Antibody data
- Antibody Data
- Antigen structure
- References [18]
- Comments [0]
- Validations
- Other assay [22]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 37-6600 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- FBXO5 Monoclonal Antibody (3D2D6)
- Antibody type
- Monoclonal
- Antigen
- Synthetic peptide
- Reactivity
- Human
- Host
- Mouse
- Isotype
- IgG
- Antibody clone number
- 3D2D6
- Vial size
- 100 μg
- Concentration
- 0.5 mg/mL
- Storage
- -20°C
Submitted references Altered G1 signaling order and commitment point in cells proliferating without CDK4/6 activity.
Kaposi's Sarcoma-Associated Herpesvirus Lytic Replication Is Independent of Anaphase-Promoting Complex Activity.
Excessive E2F Transcription in Single Cancer Cells Precludes Transient Cell-Cycle Exit after DNA Damage.
Oocytes mount a noncanonical DNA damage response involving APC-Cdh1-mediated proteolysis.
EMI1 switches from being a substrate to an inhibitor of APC/C(CDH1) to start the cell cycle.
Progression through mitosis promotes PARP inhibitor-induced cytotoxicity in homologous recombination-deficient cancer cells.
Feedback regulation between atypical E2Fs and APC/CCdh1 coordinates cell cycle progression.
New origin firing is inhibited by APC/CCdh1 activation in S-phase after severe replication stress.
Hypersensitivity to DNA damage in antephase as a safeguard for genome stability.
MASTL promotes cyclin B1 destruction by enforcing Cdc20-independent binding of cyclin B1 to the APC/C.
Centriole maturation requires regulated Plk1 activity during two consecutive cell cycles.
Human papillomavirus type 16 E7 oncoprotein inhibits the anaphase promoting complex/cyclosome activity by dysregulating EMI1 expression in mitosis.
Selective enhancing effect of early mitotic inhibitor 1 (Emi1) depletion on the sensitivity of doxorubicin or X-ray treatment in human cancer cells.
The anaphase-promoting complex or cyclosome supports cell survival in response to endoplasmic reticulum stress.
Regulation of the action of early mitotic inhibitor 1 on the anaphase-promoting complex/cyclosome by cyclin-dependent kinases.
Substrate degradation by the anaphase promoting complex occurs during mitotic slippage.
Regulation of APC/C (Cdh1) ubiquitin ligase in differentiation of human embryonic stem cells.
DNA damage triggers p21WAF1-dependent Emi1 down-regulation that maintains G2 arrest.
Liu C, Konagaya Y, Chung M, Daigh LH, Fan Y, Yang HW, Terai K, Matsuda M, Meyer T
Nature communications 2020 Oct 20;11(1):5305
Nature communications 2020 Oct 20;11(1):5305
Kaposi's Sarcoma-Associated Herpesvirus Lytic Replication Is Independent of Anaphase-Promoting Complex Activity.
Elbasani E, Gramolelli S, Günther T, Gabaev I, Grundhoff A, Ojala PM
Journal of virology 2020 Jun 16;94(13)
Journal of virology 2020 Jun 16;94(13)
Excessive E2F Transcription in Single Cancer Cells Precludes Transient Cell-Cycle Exit after DNA Damage.
Segeren HA, van Rijnberk LM, Moreno E, Riemers FM, van Liere EA, Yuan R, Wubbolts R, de Bruin A, Westendorp B
Cell reports 2020 Dec 1;33(9):108449
Cell reports 2020 Dec 1;33(9):108449
Oocytes mount a noncanonical DNA damage response involving APC-Cdh1-mediated proteolysis.
Subramanian GN, Greaney J, Wei Z, Becherel O, Lavin M, Homer HA
The Journal of cell biology 2020 Apr 6;219(4)
The Journal of cell biology 2020 Apr 6;219(4)
EMI1 switches from being a substrate to an inhibitor of APC/C(CDH1) to start the cell cycle.
Cappell SD, Mark KG, Garbett D, Pack LR, Rape M, Meyer T
Nature 2018 Jun;558(7709):313-317
Nature 2018 Jun;558(7709):313-317
Progression through mitosis promotes PARP inhibitor-induced cytotoxicity in homologous recombination-deficient cancer cells.
Schoonen PM, Talens F, Stok C, Gogola E, Heijink AM, Bouwman P, Foijer F, Tarsounas M, Blatter S, Jonkers J, Rottenberg S, van Vugt MATM
Nature communications 2017 Jul 17;8:15981
Nature communications 2017 Jul 17;8:15981
Feedback regulation between atypical E2Fs and APC/CCdh1 coordinates cell cycle progression.
Boekhout M, Yuan R, Wondergem AP, Segeren HA, van Liere EA, Awol N, Jansen I, Wolthuis RM, de Bruin A, Westendorp B
EMBO reports 2016 Mar;17(3):414-27
EMBO reports 2016 Mar;17(3):414-27
New origin firing is inhibited by APC/CCdh1 activation in S-phase after severe replication stress.
Ercilla A, Llopis A, Feu S, Aranda S, Ernfors P, Freire R, Agell N
Nucleic acids research 2016 Jun 2;44(10):4745-62
Nucleic acids research 2016 Jun 2;44(10):4745-62
Hypersensitivity to DNA damage in antephase as a safeguard for genome stability.
Feringa FM, Krenning L, Koch A, van den Berg J, van den Broek B, Jalink K, Medema RH
Nature communications 2016 Aug 26;7:12618
Nature communications 2016 Aug 26;7:12618
MASTL promotes cyclin B1 destruction by enforcing Cdc20-independent binding of cyclin B1 to the APC/C.
Voets E, Wolthuis R
Biology open 2015 Mar 6;4(4):484-95
Biology open 2015 Mar 6;4(4):484-95
Centriole maturation requires regulated Plk1 activity during two consecutive cell cycles.
Kong D, Farmer V, Shukla A, James J, Gruskin R, Kiriyama S, Loncarek J
The Journal of cell biology 2014 Sep 29;206(7):855-65
The Journal of cell biology 2014 Sep 29;206(7):855-65
Human papillomavirus type 16 E7 oncoprotein inhibits the anaphase promoting complex/cyclosome activity by dysregulating EMI1 expression in mitosis.
Yu Y, Munger K
Virology 2013 Nov;446(1-2):251-9
Virology 2013 Nov;446(1-2):251-9
Selective enhancing effect of early mitotic inhibitor 1 (Emi1) depletion on the sensitivity of doxorubicin or X-ray treatment in human cancer cells.
Shimizu N, Nakajima NI, Tsunematsu T, Ogawa I, Kawai H, Hirayama R, Fujimori A, Yamada A, Okayasu R, Ishimaru N, Takata T, Kudo Y
The Journal of biological chemistry 2013 Jun 14;288(24):17238-52
The Journal of biological chemistry 2013 Jun 14;288(24):17238-52
The anaphase-promoting complex or cyclosome supports cell survival in response to endoplasmic reticulum stress.
Chen M, Gutierrez GJ, Ronai ZA
PloS one 2012;7(4):e35520
PloS one 2012;7(4):e35520
Regulation of the action of early mitotic inhibitor 1 on the anaphase-promoting complex/cyclosome by cyclin-dependent kinases.
Moshe Y, Bar-On O, Ganoth D, Hershko A
The Journal of biological chemistry 2011 May 13;286(19):16647-57
The Journal of biological chemistry 2011 May 13;286(19):16647-57
Substrate degradation by the anaphase promoting complex occurs during mitotic slippage.
Lee J, Kim JA, Margolis RL, Fotedar R
Cell cycle (Georgetown, Tex.) 2010 May;9(9):1792-801
Cell cycle (Georgetown, Tex.) 2010 May;9(9):1792-801
Regulation of APC/C (Cdh1) ubiquitin ligase in differentiation of human embryonic stem cells.
Bar-On O, Shapira M, Skorecki K, Hershko A, Hershko DD
Cell cycle (Georgetown, Tex.) 2010 May 15;9(10):1986-9
Cell cycle (Georgetown, Tex.) 2010 May 15;9(10):1986-9
DNA damage triggers p21WAF1-dependent Emi1 down-regulation that maintains G2 arrest.
Lee J, Kim JA, Barbier V, Fotedar A, Fotedar R
Molecular biology of the cell 2009 Apr;20(7):1891-902
Molecular biology of the cell 2009 Apr;20(7):1891-902
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
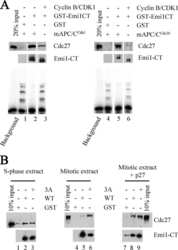
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
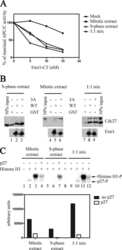
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
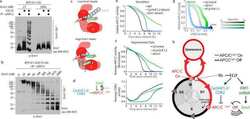
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
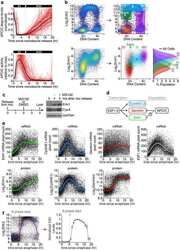
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
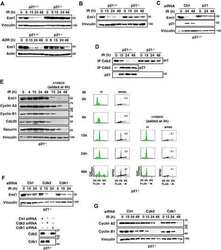
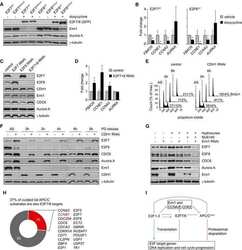
- Fig. 3. MASTL depletion significantly impairs cyclin B1 recruitment to the mitotic APC/C. (A) Cyclin B1 binds to the APC/C specifically in mitosis. HeLa cells were synchronised in G2 and M phase by a thymidine block, followed by nocodazole or taxol treatment. Cells arrested in mitosis were collected by mitotic shake-off, and treated for another 2 hours with 5 uM MG132 where indicated. Cell extracts were subjected to immunoprecipitation using a goat anti-APC4. Aliquots of the immunoprecipitates were analysed by western blotting. (B) MASTL silencing impairs the recruitment of cyclin A2 and cyclin B1 to the mitotic APC/C. HeLa cells transfected with indicated siRNAs were synchronised in G2 phase and mitosis using thymidine and taxol. Cells arrested in mitosis were obtained by mitotic shake-off and lysed directly or subsequently treated with 10 uM of RO-3306 for 90 minutes to obtain G1 phase cells. Cell extracts were subjected to immunoprecipitation using a goat anti-APC4 and aliquots of the immunoprecipitates were analysed by western blotting. Note that equal amounts of APC4 are immunoprecipitated in the mitotic fractions, whereas cyclins A2 and B1 bound to the APC/C are hardly detectable in the MASTL RNAi extracts. The asterisk (*) indicates the remaining securin signal after reprobing the blot with anti-geminin (right-hand panel). (C) Direct binding of cyclin B1 to the APC/C may depend on extensive APC3 phosphorylation in mitosis. HeLa cells treated as in (B), were subjected t
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
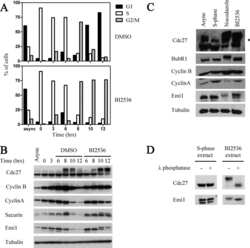
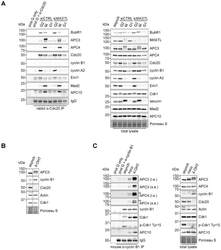
- Fig. 4. Cyclin B1 associates with the mitotic APC/C, independent of Cdc20. (A) Cyclin B1 recruitment to the APC/C does not involve direct Cdc20 binding, different from cyclin A2. HeLa cells transfected with indicated siRNAs were synchronised in G2 phase and mitosis using thymidine and taxol. Cells arrested in mitosis were obtained by mitotic shake-off and lysed directly or subsequently treated with 10 uM of RO-3306 for 90 minutes to obtain G1 phase cells. Cell extracts were subjected to immunoprecipitation using a rabbit anti-Cdc20 and aliquots of the immunoprecipitates were analysed by western blotting. Asynch refers to an asynchronous, non-transfected lysate. (B) Genetic ablation of CDC20 results in a mitotic arrest due to cyclin B1 stabilisation. Cdc20(Delta/Delta); RERT(+/Cre) mouse embryonic fibroblasts (MEFs) were asynchronously grown or thymidine synchronised for 9 hours and treated with 1 uM of 4-hydroxytamoxifen (4-OHT) to induce Cre activity, resulting in the excision of exon 2 of CDC20 (Cdc20(Delta) allele). Cells arrested in mitosis by the 4-OHT treatment were obtained by mitotic shake-off. Extracts of asynchronous or 4-OHT treated cells were subjected to western blot analysis. The dashed line indicates where two lanes have been placed next to each other. (C) Cyclin B1 binds the mitotic APC/C regardless of the presence of Cdc20. Cdc20(Delta/Delta); RERT(+/Cre) MEFs were either asynchronously grown or synchronised in mitosis using thymidine and nocodazole or 4-OHT
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
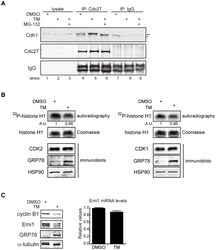
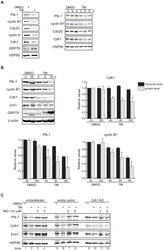
- Figure 3 Mechanisms supporting APC/C Cdh1 activity under ER stress conditions. (A) HeLa cells were treated with DMSO, 0.5 ug/ml of TM alone, or 0.5 ug/ml of TM plus 5 uM of MG-132 for 16 h. Immunoprecipitates of endogenous Cdc27 were immunoblotted for endogenous Cdh1. Immunoprecipitation using IgG served as negative control. ** indicates Cdh1-specific top band; *indicates non-specific bottom band. (B) HeLa cells were treated with DMSO or 1 ug/ml of TM for 16 h. CDK2 or CDK1 antibodies were used to immunoprecipitate endogenous CDK2 or CDK1 complexes, respectively. Immunoprecipitates were then used in in vitro kinase assays using histone H1 as substrate. Left , autoradiography of 32 P-histone H1 phosphorylated by CDK2 complexes. Coomassie stains input of histone H1 in the reactions. Intensity of the autoradioactive bands were quantified by Image J, normalized to histone H1 input, and presented as arbitrary units (A.U.). Immunoblot shows comparable levels of endogenous CDK2 and the indicated proteins before and after TM treatment. Right , autoradiography of 32 P-histone H1 phosphorylated by CDK1 complexes, analyzed as indicated for CDK2. (C) Left , total cell lysates from HeLa cells treated with DMSO or 1 ug/ml of TM for 16 h were immunoblotted for the indicated endogenous proteins. Right , quantification of endogenous Emi1 mRNA levels in HeLa cells treated with DMSO or 1 ug/ml of TM for 16 h, measured by SYBR-green qRT-PCR.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
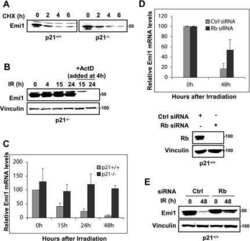
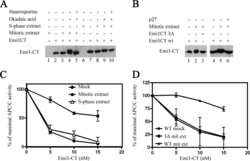
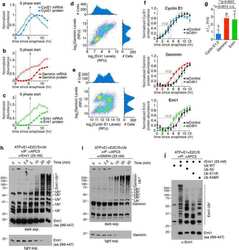
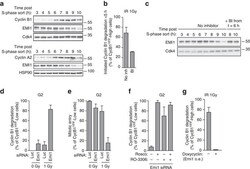
- Figure 3 Emi1 acts to maintain recovery competence in G2 cells. ( a ). Western blot showing EMI1, Cyclin A2 and Cyclin B1 protein levels at the indicated time following early-S phase sort. Representative blots of two independent experiments are shown. ( b ) Quantification of antephase cells (selected as in Fig. 2a ) that started direct Cyclin B1 degradation within 5 h from IR 1 Gy. BI was added 2,5 h before IR. Mean+-s.e.m. of three independent experiments. ( c ) Western blot showing Emi1 protein levels following early-S phase sort in the absence or presence of BI from 6 h after the sort. ( d , e ) Cyclin B1 degradation ( d ) and mitotic entry ( e ) within 10 h after 1 Gy IR was analysed in RPE CCNB YFP G2 cells (selected as in Fig. 2c ) partially depleted for EMI1. Mean+-s.d. of three independent experiments. ( f ) Cdk1 (RO-3306), Cdk2 (Roscovitine) or both were inhibited in undamaged RPE CCNB YFP cells partially depleted of EMI1. Direct Cyclin B1 degradation of cells in G2 phase at the moment of Cdk inhibition was analysed. Mean+-s.d. of three independent experiments. ( g ) EMI1 Turq overexpression was induced in RPE CCNB YFP cells 2 h before IR 1 Gy using doxycycline. Direct Cyclin B1 degradation was scored in antephase cells (selected as in Fig. 2a ). Mean+-s.e.m. of three independent experiments.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
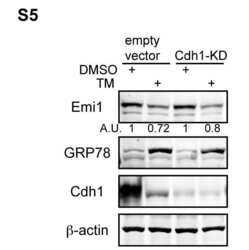
- Figure 1 APC/C Cdh1 is activated under ER stress conditions. (A) Left , HeLa cells were treated with DMSO or 2.5 ug/ml of tunicamycin (TM) for 8 h. Total cell lysates were immunoblotted for the indicated endogenous proteins. GRP78 served as a marker of ER stress. Right , HeLa cells were harvested at the indicated times after addition of DMSO or 2.5 ug/ml of TM. Total cell lysates were immunoblotted for the indicated endogenous proteins. HSP90 was used as a loading control. (B) HeLa cells were treated with DMSO or 1 ug/ml of tunicamycin for the indicated times. Immunoblots , total cell lysates were immunoblotted for the indicated endogenous proteins. Graphs , transcript levels as measured by SYBR-green qRT-PCR and protein levels as quantified by LiCOR-Odyssey software on immunoblots are compared for the indicated proteins. All measurements were normalized to the DMSO-0 h sample with relative value of 1. (C) HeLa cells were transfected with pSUPER (empty vector) or pSUPER-Cdh1-shRNA (Cdh1-KD). 24 h after transfection, cells were treated with DMSO, 1 ug/ml of TM alone, or 1 ug/ml of TM plus MG-132 (2 or 5 uM) for 16 h. Total cell extracts were immunoblotted for the indicated endogenous proteins.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
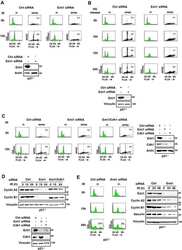
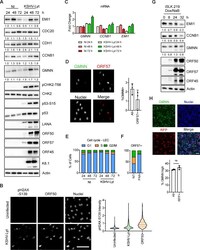
- FIG 2 KSHV lytic replication occurs in G 1 while the APC/C is active. (A) LECs were either infected with KSHV-Lyt using 4 IU/cell or uninfected. Cells were lysed at the indicated times and subjected to immunoblot analysis for the indicated proteins. Numbers below the blots indicate actin-normalized relative band intensity. (B) LECs were infected as in (A) or uninfected. At 48 h p.i., cells were stained with anti-ORF50 and anti-pH2AX-S139 antibodies. Quantification of the pH2AX-S139 intensity per nucleus is shown in the violin plot ( n = >850 cells; scale bar = 100 mum). (C) LECs were infected as in (A) and harvested at the indicated time points. The mRNA levels of target genes were analyzed by RT-qPCR by using Actin mRNA as an internal control ( n = 2 biological replicates; error bars show SD). (D) LECs were infected as in (A) and at 48 h p.i. stained with anti-GMNN and anti-ORF57 antibodies. The graph shows the percentage of the GMNN-positive cells in the total population and in the ORF57-positive population ( n = 3 biological replicates; >500 cells/biological replicate were analyzed). (E) LECs were either infected with KSHV-Lyt as in (A) or uninfected. The cell cycle was analyzed by flow cytometry after PI staining. The graph shows the proportion of cells at each phase of the cell cycle. (F) LEC were infected as in (A), treated where indicated with PAA (100 mug/ml) and at 48 h p.i. stained with PI and antibodies against ORF57. The graph shows the portion of ORF57-positive c
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
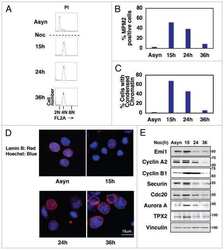
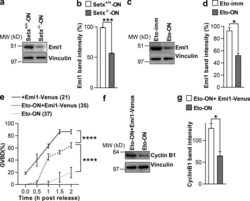
- Figure 7. Reduced Emi1 levels inhibit M-phase entry following DNA damage. (a and b) Representative immunoblot (a) and quantification of Emi1 band intensities (b) in Setx +/+ -ON and Setx -/- -ON oocytes (50 oocytes per lane). (c and d) Representative immunoblot (c) and quantification of Emi1 band intensities (d) in Eto-immediate and Eto-ON oocytes (50 oocytes per lane). (e) GVBD rates for oocytes microinjected with EMI1-Venus cRNA, Eto-ON oocytes microinjected with EMI1-Venus cRNA, and uninjected Eto-ON oocytes. Note that ~20% of oocytes overexpressing Emi1-Venus overcame IBMX-mediated inhibition. ( f and g ) Representative immunoblot (f) and quantification of cyclin B1 band intensities (g) in Eto-ON oocytes with or without Emi1-venus overexpression (80 oocytes per lane). Oocyte numbers are shown in parentheses from a minimum of three independent experiments. Error bars are mean +- SEM. Two-tailed Student's t test (b, d, and g) or two-way ANOVA (e) was used for statistical analysis. *, P < 0.05; ***, P < 0.001; ****, P < 0.0001. In immunoblots, vinculin served as loading control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
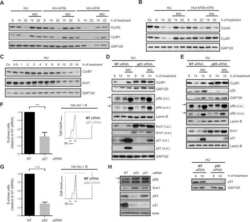
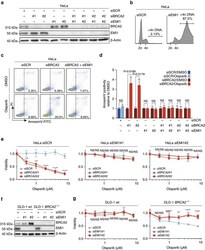
- Figure 6 Mitotic progression promotes PARP-inhibitor cytotoxicity. ( a ) HeLa cells were transfected with indicated siRNAs, and immunoblotting for BRCA2, EMI1 and beta-Actin was performed at 48 h after transfection. Lines next to blots indicate positions of molecular weight markers. ( b ) HeLa cells were transfected with indicated siRNAs for 48 h, and subsequently fixed. DNA content was analysed by flow cytometry. The percentage of cells containing >4n DNA is indicated. ( c ) HeLa cells were transfected with BRCA2 siRNAs or control siRNA (SCR), either alone or in combination with EMI1 siRNAs. After 24 h, cells were replated and treated with olaparib (0.5 muM) for 72 h, after which cells were stained with annexin-V-FITC and propidium iodide, and were analysed by flow cytometry. ( d ) HeLa cells were treated and analysed as for c . Averages and s.d. of three independent experiments are shown. P values were calculated using two-tailed Student's t -test. ( e ) HeLa cells were transfected with two independent BRCA2 siRNAs or control siRNA (SCR), either alone or in combination with siRNAs targeting EMI1. After 24 h, cells were replated and allowed to attach for 3 h. Subsequently, cells were treated with indicated concentrations of olaparib for 72 h, and viability was assessed by MTT conversion. Graphs are representative of three independent experiments, and error bars indicate s.d. of three technical replicates. P values were calculated using two-tailed Student's t -test. ( f , g )
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot ELISA
ELISA Other assay
Other assay