibody data
- Antibody Data
- Antigen structure
- References [19]
- Comments [0]
- Validations
- Western blot [1]
- Immunohistochemistry [2]
- Flow cytometry [1]
- Other assay [5]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-744 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-Tau (Thr217) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
Submitted references Amelioration of Alzheimer's disease pathology by mitophagy inducers identified via machine learning and a cross-species workflow.
Cerebrospinal fluid p-tau231 as an early indicator of emerging pathology in Alzheimer's disease.
Systematic Phenotyping and Characterization of the 3xTg-AD Mouse Model of Alzheimer's Disease.
Dephosphorylation Passivates the Seeding Activity of Oligomeric Tau Derived From Alzheimer's Brain.
Head-to-head comparison of clinical performance of CSF phospho-tau T181 and T217 biomarkers for Alzheimer's disease diagnosis.
Icariin protects against sodium azide-induced neurotoxicity by activating the PI3K/Akt/GSK-3β signaling pathway.
Truncation of Tau selectively facilitates its pathological activities.
Novel tau biomarkers phosphorylated at T181, T217 or T231 rise in the initial stages of the preclinical Alzheimer's continuum when only subtle changes in Aβ pathology are detected.
Pathological Tau From Alzheimer's Brain Induces Site-Specific Hyperphosphorylation and SDS- and Reducing Agent-Resistant Aggregation of Tau in vivo.
A post-translational modification signature defines changes in soluble tau correlating with oligomerization in early stage Alzheimer's disease brain.
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Expression of Tau Pathology-Related Proteins in Different Brain Regions: A Molecular Basis of Tau Pathogenesis.
Liraglutide Improves Water Maze Learning and Memory Performance While Reduces Hyperphosphorylation of Tau and Neurofilaments in APP/PS1/Tau Triple Transgenic Mice.
Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment.
Age-dependent effects of A53T alpha-synuclein on behavior and dopaminergic function.
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Interaction of tau isoforms with Alzheimer's disease abnormally hyperphosphorylated tau and in vitro phosphorylation into the disease-like protein.
Xie C, Zhuang XX, Niu Z, Ai R, Lautrup S, Zheng S, Jiang Y, Han R, Gupta TS, Cao S, Lagartos-Donate MJ, Cai CZ, Xie LM, Caponio D, Wang WW, Schmauck-Medina T, Zhang J, Wang HL, Lou G, Xiao X, Zheng W, Palikaras K, Yang G, Caldwell KA, Caldwell GA, Shen HM, Nilsen H, Lu JH, Fang EF
Nature biomedical engineering 2022 Jan;6(1):76-93
Nature biomedical engineering 2022 Jan;6(1):76-93
Cerebrospinal fluid p-tau231 as an early indicator of emerging pathology in Alzheimer's disease.
Ashton NJ, Benedet AL, Pascoal TA, Karikari TK, Lantero-Rodriguez J, Brum WS, Mathotaarachchi S, Therriault J, Savard M, Chamoun M, Stoops E, Francois C, Vanmechelen E, Gauthier S, Zimmer ER, Zetterberg H, Blennow K, Rosa-Neto P
EBioMedicine 2022 Feb;76:103836
EBioMedicine 2022 Feb;76:103836
Systematic Phenotyping and Characterization of the 3xTg-AD Mouse Model of Alzheimer's Disease.
Javonillo DI, Tran KM, Phan J, Hingco E, Kramár EA, da Cunha C, Forner S, Kawauchi S, Milinkeviciute G, Gomez-Arboledas A, Neumann J, Banh CE, Huynh M, Matheos DP, Rezaie N, Alcantara JA, Mortazavi A, Wood MA, Tenner AJ, MacGregor GR, Green KN, LaFerla FM
Frontiers in neuroscience 2021;15:785276
Frontiers in neuroscience 2021;15:785276
Dephosphorylation Passivates the Seeding Activity of Oligomeric Tau Derived From Alzheimer's Brain.
Wu R, Li L, Shi R, Zhou Y, Jin N, Gu J, Tung YC, Liu F, Chu D
Frontiers in molecular neuroscience 2021;14:631833
Frontiers in molecular neuroscience 2021;14:631833
Head-to-head comparison of clinical performance of CSF phospho-tau T181 and T217 biomarkers for Alzheimer's disease diagnosis.
Karikari TK, Emeršič A, Vrillon A, Lantero-Rodriguez J, Ashton NJ, Kramberger MG, Dumurgier J, Hourregue C, Čučnik S, Brinkmalm G, Rot U, Zetterberg H, Paquet C, Blennow K
Alzheimer's & dementia : the journal of the Alzheimer's Association 2021 May;17(5):755-767
Alzheimer's & dementia : the journal of the Alzheimer's Association 2021 May;17(5):755-767
Icariin protects against sodium azide-induced neurotoxicity by activating the PI3K/Akt/GSK-3β signaling pathway.
Zhang Y, Huang N, Lu H, Huang J, Jin H, Shi J, Jin F
PeerJ 2020;8:e8955
PeerJ 2020;8:e8955
Truncation of Tau selectively facilitates its pathological activities.
Gu J, Xu W, Jin N, Li L, Zhou Y, Chu D, Gong CX, Iqbal K, Liu F
The Journal of biological chemistry 2020 Oct 2;295(40):13812-13828
The Journal of biological chemistry 2020 Oct 2;295(40):13812-13828
Novel tau biomarkers phosphorylated at T181, T217 or T231 rise in the initial stages of the preclinical Alzheimer's continuum when only subtle changes in Aβ pathology are detected.
Suárez-Calvet M, Karikari TK, Ashton NJ, Lantero Rodríguez J, Milà-Alomà M, Gispert JD, Salvadó G, Minguillon C, Fauria K, Shekari M, Grau-Rivera O, Arenaza-Urquijo EM, Sala-Vila A, Sánchez-Benavides G, González-de-Echávarri JM, Kollmorgen G, Stoops E, Vanmechelen E, Zetterberg H, Blennow K, Molinuevo JL, ALFA Study
EMBO molecular medicine 2020 Dec 7;12(12):e12921
EMBO molecular medicine 2020 Dec 7;12(12):e12921
Pathological Tau From Alzheimer's Brain Induces Site-Specific Hyperphosphorylation and SDS- and Reducing Agent-Resistant Aggregation of Tau in vivo.
Miao J, Shi R, Li L, Chen F, Zhou Y, Tung YC, Hu W, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2019;11:34
Frontiers in aging neuroscience 2019;11:34
A post-translational modification signature defines changes in soluble tau correlating with oligomerization in early stage Alzheimer's disease brain.
Ercan-Herbst E, Ehrig J, Schöndorf DC, Behrendt A, Klaus B, Gomez Ramos B, Prat Oriol N, Weber C, Ehrnhoefer DE
Acta neuropathologica communications 2019 Dec 3;7(1):192
Acta neuropathologica communications 2019 Dec 3;7(1):192
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Zhou Y, Shi J, Chu D, Hu W, Guan Z, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2018;10:27
Frontiers in aging neuroscience 2018;10:27
Expression of Tau Pathology-Related Proteins in Different Brain Regions: A Molecular Basis of Tau Pathogenesis.
Hu W, Wu F, Zhang Y, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2017;9:311
Frontiers in aging neuroscience 2017;9:311
Liraglutide Improves Water Maze Learning and Memory Performance While Reduces Hyperphosphorylation of Tau and Neurofilaments in APP/PS1/Tau Triple Transgenic Mice.
Chen S, Sun J, Zhao G, Guo A, Chen Y, Fu R, Deng Y
Neurochemical research 2017 Aug;42(8):2326-2335
Neurochemical research 2017 Aug;42(8):2326-2335
Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment.
Smolek T, Madari A, Farbakova J, Kandrac O, Jadhav S, Cente M, Brezovakova V, Novak M, Zilka N
The Journal of comparative neurology 2016 Mar 1;524(4):874-95
The Journal of comparative neurology 2016 Mar 1;524(4):874-95
Age-dependent effects of A53T alpha-synuclein on behavior and dopaminergic function.
Oaks AW, Frankfurt M, Finkelstein DI, Sidhu A
PloS one 2013;8(4):e60378
PloS one 2013;8(4):e60378
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Yu Y, Run X, Liang Z, Li Y, Liu F, Liu Y, Iqbal K, Grundke-Iqbal I, Gong CX
Journal of neurochemistry 2009 Mar;108(6):1480-94
Journal of neurochemistry 2009 Mar;108(6):1480-94
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Haase C, Stieler JT, Arendt T, Holzer M
Journal of neurochemistry 2004 Mar;88(6):1509-20
Journal of neurochemistry 2004 Mar;88(6):1509-20
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Haase C, Stieler JT, Arendt T, Holzer M
Journal of neurochemistry 2004 Mar;88(6):1509-20
Journal of neurochemistry 2004 Mar;88(6):1509-20
Interaction of tau isoforms with Alzheimer's disease abnormally hyperphosphorylated tau and in vitro phosphorylation into the disease-like protein.
Alonso AD, Zaidi T, Novak M, Barra HS, Grundke-Iqbal I, Iqbal K
The Journal of biological chemistry 2001 Oct 12;276(41):37967-73
The Journal of biological chemistry 2001 Oct 12;276(41):37967-73
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
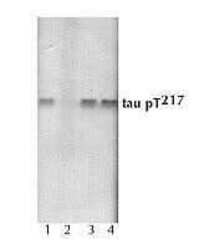
- Experimental details
- SF-9 cell extracts expressing human four repeat were resolved by SDS-PAGE on a 10% Tris-glycine gel. The proteins were transferred tonitrocellulose. Membranes were incubated with 0.50 µg/mL phospho tau (pT217) polyclonal antibody (Product # 44-744).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
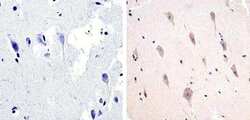
- Experimental details
- Immunohistochemistry analysis of Phospho-Tau (pT217) showing staining in the cytoplasm of paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Phospho-Tau (pT217) polyclonal antibody (Product # 44-744) diluted in 3% BSA-PBS at a dilution of 1:100 overnight at 4ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
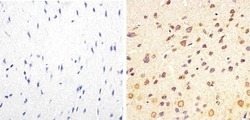
- Experimental details
- Immunohistochemistry analysis of Phospho-Tau (pT217) showing staining in the cytoplasm of paraffin-embedded mouse brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Phospho-Tau (pT217) polyclonal antibody (Product # 44-744) diluted in 3% BSA-PBS at a dilution of 1:50 overnight at 4ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Flow cytometry analysis of Tau [pT217] was done on SH-SY5Y cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with Tau [pT217] Rabbit Polyclonal Antibody (44744, red histogram) or with rabbit isotype control (pink histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Goat Anti-Rabbit Secondary Antibody (A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10,000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
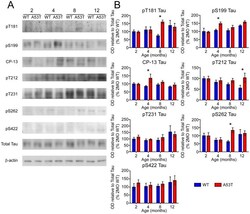
- Experimental details
- Figure 6 Screen for additional pJNK-Tau sites in striatum. (A) Phosphorylation of Tau protein at epitopes subject to phosphorylation by JNK was analyzed by immunoblot. (B) Band optical density (OD) from phosphorylation-specific probes relative to total Tau expression is presented as percent of 2 month-old WT (mean +- SEM) and was analyzed by two-way ANOVA with Bonferroni post-hoc tests comparing each A53T group to age-matched controls (*p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Effect of ICA on the phosphorylation level of Tau in NaN 3 -injured PC12 cells. (A) Representative bands showing PHF-1 and p-T217 in PC12 cells in the different groups. (B) Quantitative analysis of PHF-1 levels. (C) Quantitative analysis of p-T217 levels. Phosphorylated Tau was normalized to total Tau. The data are shown as the mean +- SEM, n = 5 (* P < 0.05 vs. the control group, # P < 0.05 vs. the NaN 3 group).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
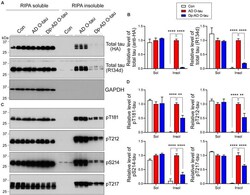
- FIGURE 5 Dephosphorylation of AD O-tau suppressed itsability to template tau aggregation. (A-D) HEK-293FT cellswere transfected with pCI/HA-tau 15 1-3 91 , treated with AD O-tau or Dp-AD O-tau and lysed withRIPA buffer. RIPA-soluble and-insoluble fractions were separated bycentrifugation and analyzed with Western blots developed withantibodies toward HA, total tau (R134d), GAPDH (A) , orvarious phosphorylated tau (C) . The experiment wasperformed in triplicate. Relative levels of total (B) orphosphorylated (D) tau were measured. ** P < 0.01. **** P < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Both HMW-tau and LMW-tau are selectively hyperphosphorylated in AD brain. (A) AD and control human brain homogenates were analyzed by western blots developed with the indicated site-specific and phosphorylation dependent anti-tau antibodies. (B,C) Blots were analyzed by densitometry. The levels of hyperphosphorylated HMW-tau (B) and LMW-tau (C) are shown as scattered dots with mean +- SD. ** p < 0.01; *** p < 0.001; **** p < 0.0001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 Compared to Braak 0-I samples, many but not all, tau phosphorylation events are increased in native Braak III-IV samples. Normalized phospho-tau signals obtained from ELISA measurements of samples from a ) entorhinal cortices (EC), b ) hippocampi (Hip) and c ) temporal cortices (TC). Student's t-tests: *, p < 0.05, **, p < 0.01, ***, p < 0.001
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot