Antibody data
- Antibody Data
- Antigen structure
- References [31]
- Comments [0]
- Validations
- Western blot [2]
- Immunocytochemistry [2]
- Flow cytometry [1]
- Other assay [9]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA1-063A - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- EEA1 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Synthetic peptide
- Description
- PA1-063A detects early endosomal antigen 1 (EEA1) from mouse and human samples. Previous lots of this antibody have been used on hamster, canine, and rat samples. PA1-063A has been successfully used in Western blot procedures. By Western blot, this antibody detects an ~160 kDa protein representing EEA1 from mouse brain extract. Previous lots of this antibody have been used in immunoprecipitation and immunofluorescence procedures. The PA1-063A immunogen is a synthetic peptide corresponding to residues T(1391) P S S K K P V R V C D A C F N D L Q G(1410) of human EEA1. The PA1-063A immunizing peptide (Cat. # PEP-079) is available for use in neutralization and control experiments.
- Reactivity
- Human, Mouse
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 μg
- Concentration
- 1 mg/mL
- Storage
- -20°C, Avoid Freeze/Thaw Cycles
Submitted references ER-lysosome lipid transfer protein VPS13C/PARK23 prevents aberrant mtDNA-dependent STING signaling.
Osteoblastic Swedish mutant APP expedites brain deficits by inducing endoplasmic reticulum stress-driven senescence.
Analysis of the intracellular traffic of IgG in the context of Down syndrome (trisomy 21).
GIPR antagonist antibodies conjugated to GLP-1 peptide are bispecific molecules that decrease weight in obese mice and monkeys.
Uncoupling endosomal CLC chloride/proton exchange causes severe neurodegeneration.
Photoactivation of sulfonated polyplexes enables localized gene silencing by DsiRNA in breast cancer cells.
A conserved dendritic-cell regulatory program limits antitumour immunity.
The class II phosphoinositide 3-kinases PI3K-C2α and PI3K-C2β differentially regulate clathrin-dependent pinocytosis in human vascular endothelial cells.
Coxiella burnetii Type 4B Secretion System-dependent manipulation of endolysosomal maturation is required for bacterial growth.
Reduction of aluminum ion neurotoxicity through a small peptide application - NAP treatment of Alzheimer's disease.
Inducing cell death in vitro in cancer cells by targeted delivery of cytochrome c via a transferrin conjugate.
Human plasma C3 is essential for the development of memory B, but not T, lymphocytes.
Clathrin coat controls synaptic vesicle acidification by blocking vacuolar ATPase activity.
Caspase-mediated proteolysis of the sorting nexin 2 disrupts retromer assembly and potentiates Met/hepatocyte growth factor receptor signaling.
GM130 gain-of-function induces cell pathology in a model of lysosomal storage disease.
UNC93B1 physically associates with human TLR8 and regulates TLR8-mediated signaling.
Abnormal trafficking and degradation of TLR4 underlie the elevated inflammatory response in cystic fibrosis.
The phospholipase A₂ enzyme complex PAFAH Ib mediates endosomal membrane tubule formation and trafficking.
Physiological relevance of cell-specific distribution patterns of CFTR, NKCC1, NBCe1, and NHE3 along the crypt-villus axis in the intestine.
H-ras resides on clathrin-independent ARF6 vesicles that harbor little RAF-1, but not on clathrin-dependent endosomes.
Essential role of endocytosis of the type II transmembrane serine protease TMPRSS6 in regulating its functionality.
HIV-1 Nef binds a subpopulation of MHC-I throughout its trafficking itinerary and down-regulates MHC-I by perturbing both anterograde and retrograde trafficking.
Mitochondrially localized EGFR is independent of its endocytosis and associates with cell viability.
Endocytosis in cultured neurons is altered by chronic alcohol exposure.
Modulation of endocytic trafficking and apical stability of CFTR in primary human airway epithelial cultures.
Differential functions of ApoER2 and very low density lipoprotein receptor in Reelin signaling depend on differential sorting of the receptors.
Assembly of connexin43 into gap junctions is regulated differentially by E-cadherin and N-cadherin in rat liver epithelial cells.
E-cadherin differentially regulates the assembly of Connexin43 and Connexin32 into gap junctions in human squamous carcinoma cells.
Urocortin trafficking in cerebral microvessel endothelial cells.
Urocortin trafficking in cerebral microvessel endothelial cells.
Misfolded BRICHOS SP-C mutant proteins induce apoptosis via caspase-4- and cytochrome c-related mechanisms.
Hancock-Cerutti W, Wu Z, Xu P, Yadavalli N, Leonzino M, Tharkeshwar AK, Ferguson SM, Shadel GS, De Camilli P
The Journal of cell biology 2022 Jul 4;221(7)
The Journal of cell biology 2022 Jul 4;221(7)
Osteoblastic Swedish mutant APP expedites brain deficits by inducing endoplasmic reticulum stress-driven senescence.
Pan JX, Sun D, Lee D, Xiong L, Ren X, Guo HH, Yao LL, Lu Y, Jung C, Xiong WC
Communications biology 2021 Nov 25;4(1):1326
Communications biology 2021 Nov 25;4(1):1326
Analysis of the intracellular traffic of IgG in the context of Down syndrome (trisomy 21).
Cejas RB, Tamaño-Blanco M, Blanco JG
Scientific reports 2021 May 26;11(1):10981
Scientific reports 2021 May 26;11(1):10981
GIPR antagonist antibodies conjugated to GLP-1 peptide are bispecific molecules that decrease weight in obese mice and monkeys.
Lu SC, Chen M, Atangan L, Killion EA, Komorowski R, Cheng Y, Netirojjanakul C, Falsey JR, Stolina M, Dwyer D, Hale C, Stanislaus S, Hager T, Thomas VA, Harrold JM, Lloyd DJ, Véniant MM
Cell reports. Medicine 2021 May 18;2(5):100263
Cell reports. Medicine 2021 May 18;2(5):100263
Uncoupling endosomal CLC chloride/proton exchange causes severe neurodegeneration.
Weinert S, Gimber N, Deuschel D, Stuhlmann T, Puchkov D, Farsi Z, Ludwig CF, Novarino G, López-Cayuqueo KI, Planells-Cases R, Jentsch TJ
The EMBO journal 2020 May 4;39(9):e103358
The EMBO journal 2020 May 4;39(9):e103358
Photoactivation of sulfonated polyplexes enables localized gene silencing by DsiRNA in breast cancer cells.
Puri A, Viard M, Zakrevsky P, Zampino S, Chen A, Isemann C, Alvi S, Clogston J, Chitgupi U, Lovell JF, Shapiro BA
Nanomedicine : nanotechnology, biology, and medicine 2020 Jun;26:102176
Nanomedicine : nanotechnology, biology, and medicine 2020 Jun;26:102176
A conserved dendritic-cell regulatory program limits antitumour immunity.
Maier B, Leader AM, Chen ST, Tung N, Chang C, LeBerichel J, Chudnovskiy A, Maskey S, Walker L, Finnigan JP, Kirkling ME, Reizis B, Ghosh S, D'Amore NR, Bhardwaj N, Rothlin CV, Wolf A, Flores R, Marron T, Rahman AH, Kenigsberg E, Brown BD, Merad M
Nature 2020 Apr;580(7802):257-262
Nature 2020 Apr;580(7802):257-262
The class II phosphoinositide 3-kinases PI3K-C2α and PI3K-C2β differentially regulate clathrin-dependent pinocytosis in human vascular endothelial cells.
Aung KT, Yoshioka K, Aki S, Ishimaru K, Takuwa N, Takuwa Y
The journal of physiological sciences : JPS 2019 Mar;69(2):263-280
The journal of physiological sciences : JPS 2019 Mar;69(2):263-280
Coxiella burnetii Type 4B Secretion System-dependent manipulation of endolysosomal maturation is required for bacterial growth.
Samanta D, Clemente TM, Schuler BE, Gilk SD
PLoS pathogens 2019 Dec;15(12):e1007855
PLoS pathogens 2019 Dec;15(12):e1007855
Reduction of aluminum ion neurotoxicity through a small peptide application - NAP treatment of Alzheimer's disease.
Yang MH, Chen SC, Lin YF, Lee YC, Huang MY, Chen KC, Wu HY, Lin PC, Gozes I, Tyan YC
Journal of food and drug analysis 2019 Apr;27(2):551-564
Journal of food and drug analysis 2019 Apr;27(2):551-564
Inducing cell death in vitro in cancer cells by targeted delivery of cytochrome c via a transferrin conjugate.
Saxena M, Delgado Y, Sharma RK, Sharma S, Guzmán SLPL, Tinoco AD, Griebenow K
PloS one 2018;13(4):e0195542
PloS one 2018;13(4):e0195542
Human plasma C3 is essential for the development of memory B, but not T, lymphocytes.
Jiménez-Reinoso A, Marin AV, Subias M, López-Lera A, Román-Ortiz E, Payne K, Ma CS, Arbore G, Kolev M, Freeley SJ, Kemper C, Tangye SG, Fernández-Malavé E, Rodríguez de Córdoba S, López-Trascasa M, Regueiro JR
The Journal of allergy and clinical immunology 2018 Mar;141(3):1151-1154.e14
The Journal of allergy and clinical immunology 2018 Mar;141(3):1151-1154.e14
Clathrin coat controls synaptic vesicle acidification by blocking vacuolar ATPase activity.
Farsi Z, Gowrisankaran S, Krunic M, Rammner B, Woehler A, Lafer EM, Mim C, Jahn R, Milosevic I
eLife 2018 Apr 13;7
eLife 2018 Apr 13;7
Caspase-mediated proteolysis of the sorting nexin 2 disrupts retromer assembly and potentiates Met/hepatocyte growth factor receptor signaling.
Duclos CM, Champagne A, Carrier JC, Saucier C, Lavoie CL, Denault JB
Cell death discovery 2017;3:16100
Cell death discovery 2017;3:16100
GM130 gain-of-function induces cell pathology in a model of lysosomal storage disease.
Roy E, Bruyère J, Flamant P, Bigou S, Ausseil J, Vitry S, Heard JM
Human molecular genetics 2012 Apr 1;21(7):1481-95
Human molecular genetics 2012 Apr 1;21(7):1481-95
UNC93B1 physically associates with human TLR8 and regulates TLR8-mediated signaling.
Itoh H, Tatematsu M, Watanabe A, Iwano K, Funami K, Seya T, Matsumoto M
PloS one 2011;6(12):e28500
PloS one 2011;6(12):e28500
Abnormal trafficking and degradation of TLR4 underlie the elevated inflammatory response in cystic fibrosis.
Bruscia EM, Zhang PX, Satoh A, Caputo C, Medzhitov R, Shenoy A, Egan ME, Krause DS
Journal of immunology (Baltimore, Md. : 1950) 2011 Jun 15;186(12):6990-8
Journal of immunology (Baltimore, Md. : 1950) 2011 Jun 15;186(12):6990-8
The phospholipase A₂ enzyme complex PAFAH Ib mediates endosomal membrane tubule formation and trafficking.
Bechler ME, Doody AM, Ha KD, Judson BL, Chen I, Brown WJ
Molecular biology of the cell 2011 Jul 1;22(13):2348-59
Molecular biology of the cell 2011 Jul 1;22(13):2348-59
Physiological relevance of cell-specific distribution patterns of CFTR, NKCC1, NBCe1, and NHE3 along the crypt-villus axis in the intestine.
Jakab RL, Collaco AM, Ameen NA
American journal of physiology. Gastrointestinal and liver physiology 2011 Jan;300(1):G82-98
American journal of physiology. Gastrointestinal and liver physiology 2011 Jan;300(1):G82-98
H-ras resides on clathrin-independent ARF6 vesicles that harbor little RAF-1, but not on clathrin-dependent endosomes.
McKay J, Wang X, Ding J, Buss JE, Ambrosio L
Biochimica et biophysica acta 2011 Feb;1813(2):298-307
Biochimica et biophysica acta 2011 Feb;1813(2):298-307
Essential role of endocytosis of the type II transmembrane serine protease TMPRSS6 in regulating its functionality.
Béliveau F, Brulé C, Désilets A, Zimmerman B, Laporte SA, Lavoie CL, Leduc R
The Journal of biological chemistry 2011 Aug 19;286(33):29035-29043
The Journal of biological chemistry 2011 Aug 19;286(33):29035-29043
HIV-1 Nef binds a subpopulation of MHC-I throughout its trafficking itinerary and down-regulates MHC-I by perturbing both anterograde and retrograde trafficking.
Yi L, Rosales T, Rose JJ, Chowdhury B, Knutson JR, Venkatesan S
The Journal of biological chemistry 2010 Oct 1;285(40):30884-905
The Journal of biological chemistry 2010 Oct 1;285(40):30884-905
Mitochondrially localized EGFR is independent of its endocytosis and associates with cell viability.
Yao Y, Wang G, Li Z, Yan B, Guo Y, Jiang X, Xi Z
Acta biochimica et biophysica Sinica 2010 Nov;42(11):763-70
Acta biochimica et biophysica Sinica 2010 Nov;42(11):763-70
Endocytosis in cultured neurons is altered by chronic alcohol exposure.
Marín MP, Esteban-Pretel G, Ponsoda X, Romero AM, Ballestín R, López C, Megías L, Timoneda J, Molowny A, Canales JJ, Renau-Piqueras J
Toxicological sciences : an official journal of the Society of Toxicology 2010 May;115(1):202-13
Toxicological sciences : an official journal of the Society of Toxicology 2010 May;115(1):202-13
Modulation of endocytic trafficking and apical stability of CFTR in primary human airway epithelial cultures.
Cholon DM, O'Neal WK, Randell SH, Riordan JR, Gentzsch M
American journal of physiology. Lung cellular and molecular physiology 2010 Mar;298(3):L304-14
American journal of physiology. Lung cellular and molecular physiology 2010 Mar;298(3):L304-14
Differential functions of ApoER2 and very low density lipoprotein receptor in Reelin signaling depend on differential sorting of the receptors.
Duit S, Mayer H, Blake SM, Schneider WJ, Nimpf J
The Journal of biological chemistry 2010 Feb 12;285(7):4896-908
The Journal of biological chemistry 2010 Feb 12;285(7):4896-908
Assembly of connexin43 into gap junctions is regulated differentially by E-cadherin and N-cadherin in rat liver epithelial cells.
Govindarajan R, Chakraborty S, Johnson KE, Falk MM, Wheelock MJ, Johnson KR, Mehta PP
Molecular biology of the cell 2010 Dec;21(23):4089-107
Molecular biology of the cell 2010 Dec;21(23):4089-107
E-cadherin differentially regulates the assembly of Connexin43 and Connexin32 into gap junctions in human squamous carcinoma cells.
Chakraborty S, Mitra S, Falk MM, Caplan SH, Wheelock MJ, Johnson KR, Mehta PP
The Journal of biological chemistry 2010 Apr 2;285(14):10761-76
The Journal of biological chemistry 2010 Apr 2;285(14):10761-76
Urocortin trafficking in cerebral microvessel endothelial cells.
Tu H, Kastin AJ, Bjorbaek C, Pan W
Journal of molecular neuroscience : MN 2007;31(2):171-81
Journal of molecular neuroscience : MN 2007;31(2):171-81
Urocortin trafficking in cerebral microvessel endothelial cells.
Tu H, Kastin AJ, Bjorbaek C, Pan W
Journal of molecular neuroscience : MN 2007;31(2):171-81
Journal of molecular neuroscience : MN 2007;31(2):171-81
Misfolded BRICHOS SP-C mutant proteins induce apoptosis via caspase-4- and cytochrome c-related mechanisms.
Mulugeta S, Maguire JA, Newitt JL, Russo SJ, Kotorashvili A, Beers MF
American journal of physiology. Lung cellular and molecular physiology 2007 Sep;293(3):L720-9
American journal of physiology. Lung cellular and molecular physiology 2007 Sep;293(3):L720-9
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
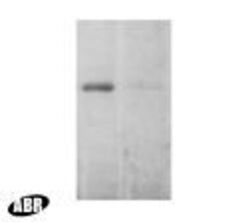
- Experimental details
- Western blot detection of EEA1 from mouse brain using Product # PA1-063A. Lane 2 shows incubation with the neutralizing peptide (PEP-079).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
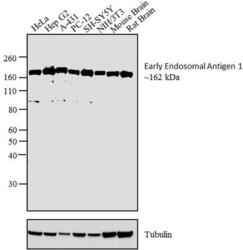
- Experimental details
- Western blot analysis was performed on whole cell extracts and tissue extracts of HeLa (Lane 1), Hep G2 (Lane 2) A-431 (Lane 3) PC-12 (Lane 4), SH-SY5Y (Lane 5), NIH/3T3 (Lane 6), Mouse brain tissue (Lane 7) and Rat brain tissue (Lane 8). The blot was probed with Anti- Early Endosomal Antigen 1 Rabbit Polyclonal Antibody (Product # PA1-063A, 2 µg/mL) and detected by chemiluminescence using Goat anti-Rabbit IgG (Heavy Chain) Superclonal™ Secondary Antibody, HRP conjugate (Product # A27036, 0.4 µg/mL, 1:2500 dilution. A ~ 162 kDa band corresponding to Early Endosomal Antigen 1 was observed across cell lines and tissues tested. Known quantity of protein samples were electrophoresed using Novex® NuPAGE® 4-12 % Bis-Tris gel (Product # NP0321BOX), XCell SureLock™ Electrophoresis System (Product # EI0002) and Novex® Sharp Pre-Stained Protein Standard (Product # LC5800). Resolved proteins were then transferred onto a nitrocellulose membrane by overnight transfer method. The membrane was probed with the relevant primary and secondary Antibody following blocking with 5 % skimmed milk. Chemiluminescent detection was performed using Pierce™ ECL Western Blotting Substrate (Product # 32106).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
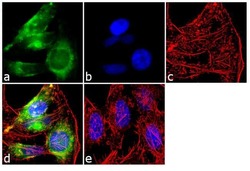
- Experimental details
- Immunofluorescent analysis of Early Endosomal Antigen 1 was performed using 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Early Endosomal Antigen 1 Rabbit Polyclonal Antibody (Product # PA1-063A) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoplasmic localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
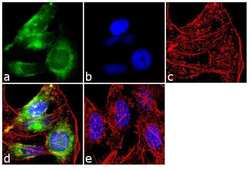
- Experimental details
- Immunofluorescent analysis of Early Endosomal Antigen 1 was performed using 70% confluent log phase HeLa cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 10 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with Early Endosomal Antigen 1 Rabbit Polyclonal Antibody (Product # PA1-063A) at 2 µg/mL in 0.1% BSA and incubated for 3 hours at room temperature and then labeled with Goat anti-Rabbit IgG (Heavy Chain) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Alexa Fluor® 555 Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing cytoplasmic localization. Panel e shows the no primary antibody control. The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
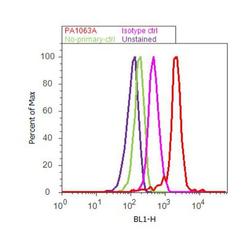
- Experimental details
- Flow cytometry analysis of Early Endosomal Antigen 1 was done on HeLa cells. Cells were fixed with 70% ethanol for 10 minutes, permeabilized with 0.25% Triton™ X-100 for 20 minutes, and blocked with 5% BSA for 30 minutes at room temperature. Cells were labeled with Early Endosomal Antigen 1 Rabbit Polyclonal Antibody (PA1-063A, red histogram) or with rabbit isotype control (pink histogram) at 3-5 ug/million cells in 2.5% BSA. After incubation at room temperature for 2 hours, the cells were labeled with Alexa Fluor® 488 Goat Anti-Rabbit Secondary Antibody (A11008) at a dilution of 1:400 for 30 minutes at room temperature. The representative 10, 000 cells were acquired and analyzed for each sample using an Attune® Acoustic Focusing Cytometer. The purple histogram represents unstained control cells and the green histogram represents no-primary-antibody control.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
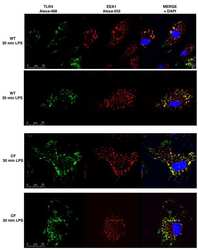
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
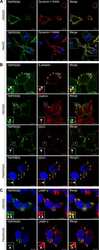
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
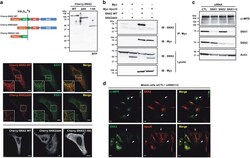
- Experimental details
- Figure 5 Cleavage of SNX2 disrupts interaction with Vps35. ( a ) HeLa cells were transfected for 16 h with plasmids encoding Cherry-fused full-length SNX2 (WT), C-terminal fragment (Delta84) or N-terminal fragment (1-84). Cherry-fused protein integrity was analyzed by immunoblotting using an RFP antibody (top). Colocalization with early endosomes was analyzed by confocal microscopy using SNX2, EEA1 early endosome marker and RFP antibodies (bottom). ( b ) HEK293T cells were transfected for 48 h with the indicated combination of empty (Myc), Myc-tagged Vps35, SNX2 WT and SNX2Delta84. Samples were immunoprecipitated (IP) using a Myc antibody and proteins were analyzed by immunoblotting (IB) with the indicated antibodies. ( c ) HeLa cells were transfected two times at a 24 h interval with the indicated combination of control (CTL), SNX1 and SNX2 small interfering RNAs (siRNAs), and proteins were analyzed after 72 h by immunoblotting using the indicated antibodies. Actin was used as a loading control. ( d ) Mixed populations of cells transfected as in ( c ) were analyzed for ci-MPR, SNX2 and Vps26 localization by confocal microscopy. Arrows point to SNX1/SNX2-depleted cells. Scale bars in ( a ) and ( d ) represent 10 mu m.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
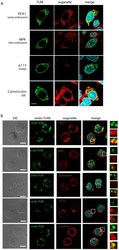
- Figure 1 Subcellular localization of TLR8 in human monocytes and HeLa transfectants. HeLa cells transiently expressing human TLR8 ( A ) and human monocytes ( B ) were incubated with anti-FLAG mAb ( A ) or anti-TLR8 mAb ( B ) followed by an Alexa Fluor 488-conjugated secondary Ab. Organelles were stained with an anti-EEA1 pAb, anti-MPR pAb, anti-p115 pAb, anti-calreticulin pAb, or anti-calnexin pAb followed by an Alexa Fluor 568-conjugated secondary Ab. Representative confocal images are shown. Green, TLR8; red, organelle markers; blue, nuclei stained with DAPI. Scale bar: ( A ) 10 um; ( B ) 5 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
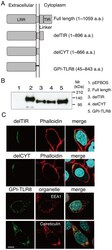
- Figure 2 Defining the TLR domain responsible for localization. A , Schematic diagram of the tail-deletion constructs of hTLR8. B , Expression of the TLR tail-deletion constructs in HEK293FT cells. Wild-type and mutant proteins transiently expressed in HEK293FT cells were immunoprecipitated with anti-FLAG mAb, resolved using SDS-PAGE, and detected using immunoblotting with anti-FLAG mAb. Molecular weight markers are shown on the right. C , Immunofluorescence images of the TLR8 deletion constructs in transfected HeLa cells. The upper and middle panels show cells stained with delTIR and delCYT together with phalloidin, which labels the plasma membrane. The lower two panels show cells stained with GPI-TLR8 together with EEA1 or calreticulin. Green, TLR8 mutants; red, organelles; blue, nuclei stained with DAPI; bar, 10 um.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
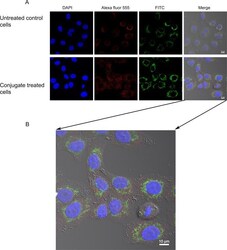
- Fig 5 Endosomal localization of Cytc-Tf conjugate in A549 cells using confocal imaging. (A) A549 cells were treated with the Cyt c-Tf conjugate for 12 h and endosome localization was probed with an endosomal marker antibody (primary antibody against EEA1 and a secondary antibody conjugated with Alexafluor-555) and Cyt c was localized with an anti-Cyt c FITC labeled antibody. The upper panel shows untreated cells and the lower panel the cells treated with Cyt c-Tf. (B) Merged image of A549 cells treated with conjugate enlarged to show nonover lapping green (Cyt c) and red (endosomes) signal.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
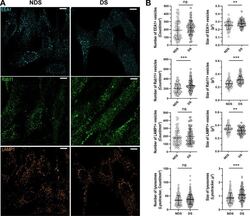
- Figure 3 Characterization of the compartments involved in the trafficking of IgG. ( A ) Representative confocal microscopy images showing the distribution of early endosomes (EEA1, cyan), recycling endosomes (Rab11, green), and lysosomes (LAMP1, orange) in diploid (NDS) and trisomic (DS) fibroblasts after incubation with IgG for 60 min at 37 degC. Scale bar: 10 um. ( B ) Number and size of EEA1 + endosomes (upper panel), Rab11 + recycling endosomes (upper middle panel), and lysosomes (LAMP1, Lysotracker) (lower middle and lower panels) in NDS (NDS-1, -2, and -3) and DS (DS-1, -2, and -3) cells. Each bar represents the mean +- SD. Each point represents measurements in individual cells (NDS, 72-296 cells/marker; DS, 82-250 cells cells/marker). **P < 0.01, ***P < 0.001, ns = not significant, Student's t test.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
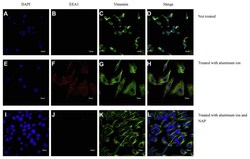
- Experimental details
- Analyses of representative samples of SH-SY5Y cells expression of DAPI, Vimentin and EEA1 were shown. Immunochemical stains for DAPI (blue), vimentin (green) and EEA1 (red) for SH-SY5Y cells cultured with aluminum ion and NAP for 24 h. (Scale bars, 10 mum, Confocal microscope, 400X).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
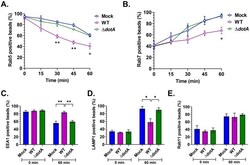
- Experimental details
- Fig 8 C . burnetii T4BSS alters latex bead phagosome maturation. Mock and C . burnetii -infected murine alveolar macrophages (2 dpi) were pulsed with 1 mum red fluorescent latex beads for 15 min, followed by fixation at 0 min post-pulse and every 15 min for 60 min. Cells were then immunostained for C . burnetii , plasma membrane, and Rab5 or Rab7 and internalized beads scored for co-localization with Rab5 or Rab7. The percent of Rab5 or Rab7 positive bead phagosomes over total number of internalized beads was plotted against time. In WT-infected cells, Rab5 was lost from bead phagosomes significantly faster than mock and DeltadotA -infected cells (A), whereas Rab7 localization to bead phagosomes was significantly slower in WT-infected cells (B). Further, bead phagosomes in mock, WT, and DeltadotA -infected cells were immunostained for EEA1, LAMP1, and Rab11 at 0 min and 60 min post-pulse. At 60 min post pulse, significantly more bead phagosomes retained EEA1, whereas WT-infected cells retained significantly more EEA1 compared to mock and DeltadotA (C), significantly fewer of those were positive for LAMP1 at 60 min post-pulse (D). There was no difference in Rab11 profile of bead phagosomes in between mock, WT, and DeltadotA -infected cells. Data shown as mean+-SEM of at least 30 bead phagosomes per condition in each of three independent experiments as analyzed by multiple t-test (A, B) or one-way ANOVA (C-E); **, P
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot