14-7379-82
antibody from Invitrogen Antibodies
Targeting: IL37
FIL1, FIL1(ZETA), FIL1Z, IL-1F7, IL-1H4, IL-1RP1, IL1F7
Antibody data
- Antibody Data
- Antigen structure
- References [4]
- Comments [0]
- Validations
- Immunocytochemistry [1]
- Other assay [2]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 14-7379-82 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- IL-37 Monoclonal Antibody (37D12), eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: This 37D12 monoclonal antibody reacts with human IL-37. IL-37 is a member of the interleukin 1 cytokine family and is also known as IL-1F7 and IL-1H4. IL-37 suppresses innate inflammatory and immune responses and exhibits anti-inflammatory characteristics resembling those of IL-1R8. IL-37 binds to the alpha chain of IL-18R with low affinity and does not recruit the essential signaling co-receptor IL-18 beta or mediate pro-inflammatory activity. IL-37 also binds to IL-18 binding protein (IL-18BP) and enhances the antagonistic effects of IL-18BP. IL-37 forms a trimeric complex with IL-18BP and IL-18R beta, which results in the blockade of IL-18 activity. Transcripts of IL-37 have been detected in human tissues, such as lung, testis, and colon tumors, as well as some human cell lines. Applications Reported: This 37D12 antibody has been reported for use in immunocytochemistry followed by cytospin. Applications Tested: This 37D12 antibody has been tested by immunocytochemistry of fixed and permeabilized normal human peripheral blood mononuclear cells and can be used at less than or equal to 20 µg/mL. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest. Purity: Greater than 90%, as determined by SDS-PAGE. Aggregation: Less than 10%, as determined by HPLC. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 37D12
- Vial size
- 100 µg
- Concentration
- 0.5 mg/mL
- Storage
- 4° C
Submitted references Characterization of the pathoimmunology of necrotizing enterocolitis reveals novel therapeutic opportunities.
IL-37 requires the receptors IL-18Rα and IL-1R8 (SIGIRR) to carry out its multifaceted anti-inflammatory program upon innate signal transduction.
A complex of the IL-1 homologue IL-1F7b and IL-18-binding protein reduces IL-18 activity.
Interleukin-1F7B (IL-1H4/IL-1F7) is processed by caspase-1 and mature IL-1F7B binds to the IL-18 receptor but does not induce IFN-gamma production.
Cho SX, Rudloff I, Lao JC, Pang MA, Goldberg R, Bui CB, McLean CA, Stock M, Klassert TE, Slevogt H, Mangan NE, Cheng W, Fischer D, Gfroerer S, Sandhu MK, Ngo D, Bujotzek A, Lariviere L, Schumacher F, Tiefenthaler G, Beker F, Collins C, Kamlin COF, König K, Malhotra A, Tan K, Theda C, Veldman A, Ellisdon AM, Whisstock JC, Berger PJ, Nold-Petry CA, Nold MF
Nature communications 2020 Nov 13;11(1):5794
Nature communications 2020 Nov 13;11(1):5794
IL-37 requires the receptors IL-18Rα and IL-1R8 (SIGIRR) to carry out its multifaceted anti-inflammatory program upon innate signal transduction.
Nold-Petry CA, Lo CY, Rudloff I, Elgass KD, Li S, Gantier MP, Lotz-Havla AS, Gersting SW, Cho SX, Lao JC, Ellisdon AM, Rotter B, Azam T, Mangan NE, Rossello FJ, Whisstock JC, Bufler P, Garlanda C, Mantovani A, Dinarello CA, Nold MF
Nature immunology 2015 Apr;16(4):354-65
Nature immunology 2015 Apr;16(4):354-65
A complex of the IL-1 homologue IL-1F7b and IL-18-binding protein reduces IL-18 activity.
Bufler P, Azam T, Gamboni-Robertson F, Reznikov LL, Kumar S, Dinarello CA, Kim SH
Proceedings of the National Academy of Sciences of the United States of America 2002 Oct 15;99(21):13723-8
Proceedings of the National Academy of Sciences of the United States of America 2002 Oct 15;99(21):13723-8
Interleukin-1F7B (IL-1H4/IL-1F7) is processed by caspase-1 and mature IL-1F7B binds to the IL-18 receptor but does not induce IFN-gamma production.
Kumar S, Hanning CR, Brigham-Burke MR, Rieman DJ, Lehr R, Khandekar S, Kirkpatrick RB, Scott GF, Lee JC, Lynch FJ, Gao W, Gambotto A, Lotze MT
Cytokine 2002 Apr 21;18(2):61-71
Cytokine 2002 Apr 21;18(2):61-71
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunocytochemistry of fixed and permeabilized human PBMC (stimulated with LPS and monensin for 5 hours) stained with 20 µg/mL of Mouse IgG1 K Isotype Control Purified (left) or 20 µg/mL of Anti-Human IL-37 (IL-1F7) Purified.Cells were then stained with F (ab')2 Anti-Mouse IgG eFluor® 570 followed by cytospin.Nuclei are stained with DAPI.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
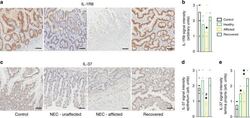
- Fig. 7 Intestinal IL-37 and IL-1R8 in human NEC infants. Immunohistochemical assessment of IL-1R8 ( a , b ) and IL-37 ( c - e ) in samples from the same infants, namely non-NEC controls (control, n = 9), largely healthy NEC tissue (unaffected, n = 3), NEC-afflicted tissue (afflicted, n = 6) and samples from intestines after NEC recovery (recovered, n = 2). ""Afflicted"" also comprises the necrotic group in the immunohistochemistry experiments. a , c One representative image for each group is shown. Scale bars: 50 um. b , d , e Quantification of the signal intensity of IL-1R8 ( b ) or IL-37 in the epithelium ( d ) or the lamina propria ( e ). Bars plotted indicate means of individual signal intensity values, which are shown as dots. One-way ANOVA P -values: * P < 0.05 and ** P < 0.01 for healthy or afflicted acute NEC compared to non-NEC controls. # P < 0.05 for healthy vs afflicted sections in acute NEC.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
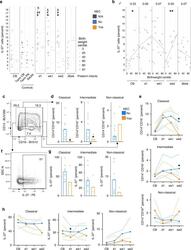
- Fig. 8 IL-37 abundance in blood cells from human NEC infants. Peripheral blood was obtained from the second cohort of premature infants (gestational age 24-29 weeks, n = 21; note that some time points are not available for some preterm infants; for exact n at each time point see dots in panels and Source Data file) at the indicated time points, from healthy term infants at birth ( n = 17) and at 4-16 weeks of age ( n = 10), as well as from healthy adult volunteers ( n = 5). The percentage of IL-37 + leukocytes among viable CD45 + cells was determined by flow cytometry. CB cord blood, Term healthy term infants, PN postnatal, d1 day 1, wk1/2 week 1/2, 36wk 36 weeks of corrected gestational age. a , b IL-37 + percentages are graphed for each individual infant (circles) with medians indicated by gray lines. Student''s t -test P- values: * P < 0.05 for indicated preterm time point vs preterm CB. # P < 0.05 and ## P < 0.01 for indicated preterm time point vs term PN. & P < 0.05 for indicated preterm time point vs adults. b Correlation of birthweight centile with IL-37 + percentage at the indicated time points. Pearson correlation P- values: * P < 0.05; ** P < 0.01. c - h Analysis of monocyte subtypes and their IL-37 + percentages at the week 2 time point ( c , d , f , g ) or longitudinally ( e , h ). Mann-Whitney U test P- values: * P < 0.05 for NEC vs non-NEC. c - e Quantification of monocyte subtypes under viable CD45 + cells, and f - h of IL-37 + percentages under the indicated
 Explore
Explore Validate
Validate Learn
Learn Immunocytochemistry
Immunocytochemistry