Antibody data
- Antibody Data
- Antigen structure
- References [20]
- Comments [0]
- Validations
- Western blot [1]
- Immunocytochemistry [4]
- Immunohistochemistry [1]
- Other assay [9]
Submit
Validation data
Reference
Comment
Report error
- Product number
- MA1-2022 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- Phospho-Histone H2A.X (Ser139) Monoclonal Antibody (3F2)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Description
- MA1-2022 was used in IHC to successfully detect H2A.X pSer140 in postnatal mouse lung section.
- Antibody clone number
- 3F2
- Concentration
- 1 mg/mL
Submitted references Borna disease virus docks on neuronal DNA double-strand breaks to replicate and dampens neuronal activity.
CHK1 protects oncogenic KRAS-expressing cells from DNA damage and is a target for pancreatic cancer treatment.
Antibacterial and safety tests of a flexible cold atmospheric plasma device for the stimulation of wound healing.
Post-translational modification of RNA m6A demethylase ALKBH5 regulates ROS-induced DNA damage response.
Y(2)O(3) Nanoparticles and X-ray Radiation-Induced Effects in Melanoma Cells.
Regulation of meiotic progression by Sertoli-cell androgen signaling.
Safety and bactericidal efficacy of cold atmospheric plasma generated by a flexible surface Dielectric Barrier Discharge device against Pseudomonas aeruginosa in vitro and in vivo.
"Mitotic Slippage" and Extranuclear DNA in Cancer Chemoresistance: A Focus on Telomeres.
Quantification of DNA Damage in Heart Tissue as a Novel Prediction Tool for Therapeutic Prognosis of Patients With Dilated Cardiomyopathy.
ATM/G6PD-driven redox metabolism promotes FLT3 inhibitor resistance in acute myeloid leukemia.
High Content Screening Analysis to Evaluate the Toxicological Effects of Harmful and Potentially Harmful Constituents (HPHC).
Elevated Expression of Hepatoma Up-Regulated Protein Inhibits γ-Irradiation-Induced Apoptosis of Prostate Cancer Cells.
Conservative site-specific and single-copy transgenesis in human LINE-1 elements.
Organization and dynamics of the nonhomologous end-joining machinery during DNA double-strand break repair.
A high-throughput chemical screen reveals that harmine-mediated inhibition of DYRK1A increases human pancreatic beta cell replication.
Profiling dose-dependent activation of p53-mediated signaling pathways by chemicals with distinct mechanisms of DNA damage.
Comparative analysis of the cytotoxic effects of okadaic acid-group toxins on human intestinal cell lines.
The NR4A2 nuclear receptor is recruited to novel nuclear foci in response to UV irradiation and participates in nucleotide excision repair.
Cytotoxicity, fractionation and dereplication of extracts of the dinoflagellate Vulcanodinium rugosum, a producer of pinnatoxin G.
Development of a two-step chromatography procedure that allows the purification of a high-purity anti-histone H1 monoclonal immunoglobulin M antibody with immunosuppressant activity.
Marty FH, Bettamin L, Thouard A, Bourgade K, Allart S, Larrieu G, Malnou CE, Gonzalez-Dunia D, Suberbielle E
iScience 2022 Jan 21;25(1):103621
iScience 2022 Jan 21;25(1):103621
CHK1 protects oncogenic KRAS-expressing cells from DNA damage and is a target for pancreatic cancer treatment.
Klomp JE, Lee YS, Goodwin CM, Papke B, Klomp JA, Waters AM, Stalnecker CA, DeLiberty JM, Drizyte-Miller K, Yang R, Diehl JN, Yin HH, Pierobon M, Baldelli E, Ryan MB, Li S, Peterson J, Smith AR, Neal JT, McCormick AK, Kuo CJ, Counter CM, Petricoin EF 3rd, Cox AD, Bryant KL, Der CJ
Cell reports 2021 Nov 30;37(9):110060
Cell reports 2021 Nov 30;37(9):110060
Antibacterial and safety tests of a flexible cold atmospheric plasma device for the stimulation of wound healing.
Boekema B, Stoop M, Vlig M, van Liempt J, Sobota A, Ulrich M, Middelkoop E
Applied microbiology and biotechnology 2021 Mar;105(5):2057-2070
Applied microbiology and biotechnology 2021 Mar;105(5):2057-2070
Post-translational modification of RNA m6A demethylase ALKBH5 regulates ROS-induced DNA damage response.
Yu F, Wei J, Cui X, Yu C, Ni W, Bungert J, Wu L, He C, Qian Z
Nucleic acids research 2021 Jun 4;49(10):5779-5797
Nucleic acids research 2021 Jun 4;49(10):5779-5797
Y(2)O(3) Nanoparticles and X-ray Radiation-Induced Effects in Melanoma Cells.
Porosnicu I, Butnaru CM, Tiseanu I, Stancu E, Munteanu CVA, Bita BI, Duliu OG, Sima F
Molecules (Basel, Switzerland) 2021 Jun 4;26(11)
Molecules (Basel, Switzerland) 2021 Jun 4;26(11)
Regulation of meiotic progression by Sertoli-cell androgen signaling.
Larose H, Kent T, Ma Q, Shami AN, Harerimana N, Li JZ, Hammoud SS, Handel MA
Molecular biology of the cell 2020 Dec 1;31(25):2841-2862
Molecular biology of the cell 2020 Dec 1;31(25):2841-2862
Safety and bactericidal efficacy of cold atmospheric plasma generated by a flexible surface Dielectric Barrier Discharge device against Pseudomonas aeruginosa in vitro and in vivo.
Dijksteel GS, Ulrich MMW, Vlig M, Sobota A, Middelkoop E, Boekema BKHL
Annals of clinical microbiology and antimicrobials 2020 Aug 19;19(1):37
Annals of clinical microbiology and antimicrobials 2020 Aug 19;19(1):37
"Mitotic Slippage" and Extranuclear DNA in Cancer Chemoresistance: A Focus on Telomeres.
Salmina K, Bojko A, Inashkina I, Staniak K, Dudkowska M, Podlesniy P, Rumnieks F, Vainshelbaum NM, Pjanova D, Sikora E, Erenpreisa J
International journal of molecular sciences 2020 Apr 16;21(8)
International journal of molecular sciences 2020 Apr 16;21(8)
Quantification of DNA Damage in Heart Tissue as a Novel Prediction Tool for Therapeutic Prognosis of Patients With Dilated Cardiomyopathy.
Ko T, Fujita K, Nomura S, Uemura Y, Yamada S, Tobita T, Katoh M, Satoh M, Ito M, Domoto Y, Hosoya Y, Amiya E, Hatano M, Morita H, Fukayama M, Aburatani H, Komuro I
JACC. Basic to translational science 2019 Oct;4(6):670-680
JACC. Basic to translational science 2019 Oct;4(6):670-680
ATM/G6PD-driven redox metabolism promotes FLT3 inhibitor resistance in acute myeloid leukemia.
Gregory MA, D'Alessandro A, Alvarez-Calderon F, Kim J, Nemkov T, Adane B, Rozhok AI, Kumar A, Kumar V, Pollyea DA, Wempe MF, Jordan CT, Serkova NJ, Tan AC, Hansen KC, DeGregori J
Proceedings of the National Academy of Sciences of the United States of America 2016 Oct 25;113(43):E6669-E6678
Proceedings of the National Academy of Sciences of the United States of America 2016 Oct 25;113(43):E6669-E6678
High Content Screening Analysis to Evaluate the Toxicological Effects of Harmful and Potentially Harmful Constituents (HPHC).
Marescotti D, Gonzalez Suarez I, Acali S, Johne S, Laurent A, Frentzel S, Hoeng J, Peitsch MC
Journal of visualized experiments : JoVE 2016 May 10;(111)
Journal of visualized experiments : JoVE 2016 May 10;(111)
Elevated Expression of Hepatoma Up-Regulated Protein Inhibits γ-Irradiation-Induced Apoptosis of Prostate Cancer Cells.
Hassan M, El Khattouti A, Ejaeidi A, Ma T, Day WA, Espinoza I, Vijayakumar S, Gomez CR
Journal of cellular biochemistry 2016 Jun;117(6):1308-18
Journal of cellular biochemistry 2016 Jun;117(6):1308-18
Conservative site-specific and single-copy transgenesis in human LINE-1 elements.
Vijaya Chandra SH, Makhija H, Peter S, Myint Wai CM, Li J, Zhu J, Ren Z, D'Alcontres MS, Siau JW, Chee S, Ghadessy FJ, Dröge P
Nucleic acids research 2016 Apr 7;44(6):e55
Nucleic acids research 2016 Apr 7;44(6):e55
Organization and dynamics of the nonhomologous end-joining machinery during DNA double-strand break repair.
Reid DA, Keegan S, Leo-Macias A, Watanabe G, Strande NT, Chang HH, Oksuz BA, Fenyo D, Lieber MR, Ramsden DA, Rothenberg E
Proceedings of the National Academy of Sciences of the United States of America 2015 May 19;112(20):E2575-84
Proceedings of the National Academy of Sciences of the United States of America 2015 May 19;112(20):E2575-84
A high-throughput chemical screen reveals that harmine-mediated inhibition of DYRK1A increases human pancreatic beta cell replication.
Wang P, Alvarez-Perez JC, Felsenfeld DP, Liu H, Sivendran S, Bender A, Kumar A, Sanchez R, Scott DK, Garcia-Ocaña A, Stewart AF
Nature medicine 2015 Apr;21(4):383-8
Nature medicine 2015 Apr;21(4):383-8
Profiling dose-dependent activation of p53-mediated signaling pathways by chemicals with distinct mechanisms of DNA damage.
Clewell RA, Sun B, Adeleye Y, Carmichael P, Efremenko A, McMullen PD, Pendse S, Trask OJ, White A, Andersen ME
Toxicological sciences : an official journal of the Society of Toxicology 2014 Nov;142(1):56-73
Toxicological sciences : an official journal of the Society of Toxicology 2014 Nov;142(1):56-73
Comparative analysis of the cytotoxic effects of okadaic acid-group toxins on human intestinal cell lines.
Ferron PJ, Hogeveen K, Fessard V, Le Hégarat L
Marine drugs 2014 Aug 21;12(8):4616-34
Marine drugs 2014 Aug 21;12(8):4616-34
The NR4A2 nuclear receptor is recruited to novel nuclear foci in response to UV irradiation and participates in nucleotide excision repair.
Jagirdar K, Yin K, Harrison M, Lim W, Muscat GE, Sturm RA, Smith AG
PloS one 2013;8(11):e78075
PloS one 2013;8(11):e78075
Cytotoxicity, fractionation and dereplication of extracts of the dinoflagellate Vulcanodinium rugosum, a producer of pinnatoxin G.
Geiger M, Desanglois G, Hogeveen K, Fessard V, Leprêtre T, Mondeguer F, Guitton Y, Hervé F, Séchet V, Grovel O, Pouchus YF, Hess P
Marine drugs 2013 Sep 2;11(9):3350-71
Marine drugs 2013 Sep 2;11(9):3350-71
Development of a two-step chromatography procedure that allows the purification of a high-purity anti-histone H1 monoclonal immunoglobulin M antibody with immunosuppressant activity.
Shimada Y, Goto T, Kawamoto S, Kiso T, Katayama A, Yamanaka Y, Aki T, Chiang KC, Nakano T, Goto S, Chen CL, Ohmori N, Ono K, Sato S
Biomedical chromatography : BMC 2008 Jan;22(1):13-9
Biomedical chromatography : BMC 2008 Jan;22(1):13-9
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
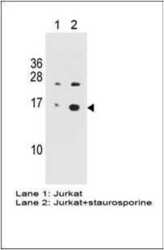
- Experimental details
- Western blot analysis of Phospho-H2AX pSer140 (Product # MA1-2022) at a concentration of 1 µg/mL on Jurkat cell untreated (Lane 1) and Jurkat cell stimulated with staurosporine (Lane 2) followed by HRP conjugated goat anti-mouse lgG (H+L) Secondary Antibody.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of Phospho-Histone H2A.X (Ser140) using a monoclonal antibody (Product # MA1-2022).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
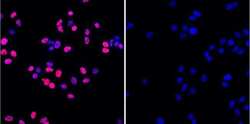
- Experimental details
- Immunofluorescence staining of Phospho-H2AX in A549 cells. Cells were treated with vehicle (control; 0.1% DMSO in media) or with 50 µM etoposide for 1 hour
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
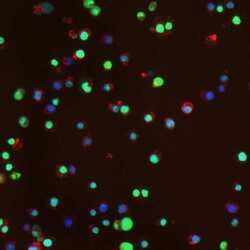
- Experimental details
- Immunofluorescence staining using Product # MA1-2022, treatment with paraquat and iron induces MnSOD and Phosphorylation of H2AX in RAW 264.7 macrophages. Cells were treated for 20 hours with paraquat (500 µM) and iron (200 µg/mL) and stained with anti-Phospho-H2AX antibody.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescence image of phosphorylated H2AX in gamma irradiated HeLa cells using Product # MA1-2022.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
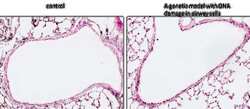
- Experimental details
- Immunohistochemistry analysis of phospho-Histone H2A.X pSer140 in postnatal mouse (PN19) lung sections. Antigen retrieval from 4% PFA, paraffin embedded sections was performed using heat induced epitope retrieval (HIER) method with sodium citrate buffer (pH 6.0). Following antigen retrieval, tissues were blocked and probed with a phospho-Histone H2A.X pSer140 mouse monoclonal antibody (Product # MA1-2022) at a dilution of 1:400. Increased staining intensity was observed in a genetic mouse model with DNA damage in airway cells.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
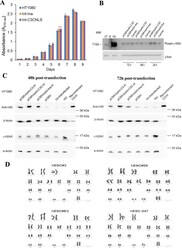
- Figure 5. Int-C3CNLS does not induce DNA damage or cytotoxicity. ( A ) MTT-based cell proliferation assays were performed to assess effects on cell proliferation rates upon Int expression in human cell lines. HT1080 cells untransfected (HT1080), and FACS sorted GFP + cells obtained after co-transfection of pCMV-EGFP with either pCMVssInt-Ina (INA; expressing inactive integrase) or pCMVssInt-C3CNLS (C3CNLS) were analyzed for the effect on cellular proliferation using the colorimetric MTT assays over the indicated time course. Data show the mean of triplicates and standard deviation of a representative experiment. n = 2. ( B ) Western blot analysis to determine phospho-gamma-H2AX levels to assess DNA damage induced by expression of Int in A549 cells. Cell lysates prepared at time points of 24, 48 and 72 h (post transfection) from A549 cells transfected with pCMVssInt-Ina or pCMVssInt-C3C (expressing Int-C3CNLS) and from control cells treated with the carrier (Lipofectamine2000 Transfection reagent) were subjected to western blot analysis using antibodies against phospho-gamma-H2AX (top panel). UT, untreated cells as negative control; HU, cells treated with hydroxy urea (10 mM for 24 h) as positive control; M, Marker lane. beta-actin was used as loading control (bottom panel). ( C ) Western blot analysis to determine phospho-gamma-H2AX levels to assess DNA damage induced by expression of Int in HT1080 cells. Forty-eight hours post transfection, top and 72 h post transfection, bo
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
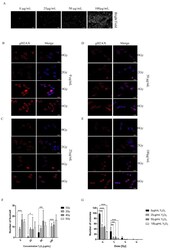
- Figure 6 Induction of active caspase-3 and/or gammaH2AX by OA, DTX-1 and -2 in Caco-2 and HT29-MTX cells. Representative images at 10x magnification of Caco-2 and HT29-MTX cells treated 24 h with 150 nM OA ( A ); DAPI staining in blue corresponds to the nucleus. Caco-2 and HT29-MTX cells were treated with increasing concentrations of OA, DTX-1 and -2 for 24 h and were then labeled for detection of active caspase-3 (green) and gammaH2AX (red) by high content analysis ( B ). The percentage of positive cells for active caspase-3 and gammaH2AX was determined using a threshold based on control conditions. Data are expressed as stacked bars representing unlabeled cells (white), active caspase-3-positive cells (green), gammaH2AX-positive (red) and positive for both active caspase-3 and gammaH2AX (orange). n = 3.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- (A) The Western blot analysis demonstrates the rescue of gamma-irradiation-induced expression and phosphorylation of ATM, the phosphorylation of H2AX, the expression of p53, Noxa and E2F1 as well as the phosphorylation of CHK1 and 2 in response to the induction of HURP expression by the addition of Dox to the culture media of LNCaP-TetOn-HURP cells. Actin was used internal control for loading and transfer. EMSA demonstrates the inhibition of gamma-irradiation-induced DNA binding activities of the transcription factors E2F1 (B), p53 (C,D) EMSA demonstrates the rescue of gamma-irradiation-induced DNA- binding activity of the transcription factor p53 in response to the induction of HURP expression by the addition of Doxycycline to the culture media of DU145-TetOn-HURP cells. (E) EMSA analysis demonstrated the ability of HURP to inhibit gamma-irradiation -induced DNA binding activity of the transcription factor p53 in LNCap and DU145 cells, whereas in p53- PC3 cells is not detectable compared to positive control. The data are representative of three independent experiments. (F,G) Immune fluorescence demonstrates the inhibition of gamma-irradiation-induced H2AX (red) in response to the induction of HURP expression (green) by the addition of Dox to the culture media of LNCaP-TetOn-HURP cells. The nucleus is stained with DAPI.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- Figure 5 DNA damage induced by X-ray radiation. ( A ) Bright field image depicting the uptake of different concentrations of NPs ( B - E ) A375 cells treated with different concentrations of NPs and irradiated with 0, 2, 4, 6 Gy and stained for gamma-H2AX. ( F ) Quantification of number of foci/cell using ImageJ software. ( G ) Clonogenic assay of samples treated with 0, 25, 50, 100 mug/mL Y 2 O 3 and exposed to 0, 2, 4, 6 Gy doses of radiation (* p < 0.05, *** p < 0.005, **** p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
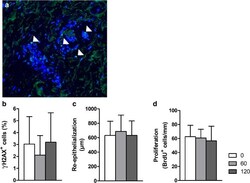
- Fig. 4 a Example of dermal sample after plasma treatment, stained for gammaH2AX. Positive nuclei are indicated (arrow head); bar is 50 mum. b Quantification of the relative number of cells (%) positive for gammaH2AX after plasma treatment of dermal tissue ( n =4-5 in duplicate). c Re-epithelialization and d proliferation of keratinocytes in BWM after 2 weeks of culture. Plasma was applied four times in 10 days. Mean values and standard deviation are shown ( n =6 for 0 and 120 s, n =4 for 60 s, all in duplicate). Significant differences were not observed
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
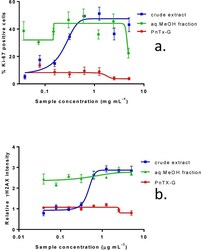
- Figure 3 Effect of crude extract of V. rugosum , the aqueous methanol fraction and PnTX-G on ( a ) Ki-67 expression and on ( b ) gammaH2AX phosphorylation in Caco-2 cells. Data are presented as the mean average nuclear intensity of Ki-67 and gammaH2AX immunofluorescence +- SEM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
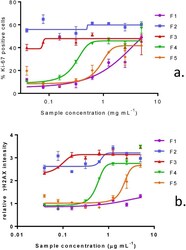
- Figure 5 Effect of purified fractions of V. rugosum on Ki-67 expression ( a ) and on gammaH2AX phosphorylation ( b ) in Caco-2 cells. Data are presented as the mean average nuclear intensity of Ki-67 and gammaH2AX immunofluorescence +- SEM.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Representative images of ( a ) gammaH2AX immunofluorescence in untreated Caco-2 cells; ( b ) Caco-2 cells treated with F3 at 156 ng mL -1 ; ( c ) Ki-67 immunofluorescence in untreated Caco-2 cells; ( d ) Caco-2 cells treated with F3 at 156 ng mL -1 .
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot