Antibody data
- Antibody Data
- Antigen structure
- References [4]
- Comments [0]
- Validations
- Western blot [1]
- Immunohistochemistry [1]
Submit
Validation data
Reference
Comment
Report error
- Product number
- NB600-1031 - Provider product page

- Provider
- Novus Biologicals
- Proper citation
- Novus Cat#NB600-1031, RRID:AB_2213184
- Product name
- Rabbit Polyclonal UNG Antibody
- Antibody type
- Polyclonal
- Description
- Protein G purified. The synthetic peptide sequence used for immunogen is 100% conserved between UNG1 and UNG2. Thus the antibody recognizes both UNG1 and UNG2. As UNG1 and UNG2 are not resolved as two species in gel electrophoresis (Huag et al., 1998), they are indistinguishable by western blot. The observed molecular weight of UNG (UNG1 and UNG2) on western blots may vary: 26 kDa, 27.5 kDa, 31 kDa, and 38 kDa forms have been described (Bharati et al., 1998
- Reactivity
- Human, Mouse, Rat, Rabbit
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 0.1 mg
- Concentration
- 0.5 mg/ml
- Storage
- Store at 4C short term. Aliquot and store at -20C long term. Avoid freeze-thaw cycles.
Submitted references Small interfering RNA-directed knockdown of uracil DNA glycosylase induces apoptosis and sensitizes human prostate cancer cells to genotoxic stress.
Known components of the immunoglobulin A:T mutational machinery are intact in Burkitt lymphoma cell lines with G:C bias.
Activation-induced deaminase cloning, localization, and protein extraction from young VH-mutant rabbit appendix.
Suppression of uracil-DNA glycosylase induces neuronal apoptosis.
Pulukuri SM, Knost JA, Estes N, Rao JS
Molecular cancer research : MCR 2009 Aug;7(8):1285-93
Molecular cancer research : MCR 2009 Aug;7(8):1285-93
Known components of the immunoglobulin A:T mutational machinery are intact in Burkitt lymphoma cell lines with G:C bias.
Xiao Z, Ray M, Jiang C, Clark AB, Rogozin IB, Diaz M
Molecular immunology 2007 Apr;44(10):2659-66
Molecular immunology 2007 Apr;44(10):2659-66
Activation-induced deaminase cloning, localization, and protein extraction from young VH-mutant rabbit appendix.
Yang G, Obiakor H, Sinha RK, Newman BA, Hood BL, Conrads TP, Veenstra TD, Mage RG
Proceedings of the National Academy of Sciences of the United States of America 2005 Nov 22;102(47):17083-8
Proceedings of the National Academy of Sciences of the United States of America 2005 Nov 22;102(47):17083-8
Suppression of uracil-DNA glycosylase induces neuronal apoptosis.
Kruman II, Schwartz E, Kruman Y, Cutler RG, Zhu X, Greig NH, Mattson MP
The Journal of biological chemistry 2004 Oct 15;279(42):43952-60
The Journal of biological chemistry 2004 Oct 15;279(42):43952-60
No comments: Submit comment
Supportive validation
- Submitted by
- Novus Biologicals (provider)
- Main image
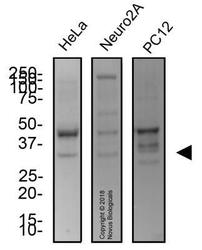
- Experimental details
- Western Blot: UNG Antibody [NB600-1031] - Total protein from human HeLa cells, muse Neuro2A cells and rat PC12 cells was separated on a 12% gel by SDS-PAGE, transferred to PVDF membrane and blocked in 5% non-fat milk in TBST. The membrane was probed with 2.0 ug/ml anti-UNG in 1% non-fat milk in TBST and detected with an anti-rabbit HRP secondary antibody using chemiluminescence.
Supportive validation
- Submitted by
- Novus Biologicals (provider)
- Main image
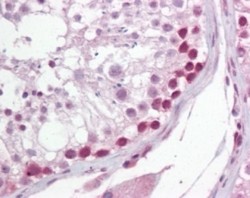
- Experimental details
- Immunohistochemistry-Paraffin: uracil-DNA glycosylase Antibody [NB600-1031] - Analysis of human testis using this antibody at 10 ug/ml.
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot Immunocytochemistry
Immunocytochemistry