44-758G
antibody from Invitrogen Antibodies
Targeting: MAPT
DDPAC, FLJ31424, FTDP-17, MAPTL, MGC138549, MSTD, MTBT1, MTBT2, PPND, PPP1R103, tau
Antibody data
- Antibody Data
- Antigen structure
- References [33]
- Comments [0]
- Validations
- Western blot [2]
- Immunohistochemistry [2]
- Other assay [18]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 44-758G - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-Tau (Ser404) Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human, Mouse, Rat
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Storage
- -20°C
Submitted references Laterality and region-specific tau phosphorylation correlate with PTSD-related behavioral traits in rats exposed to repetitive low-level blast.
A new non-aggregative splicing isoform of human Tau is decreased in Alzheimer's disease.
PKR kinase directly regulates tau expression and Alzheimer's disease-related tau phosphorylation.
IL-17 triggers the onset of cognitive and synaptic deficits in early stages of Alzheimer's disease.
Truncation of Tau selectively facilitates its pathological activities.
The behavioural and neuropathologic sexual dimorphism and absence of MIP-3α in tau P301S mouse model of Alzheimer's disease.
Dysregulation of histone acetylation pathways in hippocampus and frontal cortex of Alzheimer's disease patients.
Mitophagy Failure in APP and Tau Overexpression Model of Alzheimer's Disease.
Pathological Tau From Alzheimer's Brain Induces Site-Specific Hyperphosphorylation and SDS- and Reducing Agent-Resistant Aggregation of Tau in vivo.
A post-translational modification signature defines changes in soluble tau correlating with oligomerization in early stage Alzheimer's disease brain.
High Dietary Iron Disrupts Iron Homeostasis and Induces Amyloid-β and Phospho-τ Expression in the Hippocampus of Adult Wild-Type and APP/PS1 Transgenic Mice.
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Reinstating plasticity and memory in a tauopathy mouse model with an acetyltransferase activator.
Evidence of the impact of systemic inflammation on neuroinflammation from a non-bacterial endotoxin animal model.
Phosphorylation of nuclear Tau is modulated by distinct cellular pathways.
Tau secretion is correlated to an increase of Golgi dynamics.
A validated antibody panel for the characterization of tau post-translational modifications.
Opposing effects of progranulin deficiency on amyloid and tau pathologies via microglial TYROBP network.
Tau deletion promotes brain insulin resistance.
Accelerated aging exacerbates a pre-existing pathology in a tau transgenic mouse model.
Human Truncated Tau Induces Mature Neurofibrillary Pathology in a Mouse Model of Human Tauopathy.
Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment.
Rapid alteration of protein phosphorylation during postmortem: implication in the study of protein phosphorylation.
Neuronal uptake and propagation of a rare phosphorylated high-molecular-weight tau derived from Alzheimer's disease brain.
Quantitative phosphoproteomics of Alzheimer's disease reveals cross-talk between kinases and small heat shock proteins.
Amyloid and tau pathology of familial Alzheimer's disease APP/PS1 mouse model in a senescence phenotype background (SAMP8).
Specificity of anti-tau antibodies when analyzing mice models of Alzheimer's disease: problems and solutions.
MiR-26b, upregulated in Alzheimer's disease, activates cell cycle entry, tau-phosphorylation, and apoptosis in postmitotic neurons.
Microtubule-associated protein tau in bovine retinal photoreceptor rod outer segments: comparison with brain tau.
A proteomic analysis of MCLR-induced neurotoxicity: implications for Alzheimer's disease.
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Perez Garcia G, De Gasperi R, Gama Sosa MA, Perez GM, Otero-Pagan A, Pryor D, Abutarboush R, Kawoos U, Hof PR, Dickstein DL, Cook DG, Gandy S, Ahlers ST, Elder GA
Acta neuropathologica communications 2021 Mar 1;9(1):33
Acta neuropathologica communications 2021 Mar 1;9(1):33
A new non-aggregative splicing isoform of human Tau is decreased in Alzheimer's disease.
García-Escudero V, Ruiz-Gabarre D, Gargini R, Pérez M, García E, Cuadros R, Hernández IH, Cabrera JR, García-Escudero R, Lucas JJ, Hernández F, Ávila J
Acta neuropathologica 2021 Jul;142(1):159-177
Acta neuropathologica 2021 Jul;142(1):159-177
PKR kinase directly regulates tau expression and Alzheimer's disease-related tau phosphorylation.
Reimer L, Betzer C, Kofoed RH, Volbracht C, Fog K, Kurhade C, Nilsson E, Överby AK, Jensen PH
Brain pathology (Zurich, Switzerland) 2021 Jan;31(1):103-119
Brain pathology (Zurich, Switzerland) 2021 Jan;31(1):103-119
IL-17 triggers the onset of cognitive and synaptic deficits in early stages of Alzheimer's disease.
Brigas HC, Ribeiro M, Coelho JE, Gomes R, Gomez-Murcia V, Carvalho K, Faivre E, Costa-Pereira S, Darrigues J, de Almeida AA, Buée L, Dunot J, Marie H, Pousinha PA, Blum D, Silva-Santos B, Lopes LV, Ribot JC
Cell reports 2021 Aug 31;36(9):109574
Cell reports 2021 Aug 31;36(9):109574
Truncation of Tau selectively facilitates its pathological activities.
Gu J, Xu W, Jin N, Li L, Zhou Y, Chu D, Gong CX, Iqbal K, Liu F
The Journal of biological chemistry 2020 Oct 2;295(40):13812-13828
The Journal of biological chemistry 2020 Oct 2;295(40):13812-13828
The behavioural and neuropathologic sexual dimorphism and absence of MIP-3α in tau P301S mouse model of Alzheimer's disease.
Sun Y, Guo Y, Feng X, Jia M, Ai N, Dong Y, Zheng Y, Fu L, Yu B, Zhang H, Wu J, Yu X, Wu H, Kong W
Journal of neuroinflammation 2020 Feb 24;17(1):72
Journal of neuroinflammation 2020 Feb 24;17(1):72
Dysregulation of histone acetylation pathways in hippocampus and frontal cortex of Alzheimer's disease patients.
Schueller E, Paiva I, Blanc F, Wang XL, Cassel JC, Boutillier AL, Bousiges O
European neuropsychopharmacology : the journal of the European College of Neuropsychopharmacology 2020 Apr;33:101-116
European neuropsychopharmacology : the journal of the European College of Neuropsychopharmacology 2020 Apr;33:101-116
Mitophagy Failure in APP and Tau Overexpression Model of Alzheimer's Disease.
Martín-Maestro P, Gargini R, García E, Simón D, Avila J, García-Escudero V
Journal of Alzheimer's disease : JAD 2019;70(2):525-540
Journal of Alzheimer's disease : JAD 2019;70(2):525-540
Pathological Tau From Alzheimer's Brain Induces Site-Specific Hyperphosphorylation and SDS- and Reducing Agent-Resistant Aggregation of Tau in vivo.
Miao J, Shi R, Li L, Chen F, Zhou Y, Tung YC, Hu W, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2019;11:34
Frontiers in aging neuroscience 2019;11:34
A post-translational modification signature defines changes in soluble tau correlating with oligomerization in early stage Alzheimer's disease brain.
Ercan-Herbst E, Ehrig J, Schöndorf DC, Behrendt A, Klaus B, Gomez Ramos B, Prat Oriol N, Weber C, Ehrnhoefer DE
Acta neuropathologica communications 2019 Dec 3;7(1):192
Acta neuropathologica communications 2019 Dec 3;7(1):192
High Dietary Iron Disrupts Iron Homeostasis and Induces Amyloid-β and Phospho-τ Expression in the Hippocampus of Adult Wild-Type and APP/PS1 Transgenic Mice.
Chen M, Zheng J, Liu G, Zeng C, Xu E, Zhu W, Anderson GJ, Chen H
The Journal of nutrition 2019 Dec 1;149(12):2247-2254
The Journal of nutrition 2019 Dec 1;149(12):2247-2254
Relevance of Phosphorylation and Truncation of Tau to the Etiopathogenesis of Alzheimer's Disease.
Zhou Y, Shi J, Chu D, Hu W, Guan Z, Gong CX, Iqbal K, Liu F
Frontiers in aging neuroscience 2018;10:27
Frontiers in aging neuroscience 2018;10:27
Reinstating plasticity and memory in a tauopathy mouse model with an acetyltransferase activator.
Chatterjee S, Cassel R, Schneider-Anthony A, Merienne K, Cosquer B, Tzeplaeff L, Halder Sinha S, Kumar M, Chaturbedy P, Eswaramoorthy M, Le Gras S, Keime C, Bousiges O, Dutar P, Petsophonsakul P, Rampon C, Cassel JC, Buée L, Blum D, Kundu TK, Boutillier AL
EMBO molecular medicine 2018 Nov;10(11)
EMBO molecular medicine 2018 Nov;10(11)
Evidence of the impact of systemic inflammation on neuroinflammation from a non-bacterial endotoxin animal model.
Huang C, Irwin MG, Wong GTC, Chang RCC
Journal of neuroinflammation 2018 May 17;15(1):147
Journal of neuroinflammation 2018 May 17;15(1):147
Phosphorylation of nuclear Tau is modulated by distinct cellular pathways.
Ulrich G, Salvadè A, Boersema P, Calì T, Foglieni C, Sola M, Picotti P, Papin S, Paganetti P
Scientific reports 2018 Dec 7;8(1):17702
Scientific reports 2018 Dec 7;8(1):17702
Tau secretion is correlated to an increase of Golgi dynamics.
Mohamed NV, Desjardins A, Leclerc N
PloS one 2017;12(5):e0178288
PloS one 2017;12(5):e0178288
A validated antibody panel for the characterization of tau post-translational modifications.
Ercan E, Eid S, Weber C, Kowalski A, Bichmann M, Behrendt A, Matthes F, Krauss S, Reinhardt P, Fulle S, Ehrnhoefer DE
Molecular neurodegeneration 2017 Nov 21;12(1):87
Molecular neurodegeneration 2017 Nov 21;12(1):87
Opposing effects of progranulin deficiency on amyloid and tau pathologies via microglial TYROBP network.
Takahashi H, Klein ZA, Bhagat SM, Kaufman AC, Kostylev MA, Ikezu T, Strittmatter SM, Alzheimer’s Disease Neuroimaging Initiative
Acta neuropathologica 2017 May;133(5):785-807
Acta neuropathologica 2017 May;133(5):785-807
Tau deletion promotes brain insulin resistance.
Marciniak E, Leboucher A, Caron E, Ahmed T, Tailleux A, Dumont J, Issad T, Gerhardt E, Pagesy P, Vileno M, Bournonville C, Hamdane M, Bantubungi K, Lancel S, Demeyer D, Eddarkaoui S, Vallez E, Vieau D, Humez S, Faivre E, Grenier-Boley B, Outeiro TF, Staels B, Amouyel P, Balschun D, Buee L, Blum D
The Journal of experimental medicine 2017 Aug 7;214(8):2257-2269
The Journal of experimental medicine 2017 Aug 7;214(8):2257-2269
Accelerated aging exacerbates a pre-existing pathology in a tau transgenic mouse model.
Bodea LG, Evans HT, Van der Jeugd A, Ittner LM, Delerue F, Kril J, Halliday G, Hodges J, Kiernan MC, Götz J
Aging cell 2017 Apr;16(2):377-386
Aging cell 2017 Apr;16(2):377-386
Human Truncated Tau Induces Mature Neurofibrillary Pathology in a Mouse Model of Human Tauopathy.
Zimova I, Brezovakova V, Hromadka T, Weisova P, Cubinkova V, Valachova B, Filipcik P, Jadhav S, Smolek T, Novak M, Zilka N
Journal of Alzheimer's disease : JAD 2016 Sep 6;54(2):831-43
Journal of Alzheimer's disease : JAD 2016 Sep 6;54(2):831-43
Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment.
Smolek T, Madari A, Farbakova J, Kandrac O, Jadhav S, Cente M, Brezovakova V, Novak M, Zilka N
The Journal of comparative neurology 2016 Mar 1;524(4):874-95
The Journal of comparative neurology 2016 Mar 1;524(4):874-95
Rapid alteration of protein phosphorylation during postmortem: implication in the study of protein phosphorylation.
Wang Y, Zhang Y, Hu W, Xie S, Gong CX, Iqbal K, Liu F
Scientific reports 2015 Oct 29;5:15709
Scientific reports 2015 Oct 29;5:15709
Neuronal uptake and propagation of a rare phosphorylated high-molecular-weight tau derived from Alzheimer's disease brain.
Takeda S, Wegmann S, Cho H, DeVos SL, Commins C, Roe AD, Nicholls SB, Carlson GA, Pitstick R, Nobuhara CK, Costantino I, Frosch MP, Müller DJ, Irimia D, Hyman BT
Nature communications 2015 Oct 13;6:8490
Nature communications 2015 Oct 13;6:8490
Quantitative phosphoproteomics of Alzheimer's disease reveals cross-talk between kinases and small heat shock proteins.
Dammer EB, Lee AK, Duong DM, Gearing M, Lah JJ, Levey AI, Seyfried NT
Proteomics 2015 Jan;15(2-3):508-519
Proteomics 2015 Jan;15(2-3):508-519
Amyloid and tau pathology of familial Alzheimer's disease APP/PS1 mouse model in a senescence phenotype background (SAMP8).
Porquet D, Andrés-Benito P, Griñán-Ferré C, Camins A, Ferrer I, Canudas AM, Del Valle J, Pallàs M
Age (Dordrecht, Netherlands) 2015 Feb;37(1):9747
Age (Dordrecht, Netherlands) 2015 Feb;37(1):9747
Specificity of anti-tau antibodies when analyzing mice models of Alzheimer's disease: problems and solutions.
Petry FR, Pelletier J, Bretteville A, Morin F, Calon F, Hébert SS, Whittington RA, Planel E
PloS one 2014;9(5):e94251
PloS one 2014;9(5):e94251
MiR-26b, upregulated in Alzheimer's disease, activates cell cycle entry, tau-phosphorylation, and apoptosis in postmitotic neurons.
Absalon S, Kochanek DM, Raghavan V, Krichevsky AM
The Journal of neuroscience : the official journal of the Society for Neuroscience 2013 Sep 11;33(37):14645-59
The Journal of neuroscience : the official journal of the Society for Neuroscience 2013 Sep 11;33(37):14645-59
Microtubule-associated protein tau in bovine retinal photoreceptor rod outer segments: comparison with brain tau.
Yamazaki A, Nishizawa Y, Matsuura I, Hayashi F, Usukura J, Bondarenko VA
Biochimica et biophysica acta 2013 Oct;1832(10):1549-59
Biochimica et biophysica acta 2013 Oct;1832(10):1549-59
A proteomic analysis of MCLR-induced neurotoxicity: implications for Alzheimer's disease.
Li G, Cai F, Yan W, Li C, Wang J
Toxicological sciences : an official journal of the Society of Toxicology 2012 Jun;127(2):485-95
Toxicological sciences : an official journal of the Society of Toxicology 2012 Jun;127(2):485-95
Developmental regulation of tau phosphorylation, tau kinases, and tau phosphatases.
Yu Y, Run X, Liang Z, Li Y, Liu F, Liu Y, Iqbal K, Grundke-Iqbal I, Gong CX
Journal of neurochemistry 2009 Mar;108(6):1480-94
Journal of neurochemistry 2009 Mar;108(6):1480-94
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Haase C, Stieler JT, Arendt T, Holzer M
Journal of neurochemistry 2004 Mar;88(6):1509-20
Journal of neurochemistry 2004 Mar;88(6):1509-20
Pseudophosphorylation of tau protein alters its ability for self-aggregation.
Haase C, Stieler JT, Arendt T, Holzer M
Journal of neurochemistry 2004 Mar;88(6):1509-20
Journal of neurochemistry 2004 Mar;88(6):1509-20
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot using rabbit anti-Tau (pS404) polyclonal antibody.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of Tau (pS404) was performed by loading 20 µg of Mouse Brain (lane1), Rat Brain (lane2), Mouse Pancreas (lane3) and Mouse Kidney (lane4) tissue lysate using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0322BOX), XCell SureLock™ Electrophoresis System (Product # EI0002), Novex® Sharp Pre-Stained Protein Standard (LC5800), and iBlot® Dry Blotting System (IB21001). Proteins were transferred to a nitrocellulose membrane and blocked with 5 % skim milk for 1 hour at room temperature. Tau (pS404) was detected at ~79 kDa using Tau (pS404) Rabbit Polyclonal Antibody (Product # 44-758G) at 1:1000 dilution in 5 % skim milk at 4ºC overnight on a rocking platform. Goat Anti-Rabbit IgG - HRP Secondary Antibody (G21234) at 1:5000 dilution was used and chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
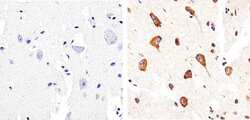
- Experimental details
- Immunohistochemistry analysis of Phospho-Tau (pS404) showing staining in the cytoplasm of paraffin-embedded human brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Phospho-Tau (pS404) polyclonal antibody (Product # 44-758G) diluted in 3% BSA-PBS at a dilution of 1:50 overnight at 4ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
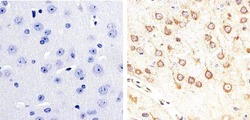
- Experimental details
- Immunohistochemistry analysis of Phospho-Tau (pS404) showing staining in the cytoplasm of paraffin-embedded mouse brain tissue (right) compared to a negative control without primary antibody (left). To expose target proteins, antigen retrieval was performed using 10mM sodium citrate (pH 6.0), microwaved for 8-15 min. Following antigen retrieval, tissues were blocked in 3% H2O2-methanol for 15 min at room temperature, washed with ddH2O and PBS, and then probed with a Phospho-Tau (pS404) polyclonal antibody (Product # 44-758G) diluted in 3% BSA-PBS at a dilution of 1:20 overnight at 4ºC in a humidified chamber. Tissues were washed extensively in PBST and detection was performed using an HRP-conjugated secondary antibody followed by colorimetric detection using a DAB kit. Tissues were counterstained with hematoxylin and dehydrated with ethanol and xylene to prep for mounting.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
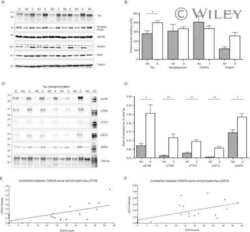
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
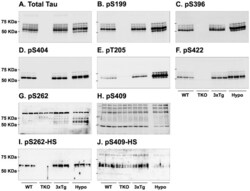
- Experimental details
- Figure 7 Analysis of tau signal with polyclonal antibodies by Western blotting. Proteins were extracted from the cortex of 3 mouse lines: control mice (WT and Hypothermic), Tau KO mice and 3xTg-AD mice. Proteins were separated by SDS-PAGE and then identified with the following polyclonal antibodies: A: Total Tau, B: pS199, C: pS396, D: pS404, E: pT205, F: pS422, G: pS262 and H: pS409. Normal anti-rabbit secondary antibodies were used to detect primary antibodies. The heat stable fraction was used to remove non-specificity: I: pS262 and J: pS409. Quantifications of the blots are available in Figure S5 .
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
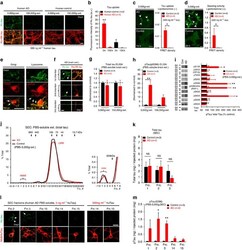
- Figure 6 Neuronal uptake of PBS-soluble HMW tau derived from human AD brain. ( a , b ) Primary neurons were incubated with AD or control brain extracts (cases were matched for age and postmortem interval ( Supplementary Table S1 )) and immunostained at day 2 ( a ). ( b ) Quantification of fluorescence intensity of human tau staining. One-way ANOVA and a subsequent Tukey-Kramer test. ( c , d ) Tau uptake ( c ) and seeding activity ( d ) assay in HEK-tau-biosensor cells. (Mann-Whitney U -test) ( e ) Subcellular localization of human tau taken up by neurons (PBS-3,000 g , 500 ng ml -1 human tau). ( f ) Neuron-to-neuron transfer of tau in a 3-chamber microfluidic device. AD brain extract (PBS-3,000 g , 500 ng ml -1 human tau) was added to the 1st chamber. Human tau positive neurons were detected in both the 1st and 2nd chamber at day 7 (arrow). ( g , h ) Quantification of total-tau ( g ) and phospho-tau ( h ) levels in AD and control brain extract (ELISA). Unpaired t -test. ( i ) Brain extracts were immunoblotted with phospho-tau specific antibodies recognizing different epitopes. Representative immunoblot and quantification of phospho-tau levels at each epitope. Unpaired t -test. ( j , k ) SEC analysis of PBS-soluble tau from AD and control brain. ( j ) Representative graph of total tau levels (ELISA) in SEC-separated samples. Small peaks for HMW fractions were detected in both groups (right panel). ( k ) Mean total tau levels of HMW SEC fractions. ( l ) Tau uptake from each SEC
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
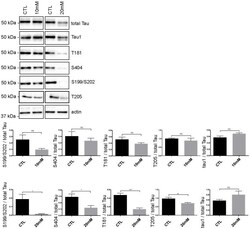
- Fig 5 Tau phosphorylation is decreased in neurons treated with 20 mM KCl. Immunoblot analysis of Tau phosphorylation in neurons treated either with 10mM or 20mM KCl. The phosphorylation of S199/S202, T181, T205 and S404 was not significantly affected by 10mM of KCl treatment whereas the phosphorylation of all these sites was decreased by 20mM of KCl treatment. The signal of the phospho-antibodies was normalized to that of total Tau (n = 5, mean +- SEM, unpaired t-test two-tailed, * P < 0.05, *** P < 0.001). The amount of dephosphorylated Tau revealed by the Tau-1 antibody was normalized to that of total Tau lysate (n = 5, mean +- SEM, unpaired t-test two-tailed, * P < 0.05).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
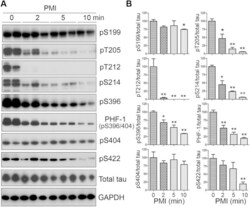
- Figure 1 Tau is rapidly dephosphorylated site-specifically in mouse brains during postmortem. Mice were scarified by cervical dislocation. The dead animal bodies were kept at room temperature for the indicated periods of time. ( A ) Phosphorylation of tau was analyzed by Western blots developed with phosphorylation-dependent and site-specific tau antibodies indicated at the right side of the blots. ( B ) The levels of tau phosphorylation at individual sites were quantified and normalized with total tau level and presented as mean +- SEM. (n = 3-4). * p < 0.05; ** p < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4 Correlations between levels of p-tau Thr231, Ser396 and Ser404 with open arm time in the elevated zero maze. Levels of p-tau Thr231 ( a ), Ser396 ( b ) and Ser404 ( c ) in the right hippocampus were previously determined by Western blotting [] at 40 weeks after blast exposure and correlated with open arm time in the elevated zero maze determined at 30 weeks following blast exposure []. Five blast and five control animals were analyzed. Kendal's tau-b correlation coefficients and p values are indicated
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 6 Immunoblotting using hTg-Tau and Tau-KO brain lysates confirms the specificity for tau for a panel of antibodies. a ) Scheme of tau (2N4R) showing the epitopes of the Tau12, Tau5, Tau1, HT7, BT2 and Dako antibodies. b ) - d' ) Immunoblots demonstrate strong reactivity of all antibodies with tau bands between 40 and 60 kD. Minor non-specific bands present in Tau-KO brain lysates were detected in d ), e ), i ), h ), p ), x ), y ) and c '). Isoform-specific antibodies in h ), i ), j ) and k ) were validated using a tau ladder with all 6 recombinant human tau isoforms. kDa sizes for the marker are given in e' ). Tg: hTg-Tau mouse, KO: Tau-KO mouse brain lysate, TL: recombinant tau ladder
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 8 Human entorhinal cortex (EC) lysates show many but not all tau PTMs. a - e Immunoblots demonstrate strong reactivity of all pan-tau antibodies with human EC ( n = 3). f - i detection with isoform specific tau antibodies reveals the presence of multiple isoforms of tau. TL: recombinant tau ladder. j - t PTM-specific antibodies demonstrate reactivity except for j nY18, k AcK280, n pT212, p pS238 and t pS422. u Immunoblotting with actin for loading control shows similar loading for three different EC lysates (n = 3). v kDa sizes for the marker
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Subcellular fractionation reveals location-specific Tau modification. ( a ) Cell lysates, cytosolic and nuclear fractions obtained from biological triplicates of C17.2 cells induced for human Tau expression for 24 hr are analysed by western blot with antibodies for Tau (Tau13), for the cytosolic GAPDH marker and for the nuclear histone 3 (H3) marker. Molecular weight markers are shown on the left. Full blots are presented in Supplementary Fig. 4 online. ( b ) Quantification is plotted as mean percent +- SD recovered in the two subcellular fractions. ( c ) Cytosolic and TCA-precipated nuclear fractions are analysed by western blot with antibodies for pan-Tau, against the indicated phosphosites or for dephosphorylated Ser 195 / 198 / 199 / 202 (biological triplicates). ( d ) Quantification of modified Tau normalized for the amount of total Tau detected in each sample. To facilitate the comparison between sites, each modified site is further normalized with the respective values measured in the cytosolic fraction. Values are mean +- SD, n = 3. 2way ANOVA and Bonferroni's multiple comparison test, *p < 0.05, ***p < 0.001.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 DNA damage induces de-phosphorylation of nuclear Tau. ( a ) The effect of Etoposide and Vinblastine treatment on mouse C17.2 cells is shown by confocal microscopy upon immune staining of PFA-fixed cells with antibodies against the microtubule marker beta-tubulin (in cyan) or the DNA damage marker gammaH2A-X (in red). ( b ) Confocal microscopic quantification of the activated kinases in the nucleus (DAPI mask). Mean percent +- sem relative to the respective controls. 2-tailed unpaired Mann-Whitney test, ****p < 0.0001. ( c ) Confocal microscopy images of C17.2 cells with induced Tau 441 expression treated in the absence or presence of 60 uM Etoposide for 5 hr and stained for with the human Tau antibody (Tau13). Quantification of immune fluorescent detection of human Tau in DAPI-stained nuclei. Mean percent +- sem relative to the control. 2-tailed unpaired Mann-Whitney test, ****p < 0.0001. ( d ) Cytosolic and nuclear fractions obtained from biological replicates of C17.2 cells with induced Tau expression and treated as indicated are analysed by western blot with antibodies for the two phosphosites pT 181 and pS 404 and for the dephosphorylated Tau1 epitope are quantified upon normalization for total Tau. Data represent mean percent +- SD relative to the untreated controls. Non-parametric Kruskal-Wallis test, *p < 0.05, **p < 0.01.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 2 Compared to Braak 0-I samples, many but not all, tau phosphorylation events are increased in native Braak III-IV samples. Normalized phospho-tau signals obtained from ELISA measurements of samples from a ) entorhinal cortices (EC), b ) hippocampi (Hip) and c ) temporal cortices (TC). Student's t-tests: *, p < 0.05, **, p < 0.01, ***, p < 0.001
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 5 Quantification and statistical analysis of IHC positive mouse brain sections Mouse brain slides were stained via IHC with primary antibodies of anti-pTau(S202/T205) (AT8), anti-pTau(S396) (PHF 13), anti-pTau(S404), Iba1 (EPR16588) (to detect active microglia), and anti-GFAP (2E1.E9) (to detect astrocytes) are showed in ( a ). Positive areas in the hippocampus were quantified using Aperio ImageScope and are shown in b - f , respectively. Statistical analysis was performed between the sex- and age-matched P301S and WTs. * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
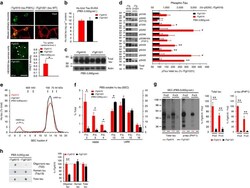
- Figure 2 Lack of PBS-soluble phosphorylated HMW tau species is associated with low tau uptake in primary neurons. ( a , top) Uptake of human tau from brain extracts from rTg4510 and rTg21221 mice by primary neurons (PBS-3,000 g , 500 ng ml -1 human tau). Neurons were immunostained with human tau-specific antibody (green) and total (human and mouse) tau antibody (red). ( a , bottom) Tau uptake assay in HEK-tau-biosensor cells. Brain extracts (10 mug protein) were applied to the cells (lipofectamie (-)). ( n =4) Unpaired t -test. Scale bar, 50 mum. ( b ) Human tau levels in brain extracts (ELISA). ( c ) Immunoblot analysis of PBS-soluble extracts with total tau antibody (DA9). Up-shifted bands in rTg4510 brain suggest phosphorylation of tau (arrow). ( d ) Brain extracts were immunoblotted with phospho-tau specific antibodies recognizing different epitopes. Representative immunoblot and quantification of phospho-tau levels at each epitope. ( n =3-4) Unpaired t -test. ( e , f ) SEC analysis of PBS-soluble tau. ( e ) Representative graph of human tau levels (ELISA) in SEC-separated samples ( f ) Mean human tau levels of HMW (Frc. 2-4) and LMW (Frc. 13-16) SEC fractions. ( n =3-6) Unpaired t -test. ( g ) Immunoblot analysis (SDS-PAGE) of SEC-separated fractions from brain extracts (total tau, DAKO). Quantification of band density is also shown (right graphs) ( n =4). Unpaired t -test. ( h ) Dot blot analysis of PBS-soluble brain extracts with tau oligomer-specific antibody (T22), h
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIGURE 3 Amounts of ferritin, p-tau (Ser404), and Abeta 1-42 protein in the hippocampus of male WT and APP/PS1 mice fed a control or high iron diet at age 30 wk (A). The immunoblot signals were quantified, and values were normalized using beta-tubulin expression (B). Values are means +- SDs, n = 7. The P value of the interaction is presented when it reaches significance, and further analysis of diet simple effects and genotype simple effects were then performed. *** P < 0.001, different because of diet; ### P < 0.001, different because of genotype (B). Abeta 1-42 , beta amyloid 1-42; APP/PS1, amyloid precursor protein/presenilin 1; APP/PS1-Ctrl, APP/PS1 mice fed a control diet; APP/PS1-High Fe, APP/PS1 mice fed a high iron diet; p-tau, phospho-tau; WT, wild-type; WT-Ctrl, wild-type mice fed a control diet; WT-High Fe, wild-type mice fed a high iron diet.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot